Peroxydisulfuric Acid – Molecular Structure, Bonding Arrangement, Oxidizing Strength, Formation and Decomposition, Industrial Relevance, Biological Considerations, and Educational Insight into Advanced Inorganic Chemistry
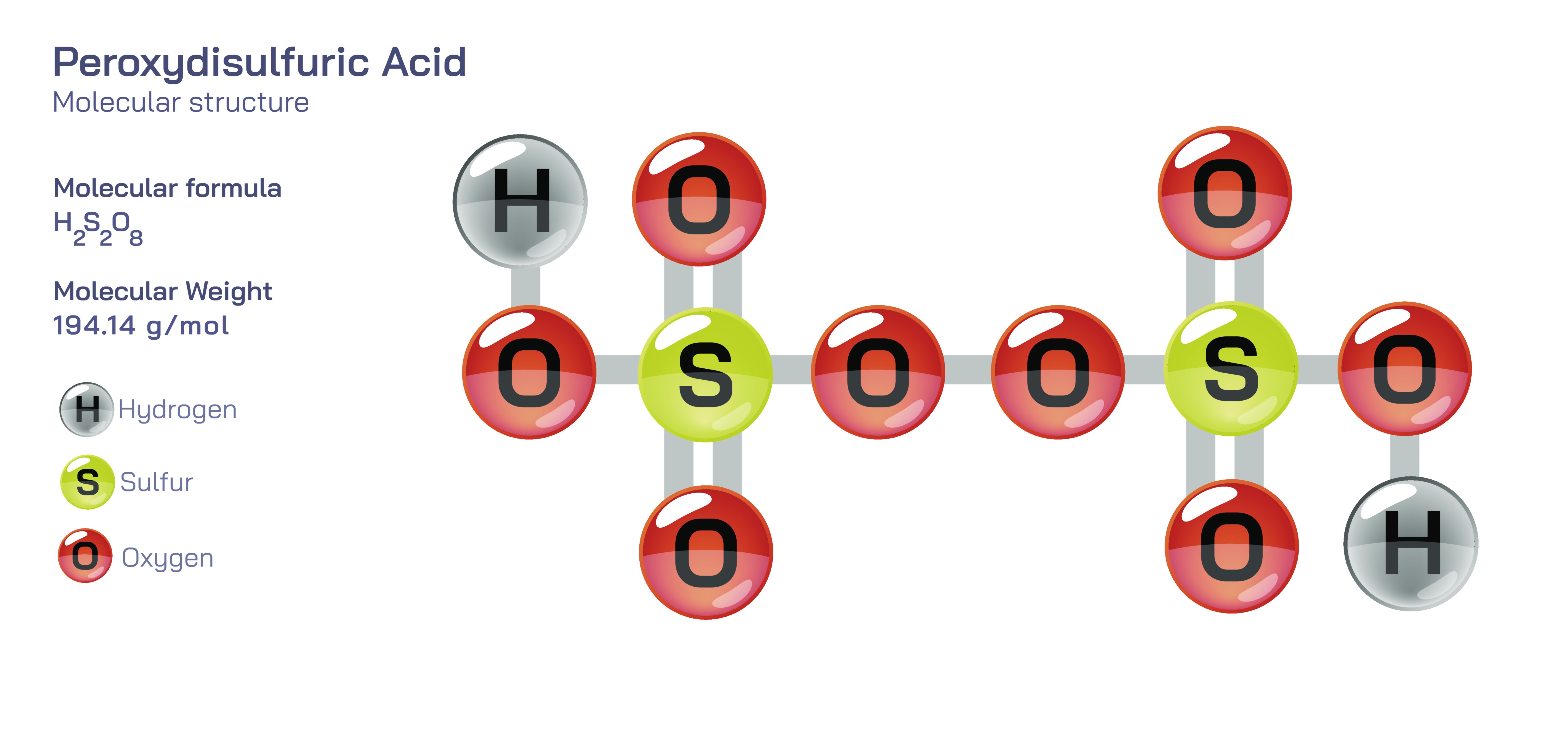
Peroxydisulfuric acid, with the chemical formula H₂S₂O₈, represents one of the most important and conceptually rich oxyacids in inorganic chemistry because its molecular architecture incorporates both the structural features of sulfuric acid derivatives and the additional complexity created by a peroxide linkage. Understanding this acid at a structural and functional level offers students a deeper appreciation for electron distribution, bond energies, oxidation states, and the relationship between molecular structure and chemical reactivity. At its core, peroxydisulfuric acid is composed of two sulfur atoms, each surrounded by four oxygen atoms in a tetrahedral geometry similar to sulfate, but the key distinction is that the two tetrahedra are joined by an –O–O– peroxide bond. Each sulfur atom is double-bonded to two oxygen atoms and single-bonded to one terminal OH group and to one of the oxygen atoms that forms the central peroxide bridge. When visualized three-dimensionally, the structure resembles two sulfate units linked together by a covalent oxygen–oxygen connection that retains the full resonance stabilization characteristic of sulfur–oxygen bonding while also introducing a reactive, energy-rich peroxide bond with unique chemical implications.
The peroxide linkage is the feature that transforms the behavior of peroxydisulfuric acid from that of a typical strong mineral acid into that of a powerful oxidizing agent. In peroxide species, the O–O bond contains stored chemical energy and is thermodynamically prone to break under the right conditions, releasing highly reactive oxygen species that can interact aggressively with other compounds. In the context of peroxydisulfuric acid, this means that proton donation is not the only reaction pathway; the molecule can also participate in strong oxidation of metallic ions, organic compounds, and even other inorganic substances. Sulfur in peroxydisulfuric acid exists in its highest oxidation state (+6), making further oxidation of sulfur chemically impossible, so the oxidative capacity of the molecule originates overwhelmingly from the destabilization and cleavage of the peroxide bond. This detail offers a valuable educational lesson: oxidizing power does not necessarily depend only on the oxidation state of atoms inside a molecule but can arise from the energetic behavior of structural bonds that are inherently short-lived under reactive conditions.
The formation of peroxydisulfuric acid is itself a demonstration of advanced chemical design. It is not found naturally in free form because the peroxide bridge is too reactive to persist without stabilization. Instead, peroxydisulfuric acid is generated industrially or in laboratory settings, often through the electrolysis of concentrated sulfuric acid. During electrolysis, sulfate species in solution undergo oxidation at the anode to form the peroxydisulfate ion (S₂O₈²⁻), which is then protonated to create the acid. The peroxydisulfate ion is relatively stable when paired with counterions such as sodium, potassium, or ammonium, and these salts—known collectively as persulfates—are widely used due to their easier handling and longer shelf life compared with the free acid. The ability to generate peroxydisulfuric acid from simpler chemical building blocks reinforces the principle that structure dictates not only reactivity but also synthesis pathways and storage requirements.
Beyond structural considerations, peroxydisulfuric acid illustrates how molecular behavior transforms across different environments. In solution, and particularly in the presence of heat or catalysts such as silver ions or metal oxides, the peroxide bond can cleave to generate sulfate radicals—highly reactive species that attack organic compounds and initiate chain reactions. These radicals make peroxydisulfuric acid and its salt derivatives important in industrial bleaching, polymer initiation, environmental remediation and oxidative cleaning processes. In wastewater treatment, persulfates derived from peroxydisulfuric acid can break down dyes, hydrocarbons, pharmaceutical residues, and other persistent pollutants through radical attack, leaving behind sulfate ions that are generally non-toxic and environmentally benign at low concentrations. This selective breakdown of contaminants depending on bond arrangement, rather than on brute chemical force, demonstrates the precision that modern oxidation chemistry can achieve.
Peroxydisulfuric acid also teaches a crucial lesson in laboratory safety and the need for understanding chemical structure before handling powerful reagents. Its oxidizing ability means the acid must be stored away from organic materials, reducing agents, combustible substances, and certain metals because contact can lead to violent reactions, ignition, or decomposition with release of heat and gas. Additionally, because the peroxide bond becomes more unstable at elevated temperatures, peroxydisulfuric acid should not be heated or concentrated beyond controlled limits. These safety considerations are not external rules but emerge naturally from the molecular design of the compound. When students see how structure translates into hazard, they learn that safe laboratory practice is rooted not in memorized warnings but in chemical understanding.
Biologically, peroxydisulfuric acid is not a reagent compatible with living systems due to its ability to oxidize proteins, lipids, and nucleic acids aggressively. However, its salts have been studied in controlled environments for their potential to disinfect surfaces and break down harmful biological contaminants. In doing so, they highlight the broader concept that toxicity is a matter of dose, exposure, and context rather than an inherent property of every chemical interaction. The useful oxidative power of peroxydisulfuric acid in wastewater purification or polymer chemistry becomes harmful inside biological tissues because the same radical reactivity that destroys pollutants would indiscriminately harm cells.
At a deeper scientific level, peroxydisulfuric acid illustrates several foundational principles that shape modern chemical education. It demonstrates how the introduction of a single new bond type—in this case the peroxide linkage—can dramatically shift chemical strength and reactivity while leaving the rest of the molecular architecture intact. It shows how resonance and electron-withdrawing effects stabilize acids while a neighboring structural element creates an entirely different type of reactivity. It proves that oxidation is a structural phenomenon rooted in electron control and bond rearrangements, not merely a label attached to compounds in textbooks. It reveals how chemical species behave differently in dilute and concentrated states and how solvation helps moderate energetic bonds that would otherwise break violently.
Ultimately, peroxydisulfuric acid serves as an elegant example of the harmony between structure and function in inorganic chemistry. Its tetrahedral sulfur centers reflect classical bonding symmetry, while the peroxide bridge introduces modern oxidative capability. Its acid strength reflects electron distribution and resonance, while its reactive decomposition reflects the energy contained within the O–O linkage. Through this compound, learners see chemistry not merely as a list of formulas but as a system of structural logic that governs how molecules behave, how they are synthesized, how they must be handled, and how they shape both technology and environmental responsibility.