Tartaric Acid – Molecular Structure, Functional Groups, Stereochemistry, Chemical Behavior, Natural Occurrence, Industrial and Biological Relevance, and its Educational Importance in Organic and Biochemical Chemistry
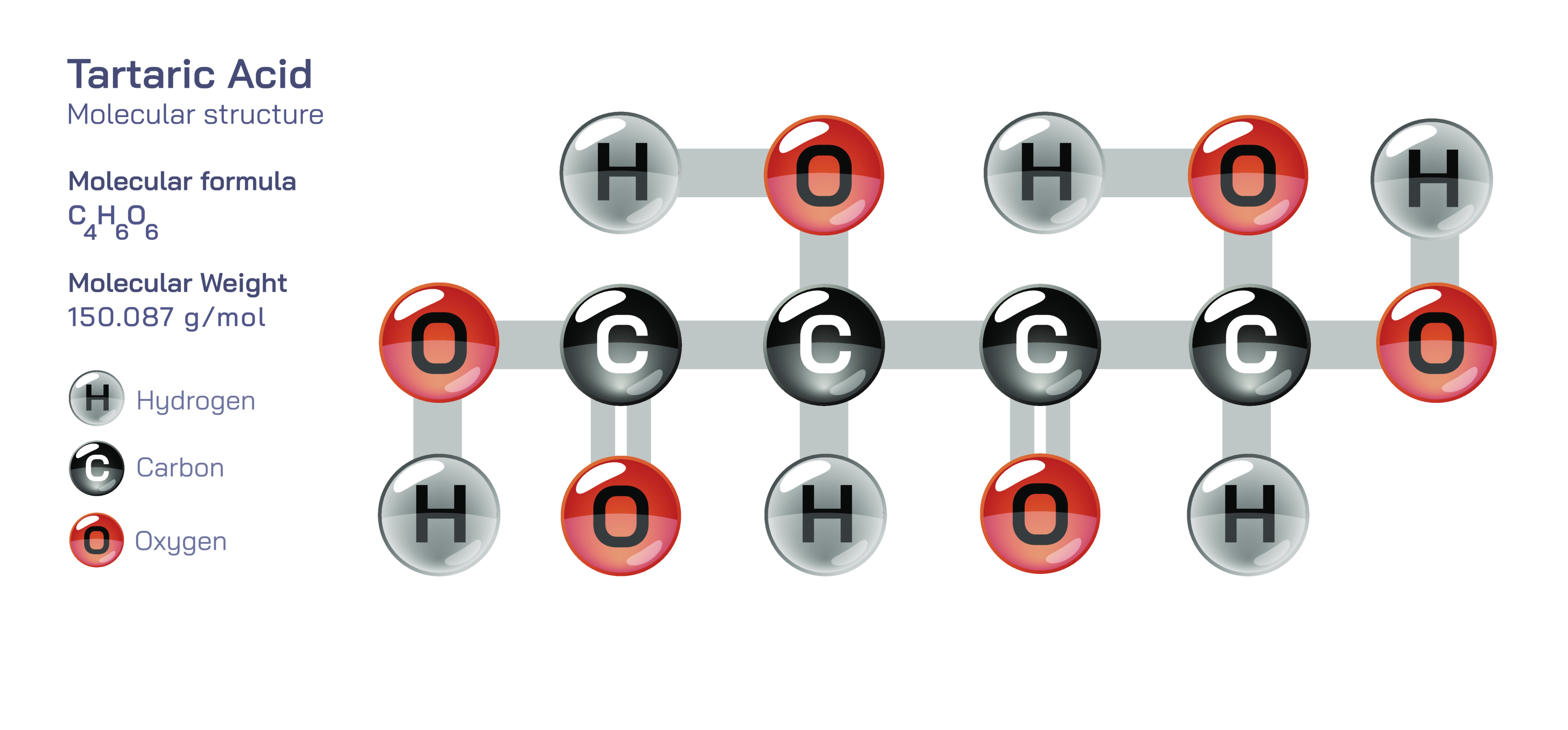
Tartaric acid is a naturally occurring organic dicarboxylic acid widely known for its contribution to the taste, acidity, and preservation qualities of fruits—especially grapes—and for its crucial role in food chemistry, pharmaceuticals, and stereochemistry education. Represented by the molecular formula C₄H₆O₆, tartaric acid is structurally composed of a four-carbon chain bearing two carboxyl groups (–COOH) and two hydroxyl groups (–OH). What makes tartaric acid particularly fascinating is not only this arrangement of functional groups but also the three-dimensional spatial orientation in which they attach. Each of the two central carbon atoms is a chiral center, meaning they each have four different substituents, creating stereoisomeric forms that give tartaric acid a unique place in the development of stereochemistry.
The general structural framework of tartaric acid can be described as HOOC–CH(OH)–CH(OH)–COOH. In this arrangement, the two –COOH groups lie at opposite ends of the molecule, while the two –OH groups are attached to the middle carbons, forming a highly polar and strongly hydrogen-bonding molecule. The presence of both acidic and alcoholic functional groups gives tartaric acid dual reactivity, allowing it to participate in acid–base reactions, esterification, and redox transformations. Its two ionizable carboxyl groups make tartaric acid a diprotic acid, releasing two protons stepwise in aqueous solution. The hydroxyl groups further influence acidity by donating electron density and stabilizing the conjugate base through hydrogen bonding. These structural details explain why tartaric acid has a sharper, cleaner acidity than some other organic acids and why it readily binds to metal ions and participates in complexation reactions.
One of the defining educational themes revealed through tartaric acid is chirality and stereoisomerism. Because the molecule possesses two stereogenic centers, four theoretically possible stereoisomers exist. However, due to molecular symmetry, tartaric acid actually appears in three distinct stereochemical forms:
D-tartaric acid
L-tartaric acid
Meso-tartaric acid
D- and L-tartaric acid are non-superimposable mirror images—enantiomers—and rotate plane-polarized light in opposite directions despite having identical physical properties in most other respects. Meso-tartaric acid, on the other hand, contains two stereocenters but is optically inactive because its internal symmetry cancels out optical rotation. This discovery played a historically significant role in the development of stereochemistry, particularly in the work of Louis Pasteur, who separated tartaric acid crystals based on their optical behavior and demonstrated the existence of molecular chirality—a turning point in the understanding of three-dimensional molecular structure.
Tartaric acid’s functional groups explain its solubility and reactivity patterns. Its multiple hydroxyl and carboxyl groups support extensive hydrogen bonding with water, giving tartaric acid excellent solubility. Hydrogen bonding within and between molecules also stabilizes crystal formation and influences melting point. The –COOH groups allow tartaric acid to neutralize bases, form salts (tartrates), and serve as a buffering agent in solutions. The hydroxyl groups contribute to reactivity such as ester formation under acidic or catalytic environment, allowing tartaric acid to act as a building block in synthetic organic chemistry.
Because tartaric acid contains both hydroxyl and carboxyl functionalities, it has the ability to chelate metals by coordinating multiple binding points to metal ions. This underlies its use in analytical chemistry and metal complex stabilization, where it prevents unwanted precipitation of metal hydroxides during reactions. The popular Fehling’s solution used historically for detecting reducing sugars relies on tartrate ions to keep copper(II) ions dissolved while permitting reduction reactions to occur.
In nature, tartaric acid appears most prominently in grapes, which is why it is central to the chemistry of wine and fruit preservation. It contributes to the acidic profile of grape juice and influences flavor, texture, and shelf life. During wine fermentation and storage, tartaric acid forms potassium hydrogen tartrate (cream of tartar), which precipitates slowly and can crystallize over time. This crystalline salt is collected and purified for use in baking powders, stabilizers for whipped egg whites, and pH-regulating additives.
In pharmaceuticals and food science, tartaric acid serves multiple functions. Its acidity and pleasant sourness make it a flavoring and acidulant. Its buffering and chelating properties aid in formulation stability. It is also used as a chiral resolving agent in organic synthesis, helping chemists separate enantiomers in racemic mixtures. This application reinforces the link between molecular shape and practical separation techniques, demonstrating how physical interactions—such as binding and crystallization—depend on stereochemistry.
In industrial chemistry, tartaric acid appears in silver mirror reactions, cleaning formulations for metal surfaces, setting agents for gypsum and cement, and complexing components in electroplating processes. This wide applicability emerges from a single unifying concept: functional groups determine the interaction of molecules with their environment. Each part of the tartaric acid structure—carboxyl groups, hydroxyl groups, stereocenters—contributes a different dimension of chemical behavior.
Educationally, tartaric acid serves as an exemplary molecule because it highlights nearly every important principle of organic chemistry—functional group chemistry, chirality, optical activity, hydrogen bonding, intermolecular interactions, acid–base behavior, solubility, metal chelation, and biomolecular relevance. Its connection to real-world products—from fruit to wine to pharmaceuticals and food preparations—makes it easier for learners to relate abstract molecular properties to everyday experiences. It demonstrates that chemistry exists not only in laboratories but in plants, foods, medicines, and materials encountered constantly in daily life.
Ultimately, tartaric acid teaches a central lesson of molecular science: the way atoms are arranged in space determines how a molecule behaves. What looks like a simple organic compound becomes a multifunctional molecular system whose structure governs acidity, solubility, taste, industrial utility, and biological compatibility. By exploring tartaric acid in depth, students gain not only chemical knowledge but also insight into the elegant logic that underlies organic chemistry, natural product chemistry, and biochemical interactions in the world around us.