Paracetamol – Molecular Structure, Bonding Characteristics, Functional Groups, Pharmacological Action, Metabolism, Therapeutic Role, Toxicity Mechanisms, and its Central Position in Modern Medicine
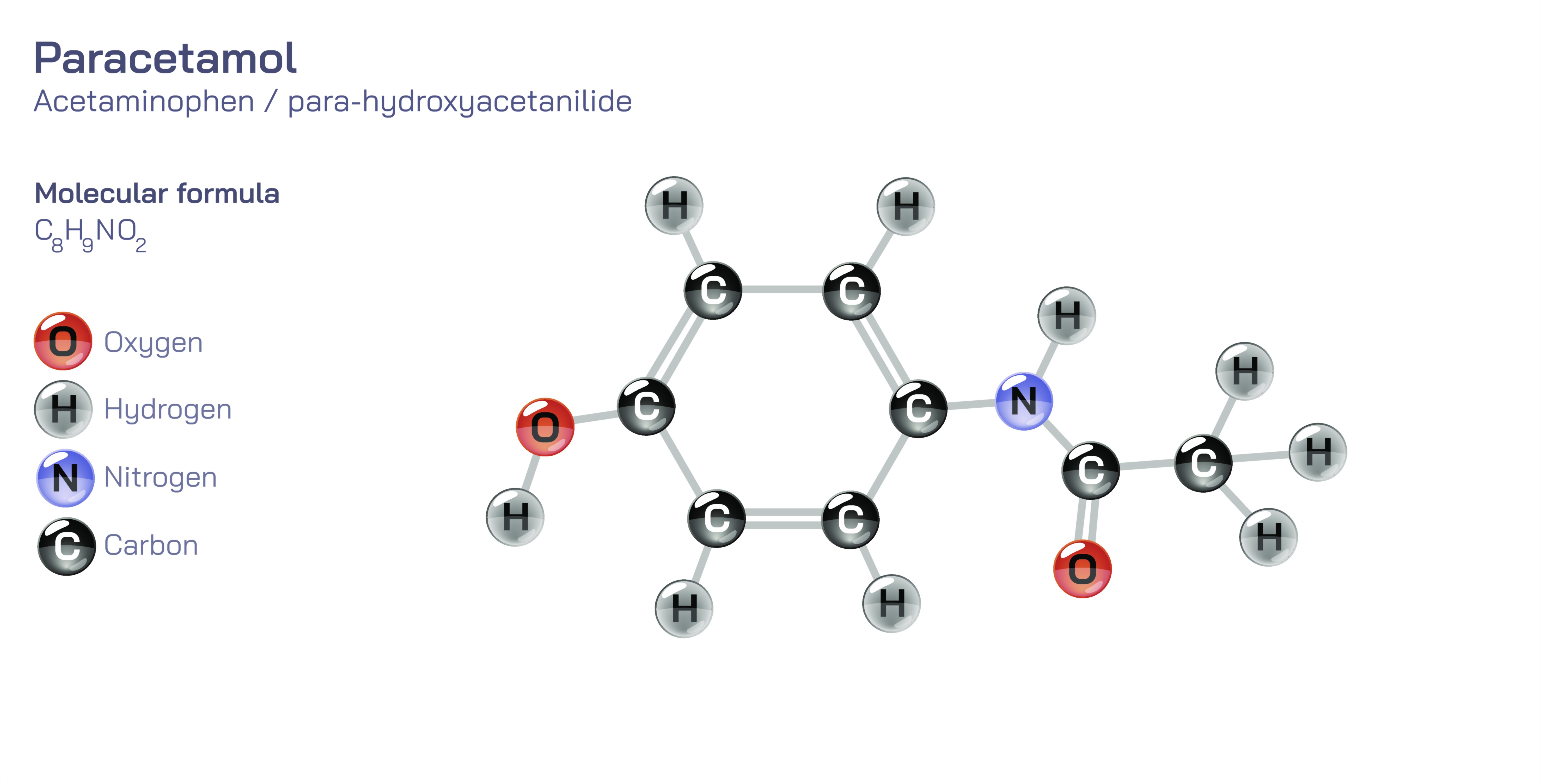
Paracetamol, also known internationally as acetaminophen, is one of the most widely used pain-relieving and fever-reducing medications in the world, and its molecular structure explains much of its behavior in the body, its clinical effectiveness, and even the risks associated with overdose. Chemically represented by the formula C₈H₉NO₂, paracetamol is classified as an aromatic amide-phenol compound. This means its carbon skeleton is built around a benzene ring that carries two distinct functional groups: a hydroxyl group (–OH) positioned on one carbon and an amide group (–NHCOCH₃) attached to another. These functional groups sit para- to each other on the benzene ring, giving the molecule both stability and directional reactivity. In two-dimensional structural diagrams, paracetamol appears as a flat aromatic hexagonal ring with an –OH group and an acetanilide-derived amide group branching off at opposite edges. In three-dimensional space, it still maintains a mostly planar shape because the aromatic ring and associated functional groups support electron delocalization and resonance, which allow the molecule to remain chemically stable while biologically active.
The phenolic hydroxyl group (–OH) plays an important role in paracetamol’s biochemical identity. Although phenols are often strongly acidic and reactive, the acidity of paracetamol’s phenolic hydrogen is reduced by the electron-withdrawing effect of the neighboring amide group. This balance makes paracetamol less irritating to the stomach lining when compared with aspirin or other salicylates, one of the key reasons it became popular for over-the-counter pain and fever control. Meanwhile, the amide group (–NHCOCH₃) influences how the molecule interacts with enzymes and how it is metabolized by the liver. Amides are generally stable structures, and paracetamol’s amide bond helps the drug remain intact long enough to circulate through the bloodstream and reach its target tissues. The para-substitution pattern across the benzene ring is another structural feature that determines pharmacological behavior. Aromatic chemistry shows that the location of substituents on the ring drives electron distribution and determines how the molecule fits into biological receptor sites. In paracetamol, this arrangement creates a shape and charge distribution well suited for modulating central nervous system pathways that regulate pain and temperature.
Despite being structurally related to anti-inflammatory compounds such as aspirin, paracetamol is not considered a traditional nonsteroidal anti-inflammatory drug (NSAID) because it lacks significant anti-inflammatory effects in most body tissues. This difference arises directly from its molecular action. Paracetamol is believed to inhibit the cyclo-oxygenase (COX) enzyme system, which is responsible for converting arachidonic acid into prostaglandins—chemical mediators involved in pain, fever, and inflammation. However, unlike NSAIDs that block COX activity strongly in peripheral tissues where inflammation occurs, paracetamol exerts comparatively weak COX inhibition outside the central nervous system. Research suggests that it works most effectively in the brain and spinal cord, where COX activity is influenced by peroxide levels and neurotransmitter states, allowing paracetamol to reduce pain signaling and lower fever through hypothalamus-mediated thermoregulation. Its molecular structure enables it to cross the blood–brain barrier easily, supporting its focused action in the central nervous system while avoiding the widespread gastrointestinal irritation typical of other COX-inhibiting painkillers.
Paracetamol’s molecular journey through the body continues after it exerts its therapeutic effects, and metabolism provides one of the most clinically important examples of how chemical structure influences bodily safety. The liver metabolizes most of the drug through glucuronidation and sulfation, two conjugation pathways that attach charged biochemical groups to paracetamol to make it more water-soluble for safe excretion through the kidneys. However, a small fraction—typically around five to ten percent—is processed through the cytochrome P450 enzyme system, particularly CYP2E1, into NAPQI (N-acetyl-p-benzoquinone imine). NAPQI is a highly reactive toxic intermediate, and under normal conditions it is neutralized instantly by glutathione, an antioxidant molecule that donates electrons to detoxify NAPQI. The issue arises when paracetamol is taken in excessive amounts or when glutathione stores become depleted due to malnutrition, chronic alcohol use, fasting, liver disease, or certain medications. In those circumstances, toxic NAPQI accumulates and begins to bind to cellular proteins inside liver tissue, leading to cell death, inflammation, and eventually acute liver failure. This dramatic shift—from safe therapy to potentially lethal toxicity—demonstrates how even small metabolic pathways can hold enormous biological significance when molecular load exceeds the body’s processing capacity.
Understanding the molecule also helps explain patterns observed in clinical practice. Because paracetamol is gentle on the stomach and does not interfere with platelets, it is preferred for individuals with peptic ulcers, clotting disorders, or aspirin sensitivity. Its structure allows safe use during pregnancy and childhood under appropriate dosing, and it is often recommended in fevers caused by viral infections due to its minimal gastrointestinal impact. Yet its central metabolic vulnerability means dosage limits are not flexible—exceeding therapeutic amounts does not increase pain relief but instead redirects metabolism toward the toxic NAPQI pathway. In overdose situations, N-acetylcysteine (NAC) serves as the antidote because it helps replenish glutathione stores and binds toxic metabolites, protecting hepatocytes from irreversible injury. Even this clinical solution relies on molecular logic: NAC succeeds because it chemically intersects with paracetamol’s metabolic pathway, highlighting how therapy, toxicity, and antidote systems are all grounded in molecular structure.
Paracetamol’s solid-state features also reflect its molecular identity. It forms crystalline white powder with characteristic intramolecular and intermolecular hydrogen bonding driven by the phenolic hydroxyl and amide groups. These interactions allow stable tablet formation and predictable dissolution profiles in the stomach and intestine. Its solubility in water is modest but increases in hot solvents or pharmaceutical suspensions, and many pediatric formulations use syrups or dispersible tablets designed to optimize absorption. Its melting point and stability under normal storage conditions contribute to safe distribution, while its chemical compatibility with many formulation excipients supports its wide availability in combination products for cold, flu, and migraine relief.
Paracetamol also reinforces fundamental chemical teaching principles. It shows how functional groups define behavior, how substitution patterns on aromatic rings regulate biological targets, how metabolism transforms molecules in multiple pathways, and how a single molecule can produce both therapeutic benefit and toxic harm depending on dose. In medicinal chemistry, paracetamol exemplifies how drug design requires balancing stability, reactivity, absorption, distribution, metabolism, and excretion. In physiology, it reveals how carefully tuned thermoregulation and pain signaling networks can be modulated through targeted structural interactions with enzymes and receptors. In toxicology, it demonstrates that the liver is both a guardian and a point of vulnerability because lifesaving detoxification reactions become dangerous when overloaded.
Ultimately, paracetamol’s molecular structure underscores a universal rule in pharmacology: there is no such thing as a safe drug—only a safe dose. When understood and used properly, paracetamol provides reliable relief from pain and fever for millions of people worldwide. When misused, the same atom arrangement that supports healing can drive catastrophic liver toxicity. This duality reminds us that biological chemistry operates with precision and consequence. Appreciating paracetamol at the molecular level deepens not only scientific understanding but also respect for the delicate biochemical balance that supports therapeutic success and protects human health.