Ozone – Molecular Structure, Bonding Arrangement, Resonance, Polarity, Chemical Reactivity, Environmental Significance, and Educational Insight into the Protective and Harmful Roles of O₃
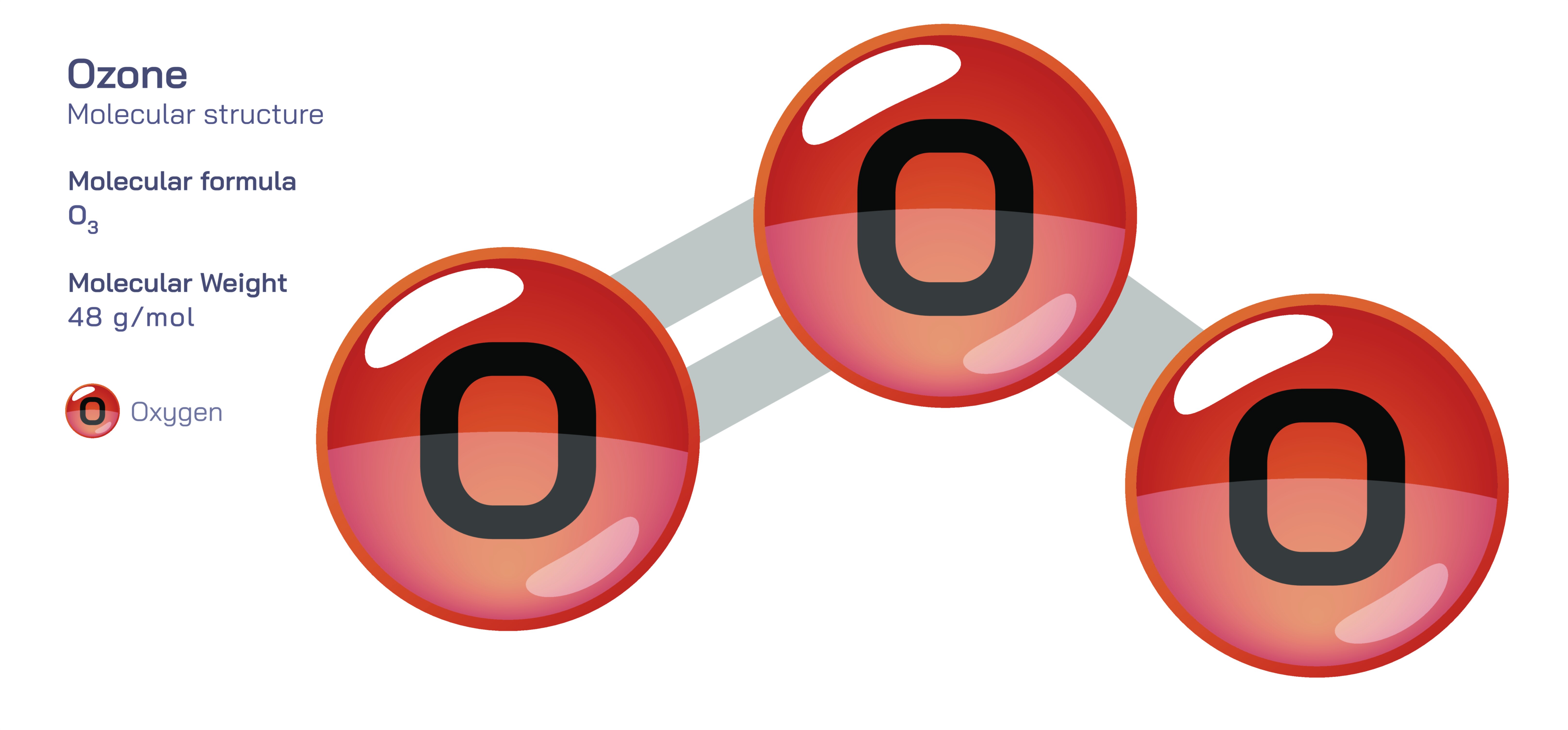
Ozone, represented by the chemical formula O₃, stands out as one of the most fascinating and scientifically important molecules in atmospheric chemistry. It is composed of three oxygen atoms, yet its behavior and structure differ profoundly from the familiar oxygen gas (O₂) that humans breathe. Rather than forming a simple linear chain of three atoms, ozone adopts a bent (angular) molecular shape, which immediately influences its polarity, chemical reactivity, and environmental function. At the center of the molecule lies a unique bonding arrangement in which electrons are not confined to single fixed bonds but are shared across multiple resonance structures. These features make ozone a vivid educational example of how molecular geometry and electron distribution determine physical stability, chemical strength, and planetary importance.
The structure of ozone begins with its atomic arrangement. Although often drawn with alternating single and double bonds between oxygen atoms, the true molecular state is not static. Instead, ozone exhibits resonance, meaning the electrons contributing to pi bonding are delocalized across the molecule. In classical Lewis structures, O₃ can be represented by two resonance forms in which the location of the double bond shifts between terminal oxygen atoms. However, neither of these drawings represents the real molecule perfectly; instead, ozone exists as a resonance hybrid in which both O–O bonds possess intermediate bond order rather than strict single or double bond classification. Experimental measurements confirm that both bonds in ozone have identical lengths—longer than a typical O=O double bond but shorter than an O–O single bond. This equalization of bond character through delocalized electrons provides stability while still preserving significant reactivity, illustrating the fundamental lesson that resonance increases molecular stability without eliminating chemical potential.
The geometry of ozone also contributes crucial insight into molecular polarity and electron distribution. Ozone features a bent shape with a bond angle of approximately 117°, slightly less than the ideal trigonal planar angle due to the repulsion caused by lone pairs of electrons on the central oxygen atom. Because this angular structure distributes charge unevenly, ozone becomes a polar molecule, with partial negative charge accumulation near the terminal oxygen atoms and a partial positive charge at the central atom. This polarity not only influences intermolecular interactions but also explains why ozone is more reactive than O₂. The uneven electron distribution makes ozone an electrophilic molecule capable of attacking electron-rich species. In everyday observations, this strong oxidative power enables ozone to break down organic and inorganic compounds rapidly, making it useful for sterilization, water purification, and industrial oxidation reactions while also posing risks when inhaled in high concentrations.
Beyond molecular structure, ozone plays a pivotal environmental role because it exists in two distinct atmospheric regions where it has contrasting effects on life. In the stratosphere, ozone forms the ozone layer, a protective shield that absorbs most of the Sun’s harmful ultraviolet (UV) radiation—particularly UV-B rays that can damage DNA and disrupt ecosystems. The formation and breakdown of stratospheric ozone operate through a delicate photochemical cycle driven by solar radiation. When UV light strikes an O₂ molecule, it can split it into two reactive oxygen atoms, each of which can then combine with O₂ to form ozone. Ozone then absorbs additional UV light and breaks back down to O₂ and atomic oxygen, maintaining a dynamic equilibrium. This cycle transforms ozone into a protective barrier that allowed life to flourish on land rather than remaining restricted to aquatic environments.
At the same time, ozone in the troposphere—the air we breathe near ground level—has health-damaging effects. It is a major component of photochemical smog, produced when nitrogen oxides (NOₓ) and volatile organic compounds (VOCs) react under sunlight. The same oxidative reactivity that makes ozone useful in sterilization becomes harmful to biological tissues, irritating the respiratory system and causing inflammation in airways. Prolonged exposure can aggravate asthma, reduce lung function, and damage crops and ecosystems. This dual nature of ozone—protective high in the stratosphere yet harmful near Earth’s surface—makes it a powerful educational example of how chemical context determines whether a substance is beneficial or dangerous. It demonstrates that molecular properties do not inherently define value; environmental placement and concentration shape outcomes.
Ozone’s reactivity also drives important processes in atmospheric chemistry beyond UV absorption. It participates in oxidation of pollutants, regulates concentrations of greenhouse gases like methane, and influences climate dynamics. At the same time, certain human-made chemicals—most famously chlorofluorocarbons (CFCs)—once threatened the ozone layer by catalyzing ozone destruction. When exposed to UV light, CFCs release chlorine atoms that react with ozone, repeatedly breaking O₃ into O₂ while regenerating reactive chlorine capable of continuing the cycle. The discovery of the Antarctic ozone hole led to global policy action, including the Montreal Protocol, which became one of the most successful environmental agreements in history. This story is fundamentally rooted in molecular chemistry: the vulnerability of ozone bonds to chlorine radicals explains a global environmental crisis and demonstrates how understanding electron-level interactions can guide planetary decision-making.
In biological systems, ozone’s reactivity has additional consequences. It oxidizes lipids and proteins in respiratory tissues, explaining why inhalation triggers coughing, chest tightness, and reduced oxygen exchange. Yet controlled medical applications, such as ozone therapy and sterilization of medical equipment, rely on the same oxidative power to kill microbes and destroy contaminants. This reinforces the chemical principle that molecular behavior is not inherently beneficial or harmful—it is dependent on how and where it is applied.
In industrial science, ozone is used for bleaching, odor removal, wastewater treatment, and disinfection. Its oxidative strength enables it to break down complex organic molecules that resist ordinary chemical treatment. However, its instability also demands careful handling since ozone decomposes back to O₂ spontaneously, releasing energy and generating free radicals. The molecule therefore illustrates why chemical engineering must account for reaction rate, energy release, and safety in designing processes that exploit reactive species.
Taken together, ozone demonstrates many of the core lessons of chemistry:
• Molecular geometry governs polarity and reactivity
• Resonance stabilizes electron distribution while maintaining chemical strength
• Environmental context determines whether a molecule is protective or dangerous
• Small-scale electron interactions influence large-scale biological and planetary outcomes
By studying ozone, learners gain insight not only into one molecule but into how chemistry operates across different levels—from atomic bonding to atmospheric cycles and from respiratory physiology to global environmental policy. Ozone’s story teaches that the smallest molecular details can produce enormous consequences for life on Earth, making it a model example of why chemistry is essential to understanding the world and protecting it for the future.