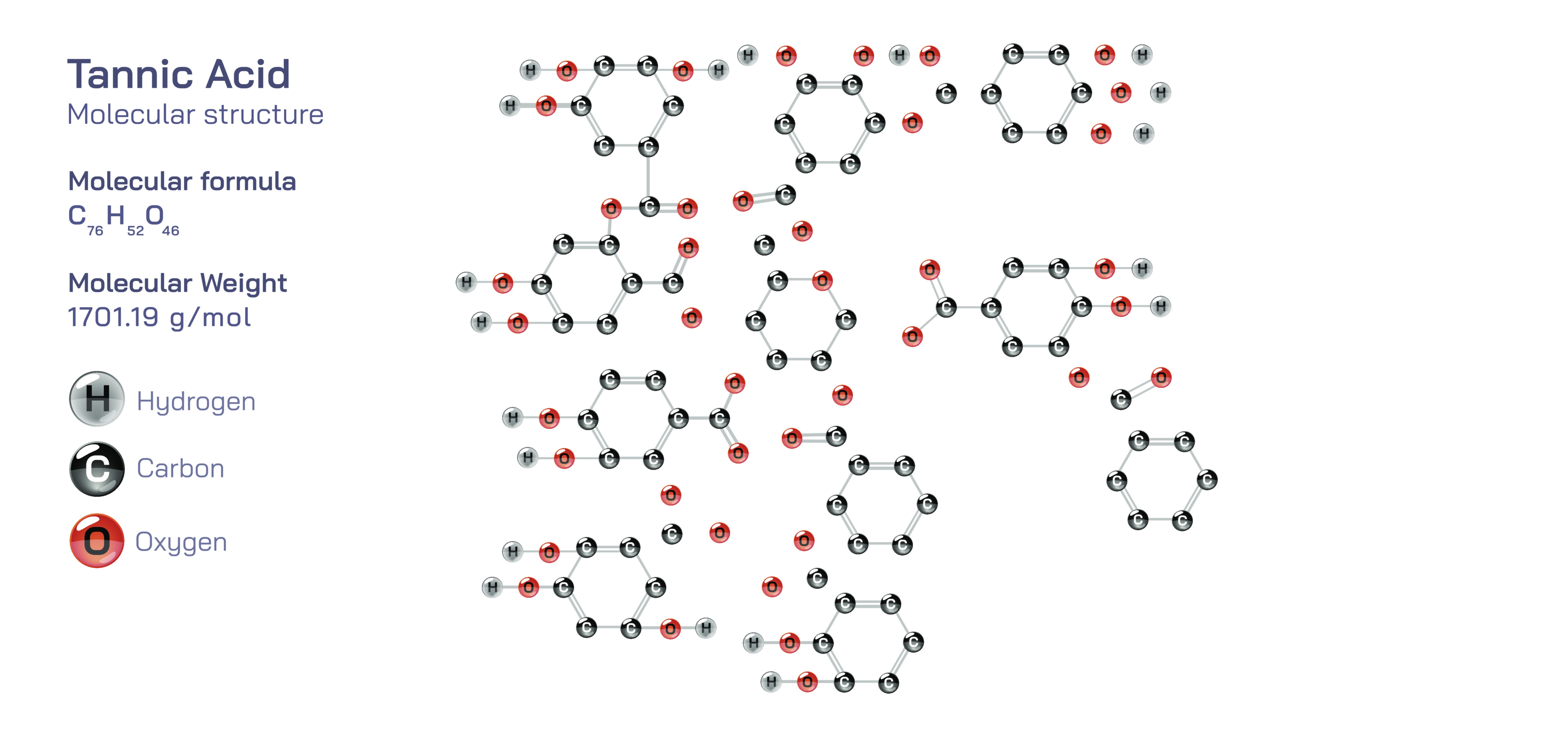
Tannic Acid – Molecular Structure, Functional Groups, Chemical Behavior, Natural Occurrence, Industrial and Biological Uses, and Educational Insight into Polyphenols and Complex Organic Chemistry
Tannic acid is one of the most intriguing and widely discussed organic compounds in both chemistry and biology because it sits at the intersection of plant biochemistry, food science, medicine, industrial processing, and environmental chemistry. Unlike simple laboratory molecules with fixed, compact structures, tannic acid is a large, naturally occurring polyphenolic compound, meaning it is composed of many phenolic units arranged together to form a structurally complex organic network with high molecular weight and extensive hydrogen-bonding capacity. Although the exact structure of tannic acid may vary depending on plant source and extraction method, a commonly referenced structural model shows a central glucose core esterified with multiple gallic acid units, giving the general formula C₇₆H₅₂O₄₆. Each branch of this structure contains phenolic groups (aromatic rings bearing hydroxyl –OH substituents), making tannic acid rich in electron-dense aromatic surfaces and capable of forming strong chemical bonds with proteins, metal ions, and other macromolecules.
The structure of tannic acid can be visualized as a hub-and-spoke arrangement. At the center lies glucose, a six-carbon carbohydrate that acts as a scaffold. Attached to this glucose are typically eight to ten molecules of gallic acid, each connected through ester linkages. Gallic acid itself contains a benzene ring with three hydroxyl groups and one carboxylic acid group. When multiple gallic acid units are bonded to a glucose molecule, the result is a multitiered structure with dozens of phenolic hydroxyl groups distributed across the molecule. This high density of hydroxyl groups gives tannic acid its most defining properties: an ability to donate hydrogen atoms (making it acidic), to accept hydrogen bonds (making it highly interactive with proteins and polysaccharides), and to participate in complex redox and coordination reactions. These features make tannic acid an outstanding educational illustration of how functional groups dictate chemical behavior in large biomolecules.
One of the most characteristic behaviors of tannic acid arises from its protein-binding ability, a property responsible for the word tannin itself, derived from the practice of tanning animal hides to make leather. In aqueous solutions, the numerous hydroxyl groups on tannic acid form stable hydrogen bonds and hydrophobic interactions with protein side chains, especially those containing proline. The result is precipitation or stiffening of the protein network, a mechanism that has been used for thousands of years in leather production. The same behavior explains the astringent taste of tannic acid in foods such as unripe fruits, tea, wine, nuts, tamarind, oak bark, and cacao. Astringency is a sensory effect produced when tannic acid temporarily binds to proteins in the mucous membranes of the mouth, reducing lubrication and causing the dry, puckering sensation familiar to anyone who has tasted strong black tea or red wine.
The chemical behavior of tannic acid is also strongly influenced by its aromatic nature. The benzene rings present throughout the structure make tannic acid highly susceptible to π–π interactions, oxidation, and metal chelation. When tannic acid reacts with metal ions such as iron(III), deeply colored complexes form, which historically led to the development of iron-gall ink, a writing medium used for centuries in manuscripts, historical documents, and artworks. These interactions illustrate clearly how electronic configuration and molecular architecture determine optical properties, bridging organic chemistry and the material sciences. Tannic acid’s metal-binding properties also make it useful in water purification, textile processing, and the development of organic coatings that protect surfaces from microbial growth.
While tannic acid acts as an acid because of its hydroxyl protons, its acidity is very different from mineral acids such as hydrochloric or sulfuric acid. The acidity of tannic acid is driven by resonance stabilization across aromatic rings, allowing hydrogen atoms to dissociate without disrupting the stability of the remaining structure. Individual phenolic groups undergo reversible deprotonation, meaning tannic acid can act both as a proton donor and as a polydentate ligand capable of binding multiple metal centers. This amphoteric and multifunctional behavior makes tannic acid a valuable example when teaching how organic acids derive acidity from resonance rather than from simple hydrogen dissociation.
Biologically, tannic acid plays important roles in the plants that produce it. Because it binds to proteins and interferes with digestion in herbivores and insects, tannic acid acts as a natural defense molecule, deterring animals and pests from feeding on vulnerable tissues such as young leaves, fruits, and seeds. In addition, tannic acid has antioxidant properties, because it can donate electrons to neutralize reactive oxygen species while distributing the resulting charge across its aromatic network. This ability to act as an antioxidant under some conditions and a pro-oxidant under others is a powerful lesson in biochemical context: molecular behavior is not fixed but depends on environment, concentration, and biological system.
In modern medicine and health sciences, tannic acid has been studied for its antimicrobial, antiviral, and anti-inflammatory properties, but it must be used with care. At low concentrations, it can help prevent oxidative damage and microbial growth; at high concentrations, its protein-binding properties can disrupt digestion, damage tissue, or irritate mucous membranes. This duality reinforces the pharmacological principle that the same natural chemical compound can be healing or harmful depending on dose, preparation, and method of use. For example, tannic acid is used externally in some medical and dermatological preparations for burns and skin irritation because of its ability to precipitate proteins and form a protective barrier. Yet ingestion of high amounts without medical supervision can interfere with nutrient absorption, especially iron.
Industrial applications of tannic acid span textiles, adhesives, corrosion inhibitors, natural dyes, wood preservation, wine aging, and pharmaceutical formulations. In environmental chemistry, tannic acid participates in complexation reactions that help remove heavy metals and contaminants from wastewater. In materials science, tannic acid supports the formation of bio-based polymers, nano-coatings, and composite films. These applications demonstrate how a natural biomolecule derived from trees, fruits, and leaves can be adapted across technological fields.
Educationally, tannic acid stands out as an invaluable molecule because it is one of the rare organic compounds that demonstrates the full spectrum of structure–function relationships. Its glucose–gallic acid framework illustrates the connection between carbohydrate chemistry and phenolic chemistry. Its abundant hydroxyl groups teach how hydrogen bonding controls solubility, taste, and biological effect. Its aromatic rings illustrate resonance stabilization and π-electron behavior. Its ability to bind metals bridges organic chemistry with coordination chemistry. Its interactions with proteins illustrate how chemical forces govern biological processes. And its presence in common food and plant sources helps students see chemistry not as an abstract discipline but as a living science woven into everyday experience.
Ultimately, tannic acid highlights the elegant complexity of natural chemistry. It is at once a structural carbohydrate derivative, a phenolic antioxidant, a protein-binding macromolecule, a reactive organic acid, and an industrially useful biochemical material. Its large, branching structure explains its enormous versatility, and its natural occurrence reminds us that biological molecules—including those that create taste, sensation, protection, and preservation—are deeply rooted in the same principles of bonding, resonance, functional groups, and reactivity that govern all of chemistry. Understanding tannic acid allows learners to connect molecular logic to plant biology, food science, medicine, material technology, and environmental science, revealing how profoundly chemistry shapes the natural and human world.