Para-Dichlorobenzene – Molecular Structure, Bonding Properties, Substitution Pattern, Chemical Reactivity, Physical Characteristics, Industrial Applications, Environmental Behavior, and Biological Considerations in Organic Chemistry Education
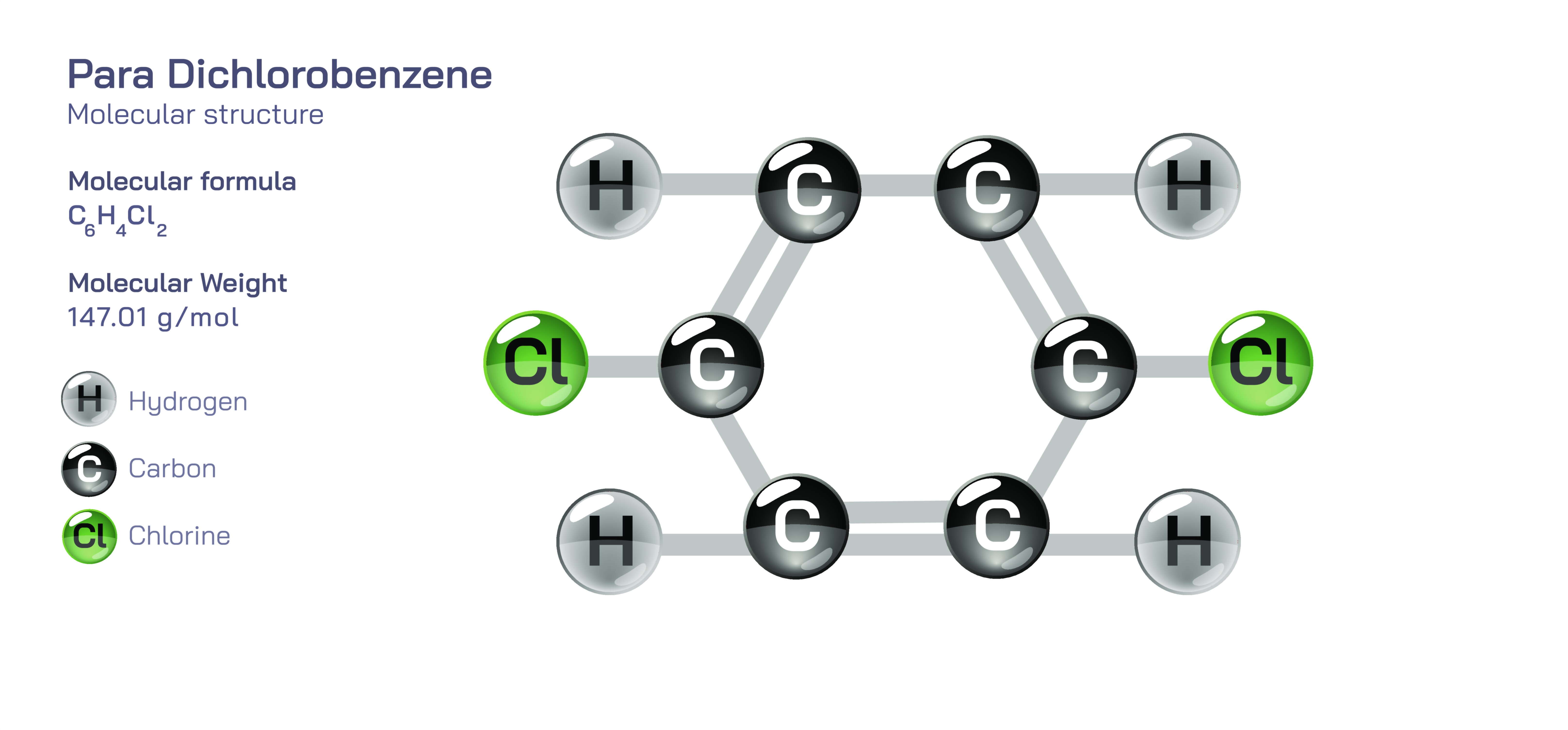
Para-dichlorobenzene is an aromatic organic compound that serves as a rich educational example of how structural arrangement, electron distribution, and substitution patterns influence the physical and chemical properties of molecules. Represented by the molecular formula C₆H₄Cl₂, it belongs to the dichlorobenzene family in which two chlorine atoms are substituted onto a benzene ring. What distinguishes para-dichlorobenzene from its ortho and meta structural isomers is the substitution pattern: the two chlorine atoms occupy opposite positions on the benzene ring, specifically the 1,4-positions. This symmetrical para configuration dramatically influences its melting point, molecular polarity, intermolecular interactions, and reactivity relative to the other isomers. The structure features a six-membered conjugated aromatic ring stabilized by delocalized π-electrons and two carbon–chlorine bonds projecting outward at opposite ends. The benzene core retains aromaticity because each carbon contributes an electron to the shared π-system, preserving the cyclic resonance stabilization responsible for benzene’s chemical behavior. The C–Cl substituents, though strongly electronegative, do not break aromaticity but modify electron distribution across the ring, creating a characteristic reactivity profile that blends electron-withdrawing substituent effects with aromatic resonance continuity.
This symmetrical arrangement is key to understanding why para-dichlorobenzene is uniquely different from its isomers not only academically but practically. While ortho-dichlorobenzene and meta-dichlorobenzene exist as oily liquids under room-temperature conditions, para-dichlorobenzene is a solid that forms crystalline white flakes. The reason lies in molecular symmetry, which enhances packing efficiency in the solid state. Because the chlorine atoms lie opposite each other rather than adjacent, para-dichlorobenzene molecules align neatly in crystal lattices, generating a relatively high melting point and stable solid phase. This makes the compound easier to store, transport, and shape into pellets, tablets, or blocks. At the molecular level, chlorine’s polarizability and the flat, rigid aromatic ring also contribute to London dispersion forces that maintain solid cohesion despite the molecule not being strongly polar. The compound sublimes readily, meaning it transitions from solid to vapor without passing through a liquid phase. This volatility is not incidental—it results from the balance between intermolecular attractions strong enough to support crystallinity but weak enough to allow vapor formation under ordinary temperatures.
Chemically, para-dichlorobenzene demonstrates typical electrophilic aromatic substitution patterns. The substituent chlorine atoms are electron-withdrawing by induction but electron-donating by resonance, generating a complex directing influence on incoming reactants. In general, the presence of the two chlorine atoms deactivates the ring toward electrophilic substitution relative to benzene because the inductive electron-withdrawing effect reduces electron density. However, resonance donation influences orientation, meaning any introduced electrophile is directed primarily to the ortho and para positions relative to each chlorine substituent, though steric factors and reduced reactivity decrease the rate. The stability of the carbon–chlorine bond and aromatic resonance prevent the compound from undergoing nucleophilic aromatic substitution easily under mild conditions. Understanding these effects helps students grasp how substituents modify aromatic ring reactivity not only through electron donation or withdrawal but through spatial configuration and resonance distribution.
Para-dichlorobenzene is historically known for its widespread industrial and household applications. The most recognizable use is as the active ingredient in moth repellents and deodorizer blocks, where its volatility enables continuous slow release of vapor that discourages insects and suppresses odor-causing microbial growth. Its sublimation behavior makes it popular in urinal cakes, closet blocks, and storage pest control formulations. In these contexts, the molecule does not act through a corrosive or toxic contact mechanism but rather through atmospheric dispersion in confined spaces. Because it vaporizes steadily at room temperature, it creates a protective chemical atmosphere in enclosed locations. Beyond household products, para-dichlorobenzene has roles in industrial chemistry as an intermediate in dye manufacturing, polymer processing, and specialty chemical synthesis. Its stability, ability to dissolve greases and waxes, and ease of sublimation also make it useful in certain cleaning and deodorizing applications.
However, the same volatility that makes para-dichlorobenzene convenient also raises concerns in environmental and health contexts. Once released into air, the vapor can persist if not diluted or ventilated. Although ingestion or skin absorption is rare in everyday use, prolonged inhalation exposure can irritate mucosal membranes and affect the liver and central nervous system in high concentrations. The compound is classified in many regions as a potential carcinogen under chronic conditions, prompting gradual reduction and regulation of household products that depend on long-term sublimation in unventilated spaces. In the environment, para-dichlorobenzene tends to partition into air rather than water but can adhere to soil particles and biodegrade slowly depending on microbial populations. Understanding its environmental fate helps students appreciate how molecular properties translate into ecological consequences beyond laboratory chemistry.
Para-dichlorobenzene also provides insight into isomeric chemistry and how subtle structural changes—shifting substituents around the same ring—produce dramatically different outcomes. All three dichlorobenzene isomers share identical molecular formulas and masses, yet their physical states, melting points, toxicological profiles, odors, and industrial applications diverge widely. The para isomer’s symmetry, crystalline packing ability, vapor pressure, and controlled sublimation make it uniquely suited for solid deodorizing and pest-repelling devices. The ortho isomer is a common solvent and degreasing agent, while the meta form is used primarily in chemical synthesis. This variation reinforces the scientific lesson that structure determines function with extraordinary precision, and that even when chemical formula remains constant, arrangement governs identity.
Educationally, para-dichlorobenzene illustrates nearly every core concept of organic molecular chemistry: aromatic resonance, inductive and resonance effects of halogens, substitution directivity, phase behavior, symmetry in molecular design, intermolecular forces, solid-state packing phenomena, and the practical relationship between chemistry, industry, and safety regulation. It ties molecular diagrams to real-world contexts, demonstrating that chemical structure guides not only reactivity but also products humans interact with daily. By examining para-dichlorobenzene as both a chemical and an industrial material, students gain not only theoretical understanding but scientific literacy—connecting molecular architecture to applications, benefits, environmental impacts, and responsible handling. The compound thus becomes an important reminder that chemistry influences life at multiple scales, from electron distribution to household technologies and public health outcomes.