Sucrose – Molecular Structure, Bonding Arrangement, Chemical Properties, Digestive Breakdown, Biological Significance, and Educational Insight into Carbohydrate Chemistry
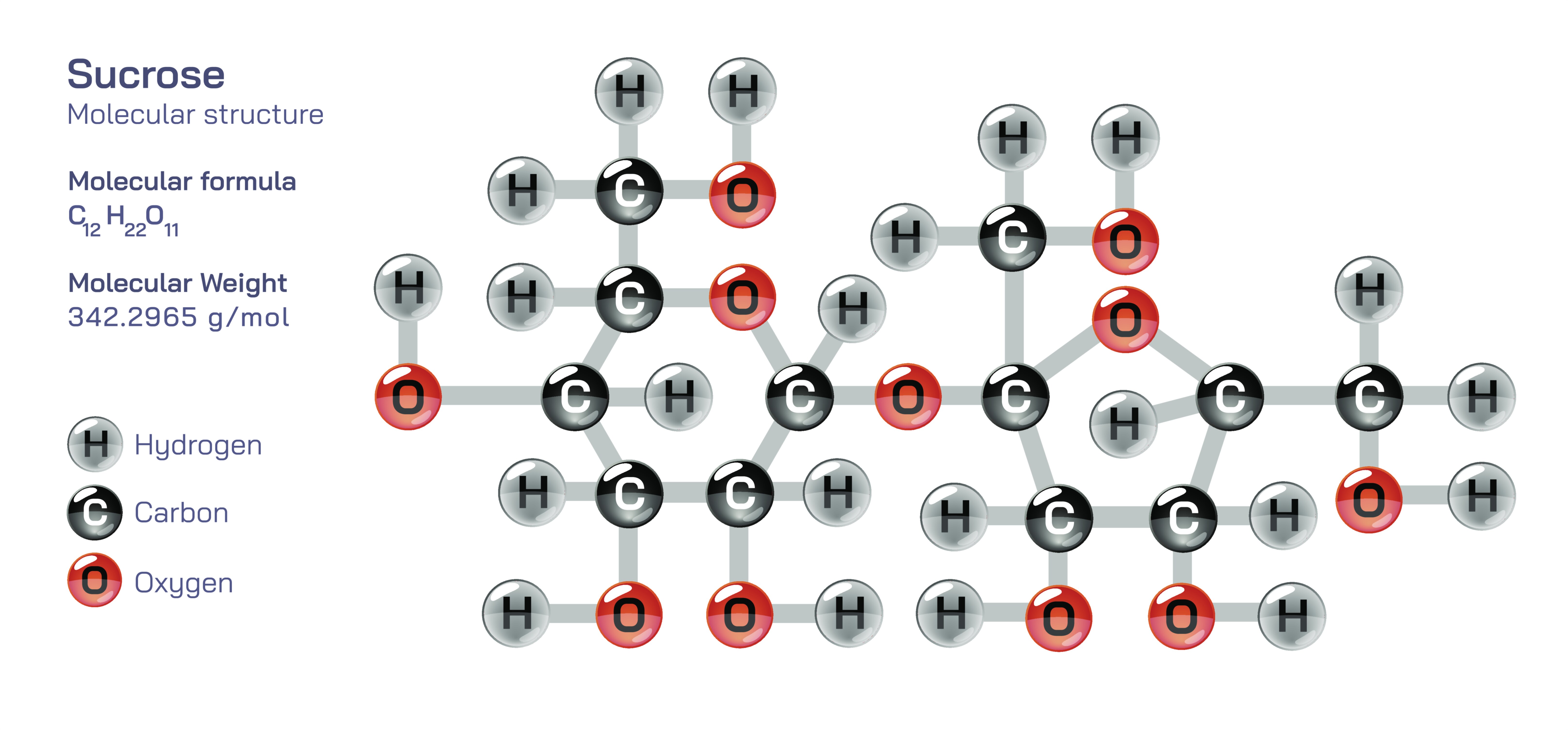
Sucrose is one of the most familiar organic compounds in daily life, yet its underlying chemistry reveals far more than its identity as “table sugar.” Represented by the molecular formula C₁₂H₂₂O₁₁, sucrose is a disaccharide, meaning it is composed of two simple sugar units linked together. These units are glucose and fructose, each of which is a monosaccharide with its own structural and metabolic characteristics. What makes sucrose particularly remarkable from a chemistry education perspective is the nature of the bond that connects these two units and the way this structural feature influences physical behavior, biochemical breakdown, sweetness, and energy production. By studying sucrose at the molecular level, students gain an opportunity to appreciate how carbohydrates store chemical energy, how their properties arise from specific bonding arrangements, and how organic structure influences biological function.
The bonding arrangement between glucose and fructose in sucrose is known as a glycosidic linkage, specifically an α(1→2) β glycosidic bond. In this arrangement, the α-anomer of glucose binds through its carbon-1 atom to the carbon-2 atom of fructose, which is in its β configuration. This link forms when a hydroxyl group from glucose and a hydroxyl group from fructose combine and release a molecule of water, making sucrose a product of a condensation reaction. The result is a covalent bond that stabilizes the interaction between the two monosaccharides. The numbering system in the bond name (1→2) indicates the specific carbon atoms involved, and this precision matters because even slight changes in position or orientation of glycosidic bonds lead to compounds with drastically different biological properties. For example, glucose linked to glucose by a different bond forms maltose, and glucose linked to galactose becomes lactose. These comparisons highlight that not only the building blocks but also the exact nature of linkage determines how sugars behave chemically and biologically.
Structurally, sucrose stands out among disaccharides because both of its anomeric carbons are involved in the glycosidic linkage. In many sugars, one anomeric carbon remains free to interconvert between cyclic and open forms, allowing the sugar to act as a reducing sugar—a compound capable of reducing certain chemical reagents because it exposes an aldehyde group. Sucrose has no free anomeric carbon, meaning it cannot easily convert into an open-chain form and is therefore classified as a nonreducing sugar. This single structural property influences sucrose’s chemical reactivity, making it less prone to participate in browning reactions without prior breakdown. For chemistry learners, sucrose offers an elegant demonstration of how molecular structure influences classification and why carbohydrate reactivity depends not only on composition but on cyclic equilibrium and functional group accessibility.
Because of its glycosidic linkage, sucrose has a distinctive set of physical properties. It is highly soluble in water, thanks to extensive hydrogen bonding between hydroxyl groups on sucrose and water molecules. This solubility explains why sucrose dissolves readily in beverages, forms syrups, and participates in osmosis across membranes. In solid form, sucrose crystallizes easily, forming the granular sugar commonly used in cooking. Its crystalline, colorless structure arises from ordered packing of sucrose molecules and strong intermolecular hydrogen bonding networks. From an educational standpoint, these solubility and crystallization properties illuminate general principles of intermolecular forces: compounds with numerous polar functional groups tend to dissolve well in polar solvents and form hydrogen-bonded lattices in the solid state.
One of the most important features of sucrose in biological chemistry is how the body metabolizes it. Digestion does not use sucrose directly; instead, the enzyme sucrase, located in the small intestine, hydrolyzes sucrose back into glucose and fructose by breaking the glycosidic bond. This hydrolysis is the reverse of the condensation reaction that formed sucrose originally. Once released, glucose enters the bloodstream and fuels cellular respiration, while fructose undergoes processing mainly in the liver before entering metabolic pathways. This breakdown illustrates a basic principle of biochemistry: the human body uses enzymes to convert complex nutrients into simpler metabolites that can enter cellular pathways. The speed and efficiency of sucrose hydrolysis help explain why sucrose produces a quick burst of energy compared with slower-digesting complex carbohydrates.
The presence of both glucose and fructose also contributes to sucrose’s sweetness profile. Although glucose is moderately sweet and fructose is very sweet, sucrose has an intermediate but well-balanced sweetness that appeals to human taste receptors. Sweetness perception depends on the interaction of sugar molecules with taste receptor proteins on the tongue, and sucrose’s molecular geometry matches these receptors in a way that produces a particularly pleasant sweetness. This feature reinforces the chemical idea that sensory qualities arise from molecular structure, not just elemental composition.
Sucrose also plays a key role in food science, an area where chemical concepts intersect with culinary practice. Heating sucrose causes it to melt, and continued heating triggers caramelization, a complex series of thermal reactions that break down sucrose into smaller molecules, producing brown pigments and characteristic flavor compounds. Caramelization is not the same as Maillard browning, which involves reducing sugars and amino acids; because sucrose is nonreducing, it participates in caramelization only after its glycosidic bond breaks under high heat. This distinction helps learners see how molecular structure determines chemical transformation during cooking and why different sugars behave differently in heat.
Sucrose’s properties also extend to industrial and biochemical applications. It is widely used not only as a food sweetener but also as a preservative because high concentrations reduce water activity, slowing microbial growth. In pharmaceuticals, sucrose stabilizes protein-based medicines and serves as a carrier in oral formulations. In plant biology, sucrose functions as the primary carbohydrate transported through the phloem, carrying energy from photosynthetic cells to non-photosynthetic tissues. These varied roles demonstrate how a single molecule can interact with chemical, biological, and physical systems due to its structural versatility.
From an educational perspective, sucrose provides a powerful entry point into carbohydrate chemistry because it allows students to explore multiple interconnected ideas. Through sucrose, learners encounter disaccharide formation, enzyme specificity, stereochemistry of glycosidic bonds, solubility and crystallization, reducing versus nonreducing sugars, energy metabolism, and the relationship between structural features and biological function. The compound shows that chemistry is not merely a collection of formulas but a language that describes structure, behavior, and transformation.
Ultimately, sucrose illustrates one of the most fundamental lessons in organic and biochemical chemistry: molecular structure controls properties and function. Even though sucrose is made of the same elements as many other carbohydrates—carbon, hydrogen, and oxygen—its specific arrangement of atoms determines how it tastes, dissolves, crystallizes, reacts, and nourishes living organisms. By studying sucrose in detail, students develop a deeper understanding of how chemical bonding, stereochemistry, and biological processing intersect, providing a strong foundation for further learning in organic chemistry, nutrition science, biochemistry, and molecular biology.