ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉōØÉÜØɽØÉŁØɽØÉÜØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉłØÉ©ØɦØÉóØÉ£ŌĆōØÉéØÉ©ØÉ»ØÉÜØÉźØÉ×ØɦØÉŁ ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉÆØÉŁØÉ×ØɽØÉ×ØÉ©ØÉ£ØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓, ØÉćØÉ▓ØÉØØɽØÉÜØÉŁØÉóØÉ©Øɦ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓ ØÉÜØɦØÉØ ØÉĆØÉØØÉ»ØÉÜØɦØÉ£ØÉ×ØÉØ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
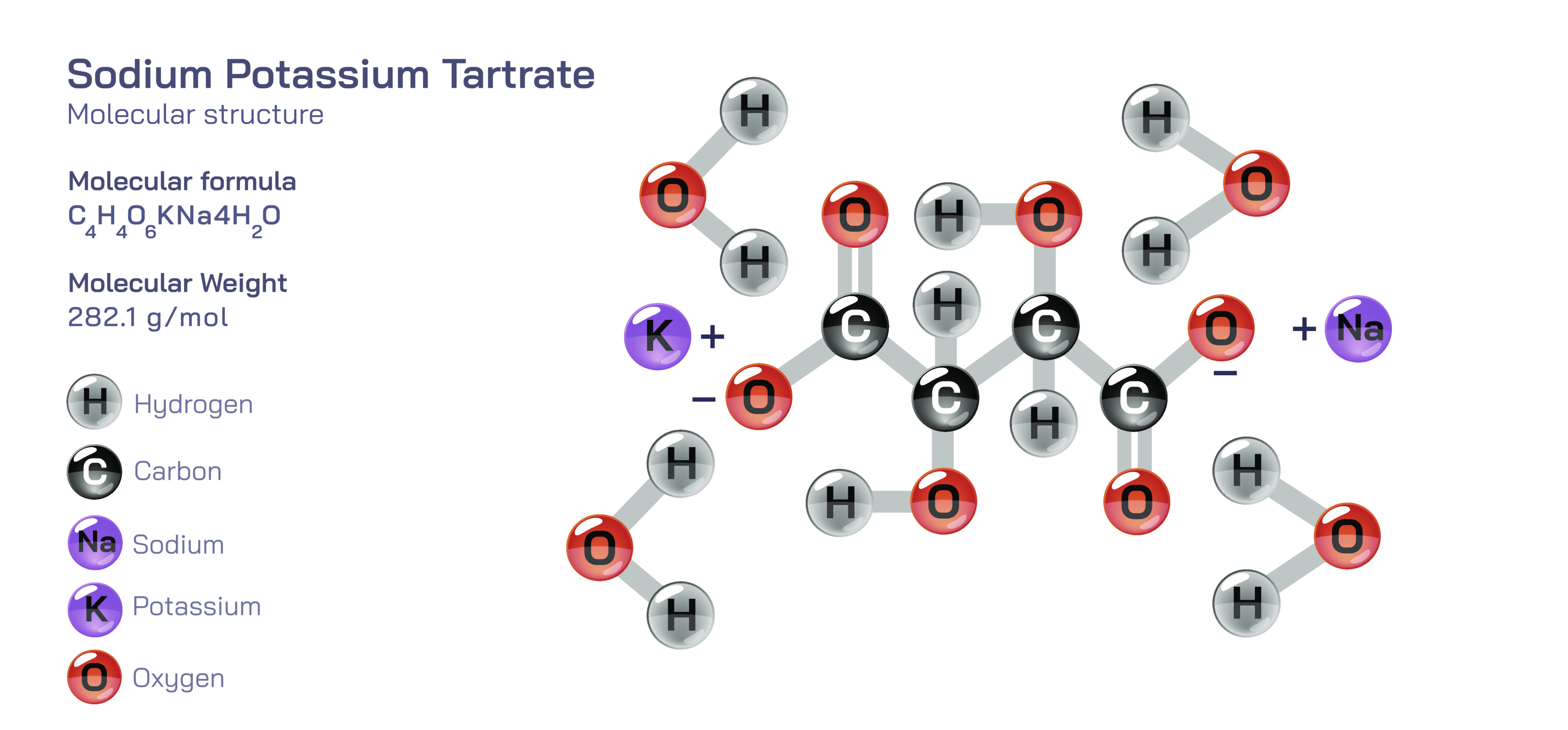
Sodium potassium tartrate, chemically represented as NaK(CŌéäHŌéäOŌéå)┬Ę4HŌééO, is an extraordinary compound that bridges the worlds of organic and inorganic chemistry in a single molecular structure. Known historically as Rochelle salt, it has played crucial roles in crystallography, early electrochemistry, piezoelectric device development and analytical laboratory procedures. What makes sodium potassium tartrate so educationally significant is not only its practical applications, but the way its structure illustrates ionic bonding, covalent bonding, stereochemistry, hydration, chirality, coordination chemistry and intermolecular interactions all at once.
3. The structure of tartrate is uniquely important because the two hydroxyl groups on adjacent stereogenic carbons generate chirality. In its most common form, the tartrate anion exists as the L-(+)-enantiomer, meaning it is optically active and can rotate plane-polarized light. This aspect introduces a crucial chemical lesson: compounds containing sodium and potassium can still be chiral, demonstrating that chirality arises from spatial arrangement of atoms rather than from elemental composition.
The chemical connectivity inside the tartrate ion is entirely covalent, with strong CŌĆōC and CŌĆōO bonds defining its backbone. However, the carboxylate groups carry negative charges, delocalized over two oxygen atoms through resonance. These negative charges allow the tartrate anion to attract and coordinate with positively charged ions. In sodium potassium tartrate, the two cations NaŌü║ and KŌü║ balance the ŌłÆ2 charge of the tartrate ion. Sodium and potassium are spectator ions in terms of organic reactivity, but they provide the electrostatic foundation that allows the compound to exist as a solid. The tartrate ion does not form a double salt randomly; the ionic radii and hydration characteristics of NaŌü║ and KŌü║ match the coordination sites on the tartrate anion, allowing a stable crystalline salt rather than unconstrained mixtures of separate sodium tartrate and potassium tartrate.
The structure becomes even more intriguing when viewed in the solid crystalline state. Sodium potassium tartrate exists as a tetrahydrate, meaning four water molecules are incorporated into each formula unit. These water molecules are not loosely trapped but play a structural role by engaging in extensive hydrogen bonding between tartrate ions, as well as coordinating partially with sodium ions. The combination of ionic attraction between NaŌü║/KŌü║ and tartrate, covalent connectivity inside the tartrate, and hydrogen bonding through lattice water creates a highly ordered crystalline structure. This arrangement reveals the powerful lesson that water can act as an architectural component in solids, not merely a solvent.
One of the most fascinating macroscopic consequences of its microscopic structure is the compoundŌĆÖs piezoelectric property. When a crystal of sodium potassium tartrate is mechanically deformedŌĆöcompressed, stretched or twistedŌĆöit produces an electrical charge across its faces. This phenomenon occurs because the crystal lattice contains asymmetrically arranged ions and dipoles, and mechanical stress causes changes in charge distribution. Conversely, applying an electric field to the crystal causes it to deform. These properties were central to early developments in radio transmitters, sonar, microphones and scientific instrumentation. The piezoelectricity of sodium potassium tartrate demonstrates the deep chemical principle that electrostatic arrangement within a solid can translate into measurable electrical behaviour without electron flow or chemical reaction.
In aqueous solution, sodium potassium tartrate dissociates into NaŌü║, KŌü║ and tartrate ions. Sodium and potassium ions remain hydrated without chemical transformation, but the tartrate ion participates in several important equilibrium processes. Because tartrate retains both hydroxyl and carboxylate groups, it can function as a weak base and a weak acid depending on pH. In strongly acidic solution, tartrate becomes protonated to regenerate tartaric acid; in strongly basic solution, the hydroxyl groups can deprotonate. This behaviour shows that organic ions can display multilevel acidŌĆōbase chemistry, demonstrating the continuum between organic and inorganic acidŌĆōbase systems.
Sodium potassium tartrate plays an especially memorable role in analytical chemistry due to its ability to act as a complexing agent for metal ions. The tartrate ion contains oxygen atoms with lone pairs that can bind to metal cations such as Cu┬▓Ōü║, preventing precipitation of metal hydroxides in alkaline solutions. This property is used in FehlingŌĆÖs solution, a classical test for aldehydes in carbohydrate chemistry. Tartrate stabilizes the copper (II) complex in a basic solution, enabling the controlled reduction of Cu┬▓Ōü║ to CuŌééO by aldehydes. Here, the compound illustrates redox chemistry, complex ion formation and stereochemistry influence on reactivity, all through one laboratory experiment.
Its behaviour on heating also reveals the link between molecular architecture and thermal stability. When heated strongly, sodium potassium tartrate decomposes, losing water of crystallization and eventually breaking down into carbonates of sodium and potassium. Unlike simple inorganic salts, its decomposition does not occur in a single step because covalent and ionic components respond differently to heat. This reinforces the concept that compounds containing both organic and inorganic features can undergo staged decomposition, reflecting the layered bonding responsible for their structure.
In biological chemistry, tartratesŌĆöincluding sodium potassium tartrateŌĆöprovide insight into chiral metabolism and stereoselective biochemical pathways. Enzymes in living organisms often recognize only one enantiomer of tartrate, reinforcing the insight that chirality governs interactions between biomolecules. Although sodium potassium tartrate itself is not commonly used biologically, the behaviour of its tartrate ion mirrors that of many biologically significant hydroxy-dicarboxylate molecules.
From an industrial perspective, sodium potassium tartrate has been used in electroplating, photography, textile processing, pharmaceutical formulations and food chemistry, especially due to its ability to bind metal ions, regulate pH and contribute to solution stability. Its versatility underscores that a compoundŌĆÖs usefulness is rooted not only in reactivity but in its ability to coordinate, dissolve, buffer and stabilize.
Educationally, sodium potassium tartrate is a rare and rich example because it unites multiple fundamental chemical concepts in one structure:
ionic bonding between NaŌü║/KŌü║ and the tartrate ion,
covalent bonding within the tartrate backbone,
chirality and optical activity,
hydration and hydrogen bonding in crystalline solids,
acidŌĆōbase equilibria in aqueous solution,
ligand and complex-forming ability,
piezoelectric effects arising from lattice asymmetry.
Ultimately, sodium potassium tartrate demonstrates one of the most important ideas of chemistry: compounds do not behave according to a single bonding type or rule ŌĆö their properties reflect the interplay of all forces acting at once. A single molecule can contain covalent bonds, ionic interactions, hydrogen bonds, resonance stabilization, stereochemistry, hydration and long-range lattice order. Through this compound, chemistry reveals itself as a unified science in which electrons, geometry and intermolecular forces determine not only microscopic structure but also the macroscopic behaviours that shape technology, biology and materials science.