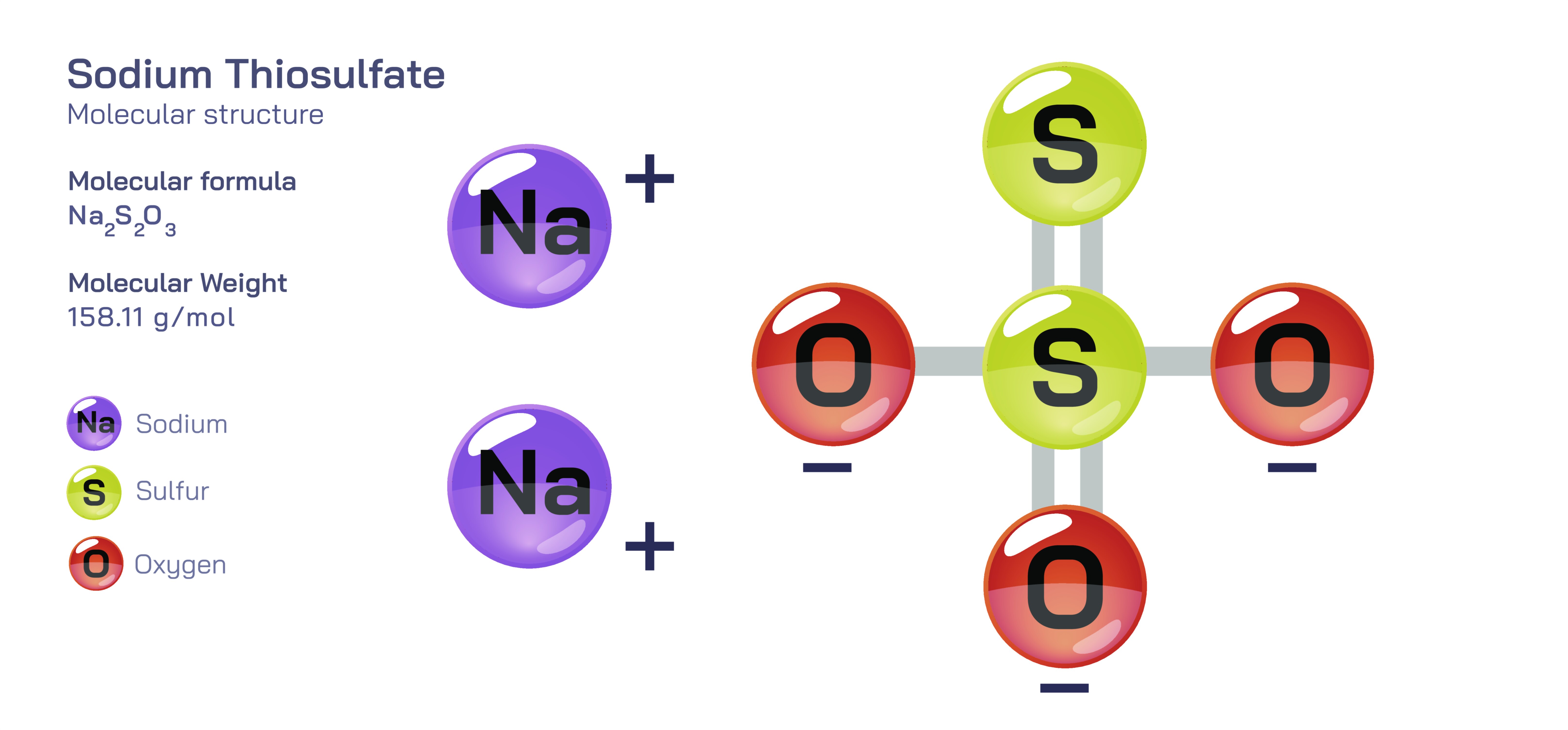
Sodium Thiosulfate – Molecular Structure, Ionic Composition, Reaction Behavior, and Educational Importance in Chemistry
Sodium thiosulfate is one of the most intellectually rich compounds encountered in chemistry education because it bridges structural chemistry, redox behavior, coordination chemistry, and kinetic experiments in a uniquely accessible way. Represented by the formula Na₂S₂O₃, it consists of two sodium ions paired with one thiosulfate ion, and this ionic pairing immediately introduces students to the concept of charge balancing in compounds formed from metal cations and polyatomic anions. Each sodium ion carries a single positive charge, while the thiosulfate anion holds an overall negative charge of two, stabilizing the salt through purely ionic attraction. When learners take the first step toward visualizing the compound, they discover that the sodium ions do not form directional bonds but instead occupy positions governed by electrostatic forces, while the true structure and reactivity arise from the thiosulfate ion. In this polyatomic ion, sulfur and oxygen atoms form a visually distinctive pattern, featuring one central sulfur atom bonded to three oxygen atoms and an additional sulfur atom bound directly to the central sulfur. This geometrical configuration allows students to see the subtle variations in bonding that differentiate thiosulfate from related ions such as sulfate and sulfite, while also revealing how substitution of an oxygen by a sulfur atom changes both structural and chemical behavior.
Exploring the structural width of the thiosulfate ion provides educators with an opportunity to highlight various fundamental molecular concepts at once. The sulfur–sulfur bond within the anion, absent in sulfate and sulfite, introduces the idea that inorganic polyatomic ions are not always composed of a single central atom surrounded by identical substituents, and that a change in atomic identity within an ion modifies both electron distribution and reactivity. Thiosulfate also provides a gentle introduction to resonance and electron delocalization, as the negative charge within the ion is not fixed on a single position but shared between atoms through electron distribution patterns that can be represented using multiple equivalent resonance forms. This electron distribution helps explain the anion’s stability and its affinity for forming complex ions with transition metals. Even the geometry of the thiosulfate ion stimulates theoretical discussion: although often depicted as a tetrahedral arrangement around the central sulfur, the presence of a sulfur substituent introduces slight distortions from perfect symmetry, allowing teachers to demonstrate how the balance between formal bonding and electron repulsion shapes real molecular structures.
When sodium thiosulfate dissolves in water, it dissociates straightforwardly into sodium ions and thiosulfate ions, and the liberated ions behave predictably in aqueous solution, reflecting the compound’s character as a strong electrolyte. Sodium ions remain chemically passive as spectator ions that maintain charge balance without influencing reactions directly, while the thiosulfate ion exhibits a reactive chemistry that becomes invaluable to laboratory instruction. One important educational application lies in the compound’s role as a reducing agent. The thiosulfate ion readily donates electrons in redox reactions, often becoming oxidized to tetrathionate or sulfate depending on reaction conditions. This property gives students a concrete way to study electron transfer, and many introductory redox experiments involve monitoring the transformation of thiosulfate under controlled conditions. In iodine–thiosulfate titration experiments, for example, students learn the essential principles of volumetric analysis by allowing thiosulfate to convert elemental iodine into iodide in a reaction that proceeds precisely and predictably. Watching the blue color of the starch–iodine complex disappear at the titration endpoint reinforces not only the value of standard solutions but also the deeper relationship between redox chemistry and analytical measurement.
Sodium thiosulfate also plays a key role in reaction kinetics experiments, making it an excellent compound for teaching rate-of-reaction theory. In one of the most famous introductory kinetic studies, sodium thiosulfate reacts with dilute acid to produce sulfur particles that turn the solution progressively cloudy. Because the reaction can be timed visually, by waiting for a marked object beneath the reaction vessel to disappear from sight, students learn how concentration, temperature, and catalysts influence the speed of chemical transformations. This experiment introduces the collision theory of reaction rates in a simple yet powerful way and offers a memorable example of how chemical reactions can be quantified even without complex instrumentation. Furthermore, the production of sulfur from this reaction highlights a broader point about chemical decomposition and the different physical forms products may adopt during reactions. While the thiosulfate ion remains dissolved at the beginning of the process, the sulfur atoms produced lose solubility and aggregate into solid particles, illustrating the transition from dissolved ions to suspended solids.
Beyond kinetics and redox transformations, sodium thiosulfate is especially valuable for introducing coordination chemistry—the study of complex ion formation between electron-pair donors and transition metals. The thiosulfate ion displays a strong ability to bind with metal cations such as silver and iron, often forming stable, soluble complexes instead of precipitates. A familiar classroom example occurs when sodium thiosulfate dissolves silver halide precipitates by forming a soluble silver–thiosulfate complex, a reaction that not only demonstrates the chemistry of complex ions but also has real historical significance in photographic development. In traditional photography, thiosulfate acted as a “fixer” to remove unreacted silver halides from photographic film, preventing further darkening when exposed to light. This example connects structural and bonding principles directly to materials science and technological history, showing students that coordination chemistry is not just theoretical but part of everyday applied science.
From a broader educational standpoint, sodium thiosulfate supports the development of conceptual connections across multiple areas of chemistry rather than restricting learners to one isolated topic. It provides structural lessons about polyatomic ions, bonding diversity, and resonance. It supports aqueous chemistry lessons through electrolyte dissociation and spectator ions. It drives foundational redox studies with iodine titration and stoichiometric calculations. It introduces kinetics through turbidity-based rate experiments. It deepens understanding of complex ions through its ability to coordinate with metals. All of these learning opportunities arise from the same molecular structure, reinforcing that chemical behavior is not random but flows directly from atomic arrangement and electron distribution. Students who engage with sodium thiosulfate gain a clearer sense of how theoretical patterns translate into experimental outcomes and how experimental evidence can be used to support theoretical understanding.
Taken together, sodium thiosulfate exemplifies how a single compound can serve as a bridge between fundamental and advanced chemistry, accessible to beginners yet still scientifically meaningful for higher levels of study. Its unique molecular structure, its dual-sulfur feature, its ability to participate in redox and coordination reactions, and its measurable kinetic behavior all arise from the precise configuration of its atoms and ions. By working with sodium thiosulfate in classroom discussions and laboratory settings, learners begin to see chemistry not merely as a memorization exercise but as an interconnected system governed by predictable laws and logical patterns. The compound becomes a powerful educational tool because it aligns structural chemistry with observable phenomena, offering a clear pathway from atomic-scale reasoning to large-scale chemical application, and demonstrating that deeper understanding always emerges from the careful study of structure, bonding, and reaction patterns at the molecular level.