ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉÅØÉ×ØɽØÉ©ØÉ▒ØÉóØÉØØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉæØÉ×ØÉØØÉ©ØÉ▒ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓ ØÉóØɦ ØÉ¢ØÉÜØÉŁØÉ×Øɽ ØÉÜØɦØÉØ ØÉĆØÉØØÉ»ØÉÜØɦØÉ£ØÉ×ØÉØ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
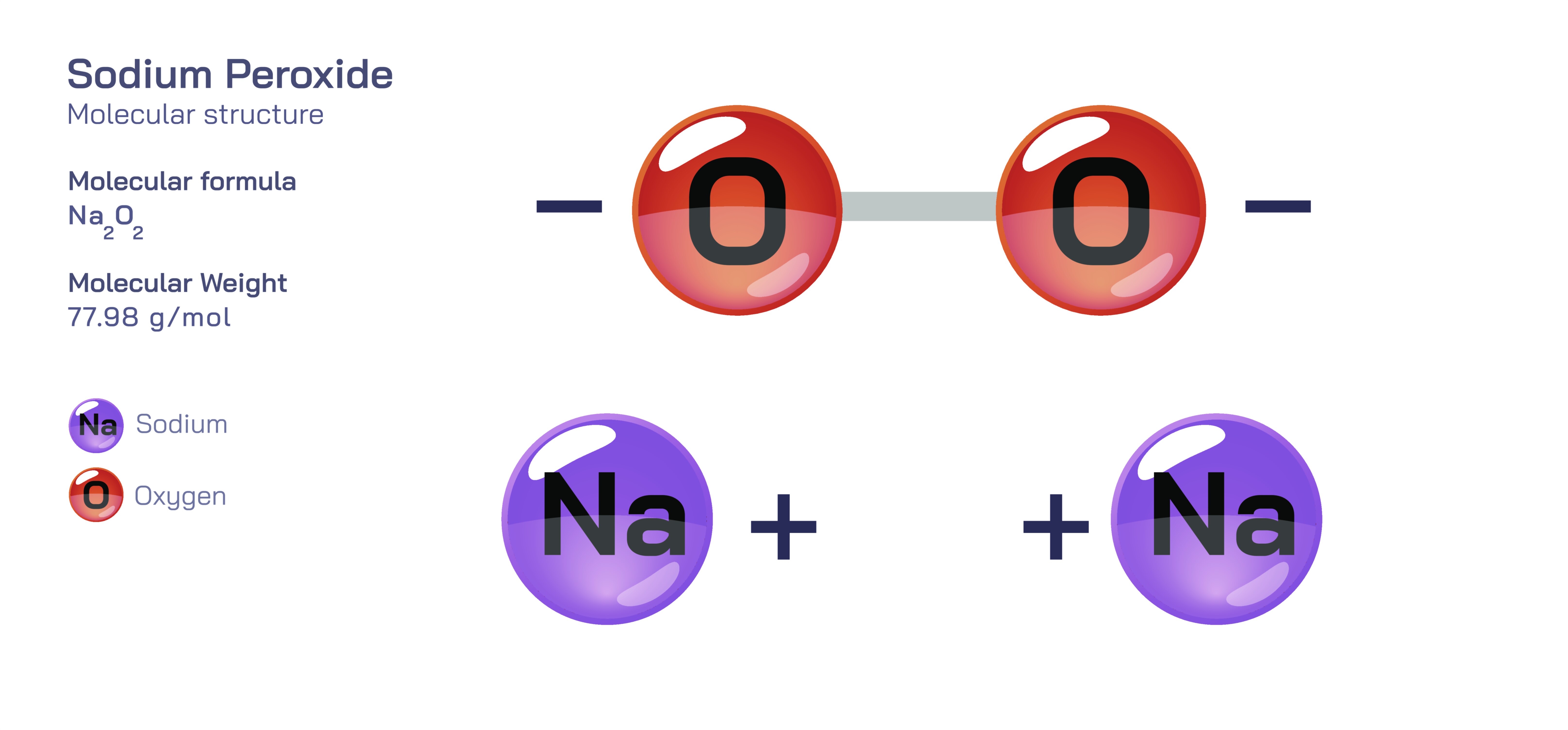
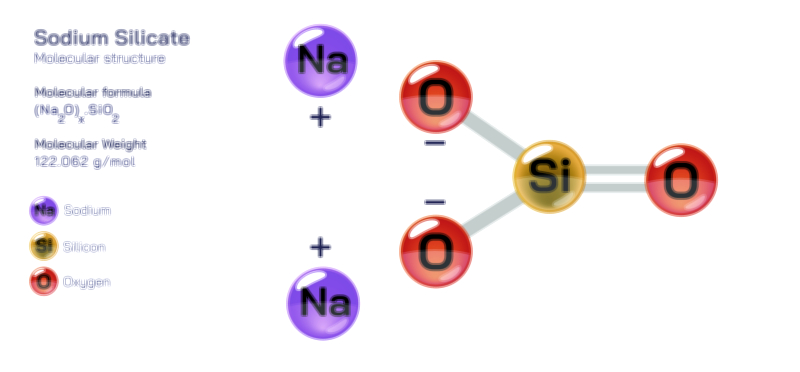
Sodium peroxide, written chemically as NaŌééOŌéé, is an inorganic ionic compound that stands at the intersection of alkali metal chemistry and oxygen chemistry. Although it appears visually as a pale yellow crystalline powder, its molecular identity reveals a rich and multilayered structure that demonstrates how electron transfer, ionic stabilization and oxygenŌĆōoxygen covalent bonding combine to create a reactive oxidizing material. Sodium peroxide bridges multiple chemical conceptsŌĆöincluding oxidation states, peroxo bonding, hydrolysis, oxygen evolution, redox thermodynamics and lattice stabilizationŌĆöand therefore serves as an excellent model compound for studying the relationship between atomic structure and macroscopic behaviour.
At the most fundamental level, sodium peroxide is formed by the interaction of elemental sodium with oxygen under controlled conditions. Sodium, a Group 1 alkali metal, carries one valence electron that is easily released to achieve a stable electron configuration. Oxygen, a Group 16 nonmetal, has six valence electrons and typically gains two electrons to complete its octet in the oxide ion, O┬▓Ōü╗. However, sodium peroxide does not contain the oxide ion. Instead, it contains the peroxide ion, OŌéé┬▓Ōü╗, a polyatomic anion consisting of two oxygen atoms joined by a single covalent bond, each carrying one extra electron. These extra electrons contribute to a total ŌłÆ2 charge on the ion. The formation of peroxide reflects an important truth in chemistry: not all ionic sodiumŌĆōoxygen compounds contain oxide; the structure depends on thermodynamic stability and ionic radius matching.
The peroxide ion is the structural and chemical centerpiece of NaŌééOŌéé. Within OŌéé┬▓Ōü╗, the oxygen atoms share electrons covalently while simultaneously accommodating extra electron density in antibonding orbitals. This leads to a bond order of 1, which makes the OŌĆōO bond weaker than the double bond in molecular oxygen and much weaker than the triple bond in ozone or the ŽĆ-bonding network in nitrate. The weakened but electron-rich bond explains much of sodium peroxideŌĆÖs behaviour: it is stable enough to exist as a crystalline solid, yet reactive enough to release oxygen or transfer electrons in redox reactions. The sodium ions in the structure, NaŌü║, are generated when sodium atoms lose their valence electrons and form positively charged cations. These cations do not participate in the chemistry directly but serve to balance the charge of the peroxide ions.
In the solid state, sodium peroxide forms an extended ionic lattice with alternating NaŌü║ and OŌéé┬▓Ōü╗ units. This lattice ensures that each peroxide ion is surrounded by multiple sodium ions, stabilizing the electrostatic interactions. Unlike simple oxides in which monatomic O┬▓Ōü╗ ions provide a compact coordination point, peroxide ions require additional structural flexibility due to their elongated shape and internal covalent bond. The lattice of sodium peroxide accommodates this geometry by allowing NaŌü║ ions to coordinate in positions that minimize electrostatic repulsion while maximizing attraction. This gives NaŌééOŌéé a high melting point, distinct crystal form and storage stability under dry conditions.
However, sodium peroxide does not remain passive when exposed to water, and here its chemistry becomes especially instructive. The peroxide ion is too reactive to persist unchanged in aqueous systems. When NaŌééOŌéé contacts water, it undergoes hydrolysis to produce sodium hydroxide (NaOH) and hydrogen peroxide (HŌééOŌéé). The reaction continues spontaneously because sodium hydroxide forms immediately and creates a strongly alkaline environment. In this high pH medium, the hydrogen peroxide produced is rapidly converted into perhydroxyl ions (HOŌééŌü╗) and may further decompose into oxygen gas (OŌéé). The ability of sodium peroxide to generate hydrogen peroxide and oxygen on contact with water gives insight into why the compound is used in oxygen-releasing applications such as life-support systems and purification environments. Through this interaction, students observe the principle that ionic compounds sometimes trigger chemical reactions rather than undergo simple dissolution.
Sodium peroxide also displays powerful oxidizing behaviour, and this behaviour is a direct consequence of the electron arrangement inside the peroxide ion. Because the peroxide ion contains electrons in antibonding orbitals, it tends to remove electrons (oxidize other substances) to reach a more stable electronic configuration. In oxidation reactions, the peroxide ion is reduced to oxide, O┬▓Ōü╗, while the other species is oxidized. This redox transformation illustrates the deeper energetic rule: peroxide ions are intermediates between molecular oxygen and oxide ions and can move toward either form depending on the reaction environment. In acidic conditions, sodium peroxide releases oxygen aggressively; in alkaline conditions controlled oxidation occurs more slowly, providing real-world demonstrations of how pH controls reactivity.
The behaviour of sodium peroxide changes drastically in acid. When reacted with mineral acids such as hydrochloric or sulfuric acid, NaŌééOŌéé decomposes rapidly, releasing oxygen gas and forming the corresponding sodium salt. The violent nature of this reaction shows that the peroxide ion is highly unstable in a proton-rich, low-pH environment. This reinforces a central acidŌĆōbase concept: basic oxygen species cannot coexist with acids because peroxide and oxide ions seek protonation and decomposition pathways immediately when hydrogen ions are present.
Heat also influences sodium peroxide dramatically. Upon heating above specific thresholds, the compound loses oxygen and converts to sodium oxide (NaŌééO). This decomposition demonstrates another major theme in inorganic chemistry: oxygen-rich anions such as peroxides contain higher energy bonds than oxides, and heating provides the energy required to shift the compound to a lower-energy oxygen state. This thermal breakdown also explains why sodium peroxide must be stored away from sources of ignition despite being nonflammable itself: the oxygen gas it produces supports combustion of flammable substances.
From an industrial and technological standpoint, sodium peroxide has played an important role in oxygen supply systems, especially in closed or underwater environments, because one mole of NaŌééOŌéé reacts with moisture to produce oxygen without the need for atmospheric interaction. In analytical chemistry, it has been used to oxidize metals in sample preparation for elemental analysis. In organic chemistry and polymer science, sodium peroxide functions as a controlled, high-strength oxidizing agent that can convert sensitive substrates in reactions where molecular oxygen or hydrogen peroxide would be too weak or uncontrollable.
In a comparative context, sodium peroxide is part of the broader family of alkali-metal oxygen compounds, which include oxides (O┬▓Ōü╗), peroxides (OŌéé┬▓Ōü╗) and superoxides (OŌééŌü╗). The identity of the compound an alkali metal forms depends heavily on the size and charge density of the metal cation. Lithium forms only the oxide, sodium forms the peroxide preferentially, and potassium, rubidium and cesium form superoxides, which are more stable with large cations. This trend shows the deep periodic principle that ionic radius compatibility determines which oxygen species are stabilized in a solid lattice. Sodium peroxide therefore becomes a stepping stone for students to understand why chemical formulas evolve systematically across a group in the periodic table.
From an educational perspective, sodium peroxide embodies multiple foundational concepts in chemistry: it demonstrates electron transfer and ionic bonding, covalent bonding within a polyatomic ion, resonance-like electron distribution in antibonding orbitals, lattice stabilization, acidŌĆōbase hydrolysis, pH-dependent speciation, thermal decomposition, periodicity in compound formation and the principle that oxidation states reflect electron energy, not simply numerical labels. The sodium ion serves as a stabilizer and structural anchor, while the peroxide ion dictates every aspect of chemical behaviourŌĆöfrom reactivity in water to oxidizing strength and decomposition.
Ultimately, sodium peroxide is far more than a bleaching reagent or laboratory oxidizer. It is a compelling example of how atomic structure and electron distribution determine macroscopic behaviour. The presence of an OŌĆōO single bond loaded with excess electrons produces a species that is stable in the dry crystalline state but explosive in the presence of acids and water. It provides oxygen on demand, serves as an oxidant when needed, and showcases the profound influence of electron organization on reactivity. Through NaŌééOŌéé, one gains a vivid understanding of the essential rule of chemistry: the properties of every substance originate from the way electrons arrange themselves within and between atoms, linking the microscopic and macroscopic worlds into a single scientific continuum.