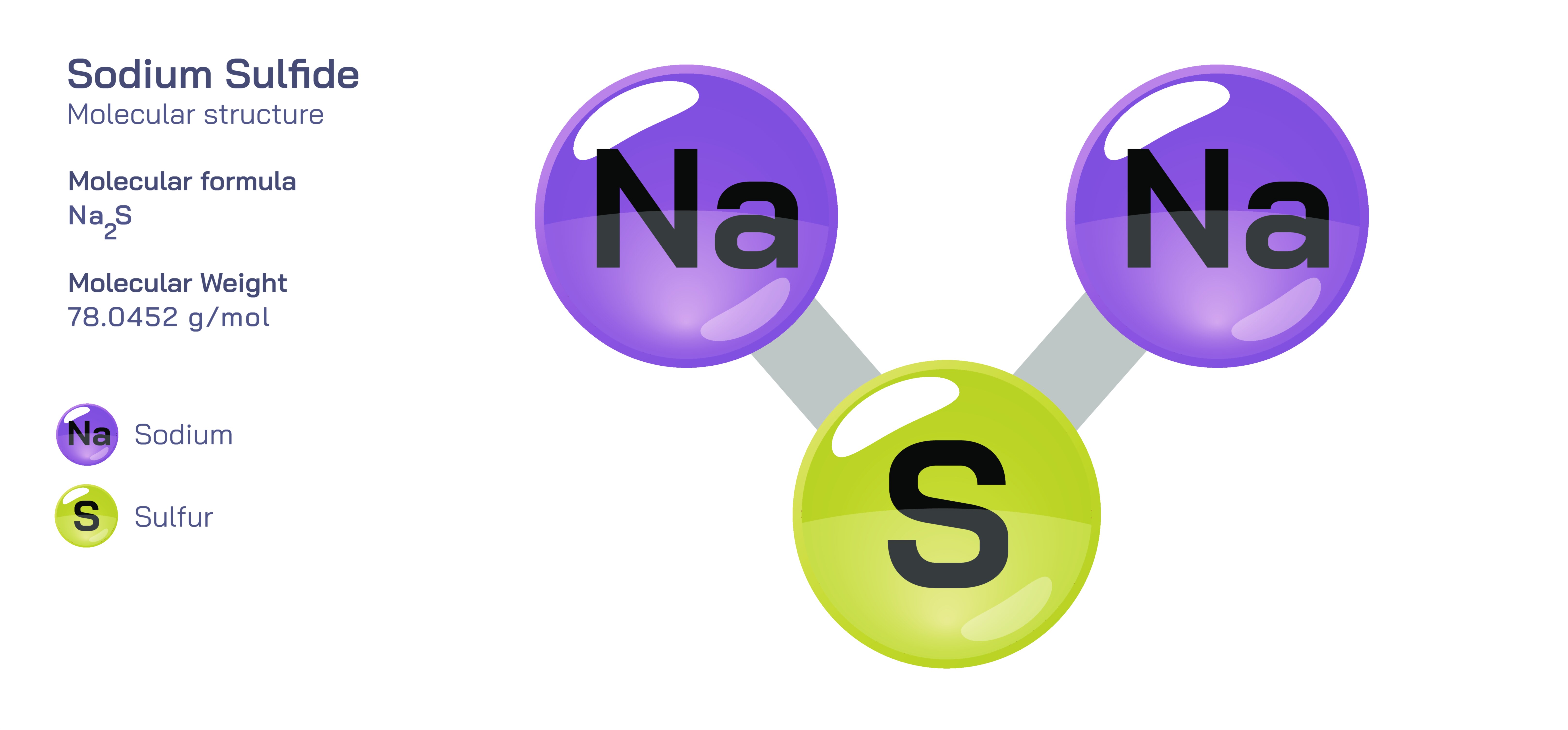
Sodium Sulfide – Molecular Structure, Ionic Bonding, Chemical Behavior, and Educational Overview
Sodium sulfide stands out in inorganic chemistry as an instructive compound that demonstrates the nature of strong ionic bonding between metallic cations and electronegative anions, making it valuable for both theoretical learning and laboratory application. With the chemical formula Na₂S, sodium sulfide consists of two sodium ions associated with one sulfide ion, a stoichiometric arrangement that balances charges through the interaction of two monovalent positive ions and a single divalent negative ion. This relationship provides an ideal opportunity for students to visualize charge neutrality in ionic compounds and to understand how electrostatic forces dictate the ratios in which ions combine. When imagining the molecular composition, it is helpful to picture the sulfur atom bearing its full negative charge after accepting electrons, while the two sodium atoms each release an electron to achieve noble gas electron configurations, resulting in the formation of a stable ionic compound driven by mutual attraction between oppositely charged species. This type of atomic exchange, common among alkali metals and chalcogens, gives sodium sulfide its fundamental identity and forms the basis for much of its characteristic chemical behavior.
The behavior of sodium sulfide in the solid state is greatly influenced by its crystalline ionic lattice, in which sodium ions and sulfide ions alternate in a highly ordered three-dimensional structure that stretches throughout the material. Unlike covalent compounds that rely on directional bonding patterns, sodium sulfide derives its integrity from nondirectional electrostatic attractions, meaning each ion interacts with several surrounding ions in a symmetrical pattern that maximizes stability. This geometric arrangement contributes to properties such as high melting point, strong lattice energy, and the ability to conduct electricity when molten or dissolved in water—all of which serve as hallmark indicators of ionic compounds in general. Sodium sulfide therefore becomes a highly effective educational tool when demonstrating the correlation between bonding and physical characteristics, because it behaves as a straightforward ionic system that aligns clearly with textbook theory. One key aspect that students often examine is the dissociation process in aqueous solution, where the compound separates into individual sodium and sulfide ions, allowing learners to relate ionic structure to behavior in solvents and to understand how electrolytes regulate conductivity and ion mobility.
In the dissolved state, sodium sulfide displays additional concepts that prove meaningful for chemical education, especially in discussions of hydrolysis and acid–base reactions. The sulfide ion, once in water, interacts strongly with the surrounding solvent and tends to produce hydroxide ions through its affinity for protons, demonstrating its basic nature and allowing students to observe pH shifts that directly result from ionic hydration. This particular feature distinguishes sodium sulfide from many other salts that remain chemically passive in aqueous media and introduces learners to the idea that not all ions behave neutrally when dissolved. These hydrolysis reactions illustrate how the chemical identity of an anion influences solution chemistry, affecting properties such as alkalinity, reactivity, and suitability for different experiments or industrial uses. Through this type of exploration, students gain a more nuanced understanding of ionic compounds, learning that behavior in water depends on more than simple dissolution and that ions carry inherent chemical tendencies that persist even outside the crystal lattice.
Another important aspect of sodium sulfide that strengthens its value in chemistry education is its participation in precipitation reactions and its ability to form insoluble metal sulfides. When sodium sulfide is introduced into solutions containing transition metal ions, the strong affinity of sulfide for many metals leads to the formation of characteristic precipitates, which often display vivid colors and offer highly visible confirmation of reaction progress. This makes sodium sulfide particularly useful in qualitative inorganic analysis, where the identification of unknown metal ions relies on the controlled formation of predictable precipitates. Through these experiments, learners observe firsthand how the sulfide ion interacts selectively with certain cations, reinforcing the relationship between ionic charge, solubility rules, and compound stability. This selectivity not only deepens understanding of reaction pathways but also reflects how sulfide chemistry plays a role in environmental systems, mining processes, metallurgy, and waste treatment—fields in which sulfide precipitation allows recovery or removal of heavy metals.
From a structural perspective, sodium sulfide also helps introduce the concept of lattice defects and hydration effects, especially because the compound can exist in anhydrous and hydrated forms depending on exposure to moisture. When sodium sulfide takes up water, it forms hydrates that integrate water molecules into the ionic framework without altering the essential stoichiometric relationship between sodium and sulfur, making it possible to teach crystallization, hygroscopic behavior, and the influence of environmental conditions on crystalline stability. The compound’s tendency to absorb moisture and transition from one form to another can be tied to lessons about thermodynamics and phase transitions, showing how physical states change without breaking ionic bonds. Moreover, its sensitivity to moisture and reaction with atmospheric gases such as carbon dioxide gives students opportunities to explore decomposition pathways and understand how air exposure can gradually transform certain inorganic materials into new products over time.
Beyond purely chemical observations, sodium sulfide also helps connect molecular structure to practical application. Its strong basicity, ability to precipitate metals, and predictable ionic dissociation make it indispensable in several industrial sectors, including leather processing, pulp treatment, ore smelting, and wastewater purification. These applications demonstrate how properties arising from atomic-level structure scale into large-scale utility, reinforcing the importance of ionic bonding concepts not just within the laboratory but across real-world processes. In industrial settings, sodium sulfide is often valued for its reactivity rather than its inertness, showing students how compounds may be deliberately chosen for chemical activity rather than stability when designing technological solutions. This distinction encourages learners to evaluate chemical reagents not only based on formula or structure but also based on the context of intended function.
Taken as a whole, sodium sulfide provides a comprehensive foundation for chemical education because it illustrates fundamental principles of ionic bonding, lattice organization, hydration, acid–base reactions, solubility rules, and precipitation behavior in a cohesive and highly observable way. Each stage of analysis—from the structure of the solid to the behavior of the dissolved ions—reveals another connection between molecular identity and macroscopic properties, helping learners build chemistry knowledge layer by layer. By working with sodium sulfide in both theoretical and experimental contexts, students gain experience in translating symbolic formulas into real behaviors, an essential skill for advancing from introductory chemistry to applied laboratory science. In this way, sodium sulfide becomes more than a formula or reagent; it becomes an educational gateway to understanding how elemental interactions shape the chemical world and how mastery of structure and bonding unlocks deeper comprehension of both scientific and industrial chemistry.