ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉÅØÉĪØÉ©ØɼØÉ®ØÉĪØÉÜØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉłØÉ©ØɦØÉóØÉ£ŌĆōØÉéØÉ©ØÉ»ØÉÜØÉźØÉ×ØɦØÉŁ ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉÅØÉĪØÉ©ØɼØÉ®ØÉÜØÉŁØÉ× ØÉłØÉ©Øɦ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓ ØÉÜØɦØÉØ ØÉĆØÉØØÉ»ØÉÜØɦØÉ£ØÉ×ØÉØ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
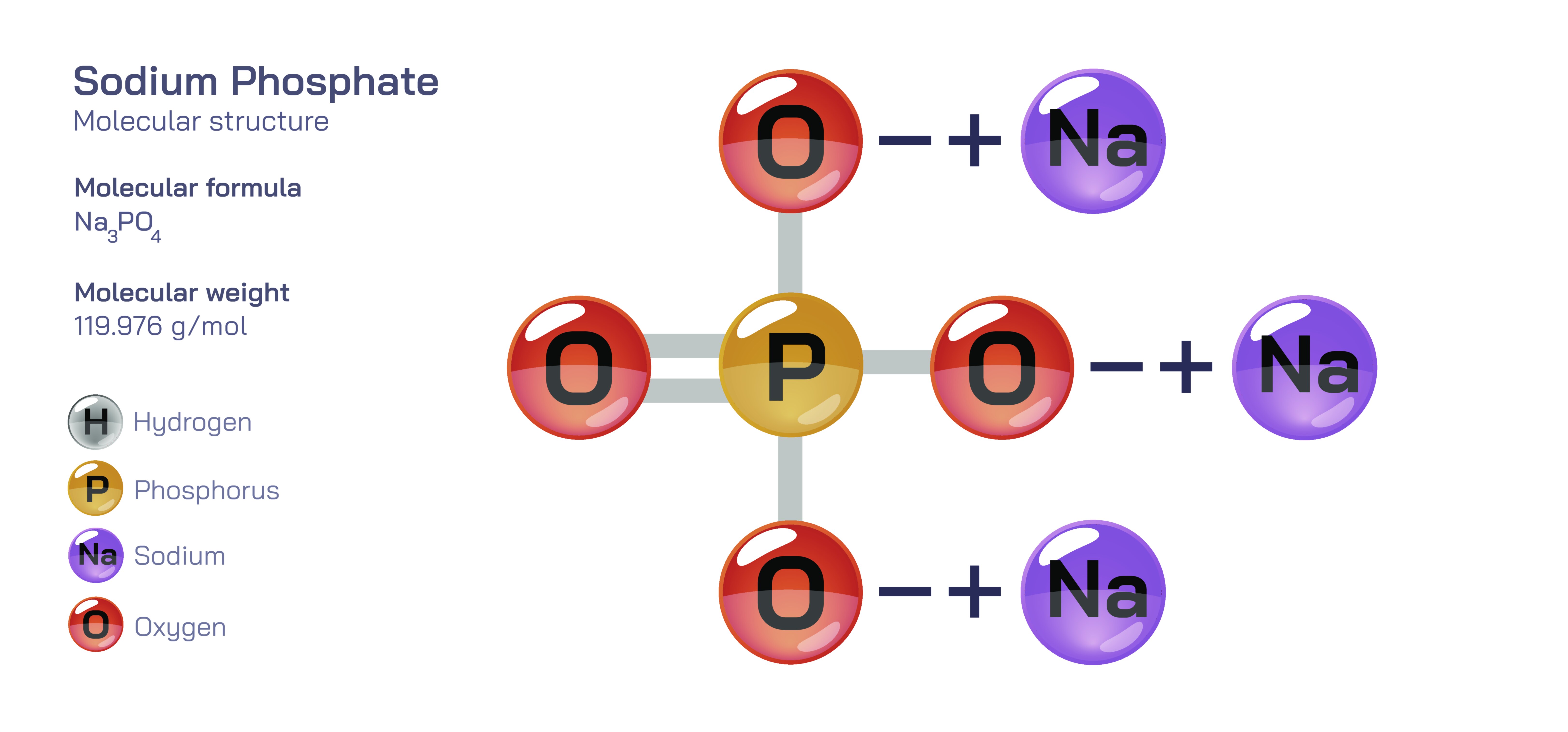
Sodium phosphate is the general name for a family of inorganic sodium salts of phosphoric acid, most commonly represented in the fully neutralized form as trisodium phosphate (NaŌéāPOŌéä). Although externally it appears as a white crystalline solid, its chemical identity reflects a sophisticated combination of ionic interactions and covalent bonding that makes phosphate-containing compounds essential to geology, biology, industry and analytical chemistry. Understanding sodium phosphate requires understanding the underlying phosphate ion, which displays one of the most important bonding patterns in chemistry ŌĆö a tetrahedral, resonance-stabilized oxyanion with both double-bond and single-bond character distributed across four oxygen atoms. Because of its stability, phosphate becomes a centerpiece of biomolecules such as DNA and ATP and a foundational building block of minerals and detergents.
At the microscopic level, sodium phosphate consists of sodium cations (NaŌü║) paired with phosphate anions (POŌéä┬│Ōü╗). Sodium becomes a cation by losing its single valence electron, reaching a closed-shell configuration. It plays a largely structural role, contributing to electrostatic stability without determining the reactive behaviour of the compound. The phosphate ion, by contrast, is a polyatomic ion composed of a central phosphorus atom covalently bonded to four oxygen atoms. Phosphorus in this arrangement exists in the +5 oxidation state, and the geometry of the ion is tetrahedral, with bond angles close to 109.5┬░, reflecting sp┬│ hybridization around the phosphorus center.
One of the most critical features of the phosphate ion is resonance. Although diagrams often depict one phosphorusŌĆōoxygen double bond and three PŌĆōO single bonds, experimental measurements show that all four bonds have very similar length and energy. This occurs because the extra electrons are not confined to a single oxygen atom; instead, they are delocalized across the entire ion. This delocalization provides the POŌéä┬│Ōü╗ ion with exceptional thermodynamic and kinetic stability, making it far less reactive than many other oxyanions. This stability explains why phosphate forms durable minerals such as apatite and why biological systems evolved to use phosphate as a fundamental structural unit in energy transfer and genetic material.
In the solid state, sodium phosphate forms an ionic lattice containing alternating NaŌü║ ions and POŌéä┬│Ōü╗ ions arranged in a repetitive three-dimensional matrix. This packing maximizes ionic attraction while minimizing repulsion between ions of like charge. Trisodium phosphate (NaŌéāPOŌéä) contains three sodium cations for every phosphate ion, neutralizing its ŌłÆ3 charge. Although the structure contains strong ionic forces, the internal integrity of the phosphate ion itself is preserved; the covalent bonds within the POŌéä┬│Ōü╗ ion remain intact and independent of the ionic bonds between phosphate and sodium. This duality ŌĆö covalent bonding inside the anion and ionic bonding between ions ŌĆö makes sodium phosphate a model compound for illustrating the coexistence of bonding types.
When sodium phosphate dissolves in water, the ionic lattice dissociates to release free NaŌü║ ions and POŌéä┬│Ōü╗ ions into solution. Sodium acts as a spectator ion, remaining solvated but chemically inactive. The phosphate ion, however, participates in acidŌĆōbase equilibria, because it is the conjugate base of phosphoric acid (HŌéāPOŌéä), a triprotic acid. Rather than existing exclusively as POŌéä┬│Ōü╗ in solution, the phosphate species undergoes progressive protonation depending on the pH. At very high pH, POŌéä┬│Ōü╗ dominates. At moderately high pH, it protonates to hydrogen phosphate (HPOŌéä┬▓Ōü╗), and at neutral to mildly acidic conditions, it becomes dihydrogen phosphate (HŌééPOŌéäŌü╗). In strongly acidic solutions, the fully protonated phosphoric acid (HŌéāPOŌéä) form exists. This elegant progression of protonation demonstrates a key educational principle: ionic species in solution are not static and change identity depending on pH.
The phosphate ion is not typically a redox-active species under ordinary conditions because phosphorus in POŌéä┬│Ōü╗ already exists in its highest common oxidation state (+5). Therefore, phosphate generally acts not as an oxidizing agent or reducing agent, but rather as a base and a ligand. As a base, it neutralizes acids by accepting protons. As a ligand, phosphate can coordinate to metal ions through oxygen atoms, forming complex ions or participating in precipitation reactions. For example, calcium phosphate is poorly soluble and precipitates easily, which forms the mineral basis of bones and teeth in biological organisms and phosphorite deposits in geology. These reactions demonstrate the importance of phosphate not through its reactivity as a redox agent, but through coordination chemistry and solubility equilibrium.
In industrial chemistry, sodium phosphate reflects the broader behaviour of alkali phosphates ŌĆö strong alkalinity in solution, high buffering capacity and the ability to chelate or bind metal ions. These properties make sodium phosphate useful in detergents, boiler water treatment, ceramics, textile processing and emulsification. The buffering behaviour arises because the phosphate ion and its protonated forms can resist changes in pH, providing chemical stability to systems ranging from washing solutions to biological cells. Even in household applications, sodium phosphate illustrates major chemical concepts: its cleaning efficiency arises partly from its ability to soften water by binding calcium and magnesium ions, preventing the formation of insoluble soap scum and allowing surfactants to operate efficiently.
Thermal behaviour reveals another aspect of phosphate chemistry. Trisodium phosphate does not decompose easily because the PŌĆōO framework is extremely stable. However, heating can drive off water of crystallization in its hydrated forms and can eventually lead to condensation reactions that form polyphosphates, where phosphate units link together through oxygen bridges. These condensed phosphate materials are major industrial compounds used in detergents, water treatment and food processing. Their formation illustrates condensation polymerization in inorganic chemistry ŌĆö showing that polymer formation is not restricted to carbon chemistry.
In biological chemistry, phosphateŌĆÖs behaviour reaches its highest significance. The same tetrahedral structure of POŌéä┬│Ōü╗ that appears in sodium phosphate forms the backbone of DNA and RNA and the energy-storing molecule adenosine triphosphate (ATP). The PŌĆōOŌĆōP bonds in ATP break and reform to store and release biological energy. The resonance stabilization that makes phosphate chemically stable also prevents accidental breakdown of genetic information and metabolic energy reserves. Thus the stability of the phosphate ion, exemplified in sodium phosphate, is essential for the persistence of life on Earth.
From an educational perspective, sodium phosphate brings together ionic bonding, covalent bonding, resonance stabilization, acidŌĆōbase chemistry, buffering behaviour, solubility equilibrium, thermal transformations and biological relevance. The sodium ion contributes structure, conductivity and solubility but does not determine the fundamental reactivity. The phosphate ion determines nearly every chemical property of the compound ŌĆö its stability, acidity, coordination ability and its behaviour in environmental, industrial and biological systems.
Ultimately, sodium phosphate illustrates one of the deepest truths of chemistry: the behaviour of a compound is governed by the structure and electrons of its ions, not merely by its formula. The POŌéä┬│Ōü╗ ion is stable because its electrons are delocalized over a tetrahedral network; it is basic because of its formal charge distribution; it buffers solutions because of stepwise protonation; it binds metals through oxygen lone pairs; and it supports life because its structure resists unwanted breakdown while enabling controlled release of energy. Through NaŌéāPOŌéä, students discover that chemistry is not an abstract table of substances, but a science in which electrons and geometry combine to shape every material and every biological process in the world around us.