ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉÄØÉ▒ØÉóØÉØØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉŗØÉÜØÉŁØÉŁØÉóØÉ£ØÉ× ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓ ØÉÜØɦØÉØ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽ.
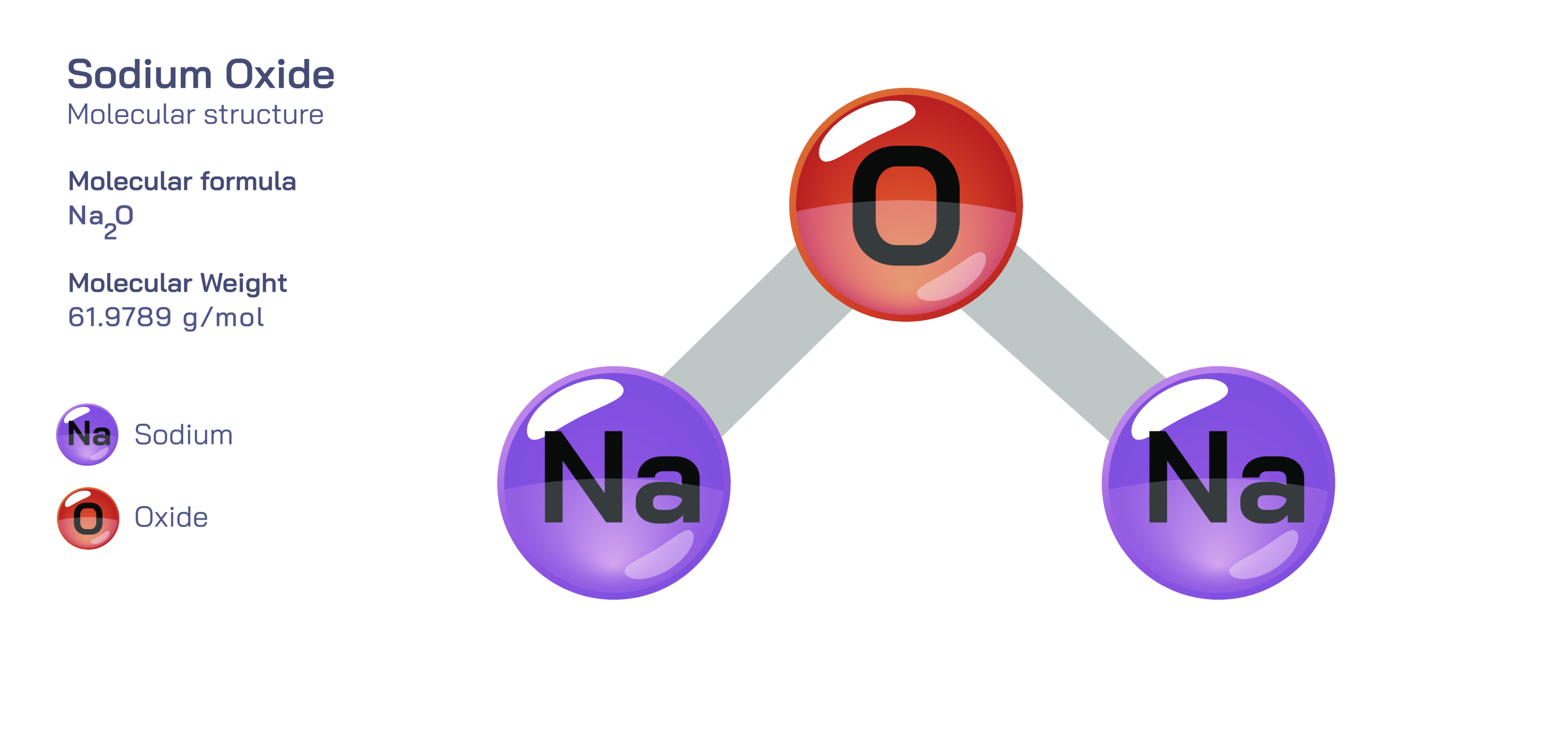
Sodium oxide, written chemically as NaŌééO, is an inorganic ionic compound formed from sodium and oxygen that reveals a deep and instructive view into ionic bonding, lattice stability, acidŌĆōbase chemistry, electron transfer, and the way chemical structure governs macroscopic reactivity. At first glance, sodium oxide appears to be just another white, crystalline solid belonging to the category of metal oxides. Yet its identity is far more meaningful when examined at the molecular and electronic levels. Sodium oxide shows how electron transfer from a reactive metal to a nonmetal generates ions, how these ions assemble into a stable crystal structure, how oxides influence the alkalinity of materials, and how the behaviour of a compound can change drastically in the presence of water. By exploring NaŌééO, one discovers not just the chemical behaviour of a specific substance but also major principles that guide inorganic chemistry as a whole.
The formation of sodium oxide begins at the atomic level, with elemental sodium and oxygen ŌĆö two elements that have completely different tendencies in electron behaviour. Sodium, belonging to Group 1 of the periodic table, contains one valence electron in its outermost shell. This electron is easily lost because removing it allows sodium to adopt the stable electronic configuration of neon. Oxygen, on the other hand, belongs to Group 16 and has six valence electrons, requiring two more electrons to complete its octet. During oxide formation, each sodium atom donates one electron, and each oxygen atom accepts two electrons. As a result, the atoms transform into ions: sodium becomes NaŌü║, and oxygen becomes the highly charged oxide ion, O┬▓Ōü╗. The oxide ion is one of the strongest bases known in inorganic chemistry because it carries a concentrated negative charge that seeks protonation or electron redistribution.
In the solid state, sodium ions and oxide ions arrange themselves into a three-dimensional ionic lattice held together by electrostatic attraction between positively charged NaŌü║ and negatively charged O┬▓Ōü╗. This lattice is not composed of individual NaŌééO molecules but rather an extended array where each oxide ion is surrounded by sodium ions, and each sodium ion is coordinated to oxide ions. The strong ionic forces in this lattice explain why sodium oxide has a high melting point, a brittle physical structure and a well-defined crystalline form. These properties also highlight an important concept: ionic bonds are not directional like covalent bonds, and the stability of the solid originates from the repeating electrostatic pattern rather than the bonding of isolated molecular units.
Even though NaŌééO is stable in the absence of moisture, its behaviour changes dramatically in the presence of water. Water has a high dielectric constant and strong polarity, meaning it can pull apart ionic lattices through hydration. The moment sodium oxide contacts water, a chemical reaction rather than simple dissolution takes place. The oxide ion is too basic to exist freely in aqueous solution; instead, it reacts with water to form hydroxide ions (OHŌü╗). The reaction converts sodium oxide into sodium hydroxide, NaOH, producing a strongly alkaline solution. This transformation demonstrates a major rule of acidŌĆōbase chemistry: metal oxides typically form bases in water. The oxide ion accepts protons from water, illustrating how strong bases arise not only from soluble hydroxide salts but also from oxides that hydrolyze to release hydroxide.
From a reactivity perspective, sodium oxide can be thought of as a highly condensed form of sodium hydroxide, because it contains two sodium atoms associated with one oxide ion, which effectively stores two equivalents of hydroxide-forming capacity. This property is crucial in industrial glass manufacturing. Sodium oxide is not added directly to molten glass; instead, sodium carbonate (NaŌééCOŌéā) is used because sodium oxide would react too violently with moisture and would be difficult to handle. Upon heating, sodium carbonate decomposes into sodium oxide, which then integrates into the glass matrix. Sodium oxide acts as a network modifier, reducing the rigidity of the silica network in glass and improving its melting properties. Thus the behaviour of sodium oxide ŌĆö strong basicity and structural modification ŌĆö is directly tied to its ion identity and charge distribution.
The electronic structure of the oxide ion helps explain why sodium oxide is such a powerful base and such a strong reducing agent at high temperatures. The O┬▓Ōü╗ ion carries two extra electrons that are held tightly by the high electronegativity of oxygen. These electrons are highly concentrated in space, making the ion extremely reactive in environments where protons or electrophilic species are available. This explains why sodium oxide reacts explosively with acids; even weak acids are proton donors when facing the oxide ion. The reactivity also explains why sodium oxide must be handled away from atmospheric moisture and carbon dioxide. In the presence of carbon dioxide, sodium oxide is converted to sodium carbonate (NaŌééCOŌéā), demonstrating a direct pathway where the oxide ion acts as a base and neutralizes acidic oxides.
Sodium oxide serves as a valuable example for studying thermal stability and decomposition. Unlike many other metal oxides, sodium oxide does not decompose into simpler oxides or elements when heated under ordinary conditions. This stability arises from the balance between ionic radii and lattice energies: the pairing of NaŌü║ and O┬▓Ōü╗ produces an energetically favourable arrangement that does not require major structural change. However, sodium oxide is not stable in the presence of atmospheric gases, which reflects the chemical principle that thermodynamic stability depends not only on internal bonding but also on the composition of the surrounding environment.
Comparing sodium oxide with other alkali metal oxides reveals deeper periodic trends. Lithium forms the normal oxide LiŌééO but also produces peroxides and superoxides more readily as the cation size increases. Sodium oxide, NaŌééO, is the preferred oxide form because the ionic radius of NaŌü║ matches well with that of O┬▓Ōü╗. Potassium, rubidium and cesium, however, form superoxides (KOŌéé, RbOŌéé, CsOŌéé) due to their larger ionic radii and lower charge density, which stabilize the OŌééŌü╗ ion rather than O┬▓Ōü╗. Thus sodium oxide becomes part of a broader chemical story linking oxygen species to alkali metals: periodic trends in ionic radius and lattice energy determine whether oxides, peroxides or superoxides are favoured.
Although sodium oxide has limited direct use in daily life because of its aggressive basic reactivity, its industrial significance is substantial. It is indispensable in glass and ceramic manufacturing, where it functions as a fluxing agent to reduce melting temperatures and improve viscosity. It is also used in metallurgy, where it participates in oxide slag formation and chemical refining. Indirectly, sodium oxide is central to the chemistry of sodium hydroxide, sodium carbonate and other sodium-based bases ŌĆö compounds that shape detergents, pulp and paper treatment, petroleum refining, soap production and water purification. The impact of sodium oxide in technology and industry reflects once again that the most influential chemicals are sometimes those that activate transformative reactions rather than those that appear as end products.
From an educational standpoint, sodium oxide beautifully demonstrates multiple pillars of chemistry without requiring complex symbolic demonstrations. It shows how ionic bonds arise from electron transfer rather than from sharing. It shows how the charge of a polyatomic or monatomic ion controls both solubility and reactivity. It shows how water can cause chemical transformations rather than simple dissolution when ions are too basic to exist in solution. It shows how thermodynamic stability depends on both internal structure and external environment. It shows how periodic trends affect the identity of oxides across the alkali metals. And it shows how the identity of a microscopic ion determines the properties of large-scale materials, such as glasses and ceramics.
Ultimately, sodium oxide is more than an ionic solid. It is an elegant example of how electron transfer drives bond formation, how ionic structure drives physical properties, and how structure determines chemical reactivity. A single oxygen atom gaining two electrons from two sodium atoms gives rise to an ion so reactive that it cannot remain unchanged in water or air; yet the same ion, when locked inside a crystal lattice or a glass network, enhances material strength and lowers melting temperature. Through NaŌééO, the deeper truth of chemistry becomes strikingly clear: the world we see is built not from formulas and symbols, but from electrons arranging themselves into the most favourable structures allowed by nature.