ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉŹØÉóØÉŁØɽØÉóØÉØØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉłØÉ©ØɦØÉóØÉ£ ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓, ØÉÆØÉŁØÉÜØÉøØÉóØÉźØÉóØÉŁØÉ▓ ØÉÜØɦØÉØ ØÉāØÉ×ØÉ×ØÉ® ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
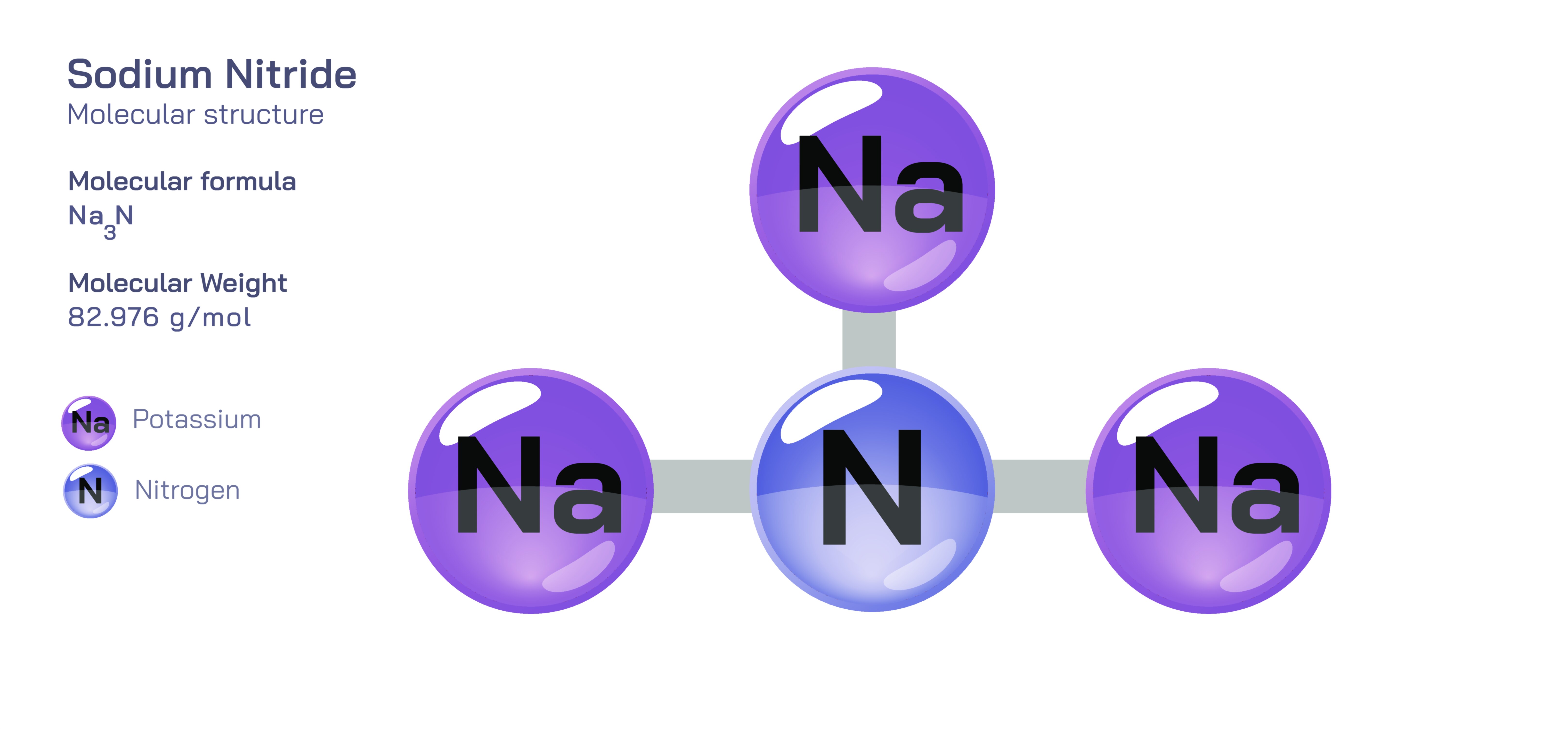
Sodium nitride, chemically represented as NaŌéāN, is one of the rarest and least stable sodium compounds, and because of this rarity it becomes a remarkable example for understanding the chemical forces that govern stability and bonding in ionic materials. Although the compound can be synthesized and observed under specific controlled conditions, it decomposes quickly under ordinary atmospheric environments. The instability of sodium nitride provides an extraordinary teaching opportunity: it demonstrates that chemical formulas do not guarantee structural viability, and that the stability of a compound is determined by the balance of electron transfer, lattice formation, ionic radii, and energetic favourability rather than by compositional predictions alone.
The structure of sodium nitride begins with an extremely unequal chemical interaction between sodium atoms and nitrogen atoms. Sodium, an alkali metal in Group 1, contains one valence electron that it readily donates, forming a sodium cation (NaŌü║) with a complete outer electron shell. Nitrogen, a Group 15 nonmetal, contains five valence electrons and generally needs three more to achieve a full stable shell. In sodium nitride, three sodium atoms each donate one electron to nitrogen. As a result, the nitrogen atom accepts three electrons and becomes N┬│Ōü╗, known as the nitride ion. This ion is among the most electron-rich and highly reactive anions in inorganic chemistry. The formal transfer of three electrons from sodium to nitrogen demonstrates an extreme case of ionic bonding because of the large electronegativity gap between the two elements.
In the solid state, sodium nitride forms a lattice consisting of NaŌü║ and N┬│Ōü╗ ions arranged in a repeating three-dimensional structure. However, the size difference between the ions creates one of the major contributors to instability. Sodium ions are relatively large and diffuse compared with the compact and highly charged nitride ion. The lattice energy ŌĆö the amount of energy stabilizing the ionic crystal ŌĆö is not sufficiently high to fully counteract the repulsion created by the density of negative charge around nitrogen. This means that NaŌéāN has an intrinsic thermodynamic tendency to break down into sodium metal and nitrogen gas under standard atmospheric conditions. The structure is most stable at very low temperatures and in inert atmospheres, emphasizing that chemical stability is not an absolute property but depends strongly on environment and energetic balance.
Sodium nitride is highly reactive because of the strong reducing character of the nitride ion (N┬│Ōü╗). With three extra electrons, this ion carries one of the highest negative formal charges of any monatomic anion. It reacts vigorously with proton sources such as water, alcohols and acids. When exposed to water, sodium nitride breaks down rapidly, forming sodium hydroxide and releasing ammonia (NHŌéā). This process demonstrates proton transfer chemistry and highlights how the nitride ion behaves as a powerful base. Instead of existing peacefully as N┬│Ōü╗, nitrogen rapidly captures hydrogen atoms because forming NŌĆöH bonds in ammonia is far more energetically favourable than remaining in the nitride state. Thus, sodium nitride reveals that stable nitrogen compounds do not always correspond to nitrogen gaining electrons fully; in fact, nitrogen achieves greater stability through covalent bonding rather than through monatomic reduction.
The decomposition pathway also reflects deeper redox principles. Although nitride carries nitrogen in its lowest possible oxidation state (ŌłÆ3), elemental nitrogen gas exists in an extraordinarily stable form with a strong triple bond between two nitrogen atoms. When sodium nitride decomposes, nitrogen is oxidized from ŌłÆ3 to 0, while sodium is reduced or remains in its +1 oxidation state depending on the mechanism and environment. The process reinforces the concept that oxidation state alone does not determine stability ŌĆö the types of bonds formed ultimately direct thermodynamics. In nitrogen gas, the triple bond gives exceptional bond strength, whereas in nitride the isolated negative charge on the nitrogen atom creates physical and electronic strain.
Sodium nitride also provides important insight into the structural trends among alkaline metal nitrides. While NaŌéāN is unstable, heavier alkali metal nitrides such as lithium nitride (LiŌéāN) are stable and isolable. This difference highlights a central educational concept: ionic radius compatibility is essential for lattice stability. Nitrides become more stable moving upward in the periodic table because smaller cations such as LiŌü║ can pack more efficiently around N┬│Ōü╗. Sodium ions are too large to generate a tightly packed lattice, and thus the stabilization energy is insufficient. This trend continues with potassium, rubidium and cesium, for which nitrides become increasingly unstable or do not form at all. Sodium nitride therefore teaches students that chemical structure is influenced not only by valence electron transfer but also by spatial and geometric compatibility between ions.
The thermal behaviour of sodium nitride reflects the same structural pressures. Even gentle heating causes the compound to decompose, evolving nitrogen gas. While many ionic solids melt before decomposing, sodium nitride breaks down chemically because decomposition provides a more energetically stable arrangement of atoms than the solid lattice. This behaviour underscores the principle that chemical decomposition is an alternative thermodynamic pathway when melting is not energetically favourable. Sodium nitride does not burn or oxidize violently; it simply reorganizes into more stable forms based on energy considerations, further supporting the concept that chemical changes are guided by energetic landscapes rather than by arbitrary reactivity.
Because sodium nitride exists only under specialised laboratory or computational conditions, there are no direct industrial or biological applications. However, its conceptual importance is enormous. It illustrates multiple paradigms of chemical education: ionic bonding and full electron transfer, stability governed by lattice energy, acidŌĆōbase reactivity of nitride, redox-driven decomposition, periodic trends across alkali metals and structural prediction limitations based purely on formula. In chemistry, sodium nitride serves as a reminder that not every theoretically conceivable compound exists in nature, and that electron counts alone do not determine viability. Structure, energy and atomic geometry decide whether a compound can persist.
In aqueous chemistry, sodium nitrideŌĆÖs rapid destruction is not merely a side reaction but an instructive demonstration of thermodynamic direction. When N┬│Ōü╗ reacts with water to form ammonia, it reflects a universal principle ŌĆö nitrogen prefers strong covalent bonding rather than extreme ionic reduction. This parallels behaviour seen in carbide, boride and phosphide chemistry, supporting wider conceptual understanding across group 15 and group 16 elements.
Ultimately, sodium nitride is a compound defined not by its longevity but by its teaching value. It illustrates that electron transfer creates ionic species, but ionic structure survives only if the resulting lattice is energetically favourable and geometrically compatible. It highlights that extreme oxidation states, whether high or low, do not guarantee stability, and that nitrogen achieves maximum stability through strong covalent bonding rather than existing as an isolated nitride ion. And it demonstrates that chemistry is not determined by symbols on paper but by the interplay of electrons, geometry, electronegativity and energy. Through the study of sodium nitride, learners gain a clearer understanding of why some compounds are stable, why others are transient and why the physical universe contains only those substances that meet the demands of energetics and electronic structure.