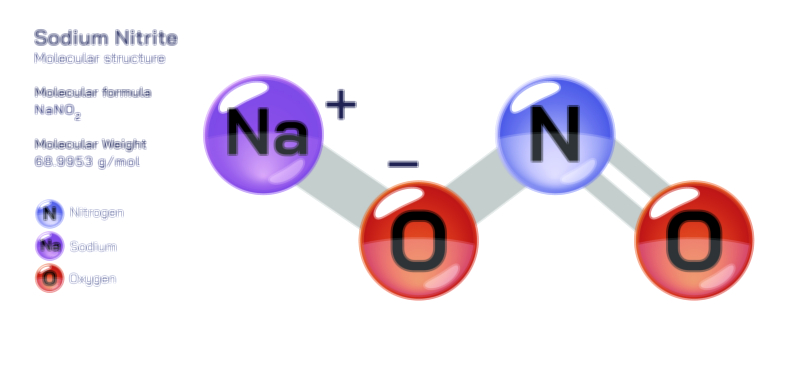
ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉćØÉ▓ØÉ®ØÉ©ØÉ£ØÉĪØÉźØÉ©ØɽØÉóØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉæØÉ×ØÉÜØÉ£ØÉŁØÉóØÉ»ØÉóØÉŁØÉ▓, ØÉÜØɦØÉØ ØÉāØÉ×ØÉ×ØÉ® ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
Sodium hypochlorite, written chemically as NaClO, is one of the most widely used chlorine-based inorganic compounds, well known for its presence in household bleach and industrial disinfectants. While its macroscopic purpose is commonly associated with cleaning and sanitation, the microscopic structure behind sodium hypochlorite reveals a far more fascinating story involving ionic bonding, covalent bonding within the hypochlorite ion, electron distribution in oxyanions, oxidationŌĆōreduction tendencies, and equilibrium behaviour in aqueous environments. Understanding sodium hypochlorite requires connecting its visible disinfecting properties with its molecular identity ŌĆö a connection that illustrates how a chemical structure translates into real-world function.
The defining structural component of sodium hypochlorite is the hypochlorite ion (ClOŌü╗). In this polyatomic anion, chlorine is covalently bonded to oxygen, forming a bent molecular architecture rather than a straight linear shape. Chlorine exists in the +1 oxidation state, an electrochemically unstable position that strongly influences reactivity. Within ClOŌü╗, oxygen is more electronegative than chlorine and pulls electron density toward itself, creating a highly polar covalent bond. Resonance within the hypochlorite ion is not as extensive as in oxyanions such as chlorate or perchlorate, but some distribution of electron density still occurs across the bond. The valence electron arrangement leaves the oxygen atom bearing most of the negative charge, while chlorine remains electron-deficient relative to its elemental state. This uneven electron placement creates the central driving force behind hypochloriteŌĆÖs oxidizing ability.
Sodium ions, NaŌü║, play a supporting yet essential role in the structure of sodium hypochlorite. When a sodium atom loses its outer valence electron, it becomes a positively charged ion stabilized by its full electron shell. In the crystalline solid, sodium ions are electrostatically paired with hypochlorite ions, forming an ionic lattice. It is important to note that NaClO does not exist as discrete ŌĆ£moleculesŌĆØ of NaŌĆōClO; instead, the solid is a repeating, three-dimensional structure of alternating NaŌü║ and ClOŌü╗ ions held in place by ionic attraction. This ionic network explains the compoundŌĆÖs crystalline form, solubility, and thermal decomposition behaviour.
The moment sodium hypochlorite enters water, a transformation occurs that dramatically illustrates solvation and equilibrium chemistry. Water molecules surround NaŌü║ and ClOŌü╗ ions, breaking the ionic lattice and dispersing them through solution. Sodium ions become fully solvated spectator ions, while the hypochlorite ion enters a dynamic chemical environment influenced by pH. In alkaline solution ŌĆö where sodium hypochlorite is most stable ŌĆö the hypochlorite ion remains largely intact. However, when the solution becomes acidic, hypochlorite converts first into hypochlorous acid (HOCl) and then, under stronger acid conditions, decomposes into chlorine gas (ClŌéé). This equilibrium behaviour reinforces a critical lesson: the chemical identity and reactivity of a substance depend strongly on proton concentration and environment, not just on the formula printed on its container.
The oxidizing behaviour of hypochlorite can be traced directly to its molecular structure. Because the chlorine atom exists in an electron-poor state, it readily accepts electrons from other molecules, converting them to oxidized forms while itself being reducedŌĆötypically to chloride (ClŌü╗), where chlorine reaches the much more stable ŌłÆ1 oxidation state. This transformation drives the compound's disinfecting properties: hypochlorite disrupts cellular molecules by oxidizing membrane proteins, enzymes, and nucleic acids in bacteria, viruses, fungi, and algae, neutralizing them through irreversible electron transfer. Thus, sodium hypochlorite does not kill microorganisms by heat or acidity or physical dissolution, but by electron-driven chemical modification of biological macromolecules.
An important aspect of its reactivity involves the hypochlorous acid formed in slightly acidic water. HOCl penetrates microbial cell walls far more readily than ClOŌü╗ and oxidizes cellular components with greater efficiency. This molecular behaviour explains why disinfecting ability peaks within a moderate pH range and decreases when the solution is too alkaline or too acidic. Understanding this relationship between structure and function is essential in fields ranging from wastewater treatment to food sanitation and medical sterilization.
In non-biological chemistry, sodium hypochlorite demonstrates additional oxidation pathways. It converts alcohols to aldehydes or ketones, iodide ions to iodine, and certain organic compounds to chlorinated derivatives. These transformations occur because the chlorine atom in the +1 oxidation state seeks electrons to reach a more stable oxidation state. Industrial bleaching of pulp and textiles also relies on hypochloriteŌĆÖs ability to disrupt conjugated double bonds in chromophoric molecules ŌĆö the structures responsible for coloration ŌĆö by breaking the electron systems that absorb visible light.
The structural behaviour of sodium hypochlorite under heat and light provides further insight into its chemistry. Hypochlorite is thermodynamically unstable relative to chloride and chlorate and decomposes slowly even at room temperature. Heat, and particularly ultraviolet light, accelerate this decomposition, producing chloride and chlorate ions. This behaviour illustrates that oxidation state transitions do not require dramatic reaction conditions ŌĆö they occur whenever the molecular structure naturally shifts toward states of lower energy. This instability is why commercial sodium hypochlorite solutions include stabilizing agents and are stored in opaque containers.
Despite strong reactivity, sodium hypochloriteŌĆÖs safety depends entirely on concentration and environment. In dilute solutions, it disinfects without significant risk. In concentrated solutions, its oxidizing power can damage skin, eyes, and organic materials. When mixed with acids, toxic chlorine gas may be released due to the equilibrium shift from hypochlorite to HOCl to ClŌéé. When mixed with ammonia or amine-containing cleaners, dangerous nitrogen-chlorine compounds may form. These behaviours demonstrate that hazards arise not from the chemical alone but from inappropriate reaction environments ŌĆö a principle central to chemical safety education.
Sodium hypochlorite is a near-perfect teaching model because it connects multiple domains of chemistry in one structure. It exemplifies ionic bonding in solids, solvation and electrolyte formation in water, acidŌĆōbase equilibria, redox transformations, stability influenced by oxidation state, biological impact driven by electron transfer, and thermal and photochemical decomposition pathways. The sodium ion contributes stability and solubility but does not drive chemical transformation. The hypochlorite ion ŌĆö through covalent bonding, charge distribution, and chlorineŌĆÖs oxidation state ŌĆö determines every major behaviour, from bleaching to disinfection to decomposition.
Ultimately, sodium hypochlorite shows that chemistry is the science of electrons shaping matter. A single chlorine atom bonded to a single oxygen atom ŌĆö carrying an uneven charge distribution and existing in a high oxidation state ŌĆö becomes a chemical system that influences sanitation, medicine, industry, and public health. Its behaviour demonstrates the governing principle of molecular science: structure determines function, and by understanding structure we can predict and control the behaviour of the material world.