ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉāØÉóØÉ£ØÉĪØɽØÉ©ØÉ”ØÉÜØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉéØÉĪØɽØÉ©ØÉ”ØÉóØÉ«ØÉ” ØÉÄØÉ▒ØÉ▓ØÉÜØɦØÉóØÉ©Øɦ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ ØÉÜØɦØÉØ ØÉāØÉ×ØÉ×ØÉ® ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
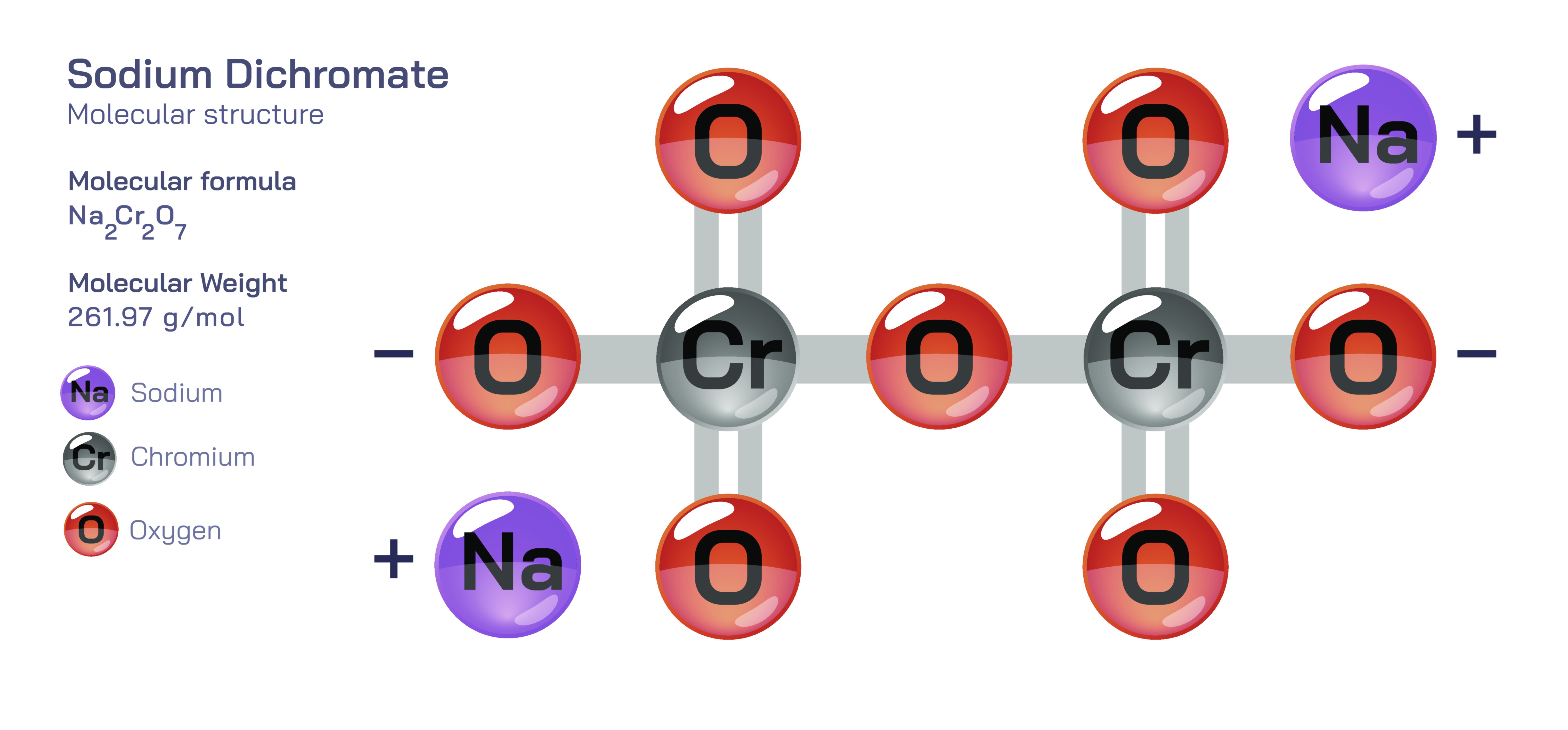
Sodium dichromate, typically written as NaŌééCrŌééOŌéć, is a highly important inorganic salt composed of sodium ions and the dichromate oxyanion. To the naked eye, sodium dichromate appears as bright orange-red crystals, but at the molecular level it represents a structurally rich and chemically powerful example of an oxyanion containing chromium in an unusually high oxidation state. This compound demonstrates how electron distribution, resonance, polyatomic ion geometry and oxidationŌĆōreduction tendencies shape the behaviours and applications of materials. Chemically, it stands beside sodium chromate as a member of the chromium(VI) family of oxyanions, but the distinction between chromate and dichromate illustrates key principles of chemical equilibria and the pH-dependent transformation of ions in solution. Understanding sodium dichromate provides insight not only into atomic bonding but into acidŌĆōbase chemistry, redox chemistry, industrial catalysis and environmental behaviour.
The core structural unit of sodium dichromate is the dichromate ion, CrŌééOŌéć┬▓Ōü╗. This ion contains two chromium atoms, each in the +6 oxidation state, linked through a shared oxygen bridge. Each chromium atom is tetrahedrally coordinated by oxygen atoms, forming two interconnected tetrahedra that share one oxygen. The remaining oxygen atoms exist as terminal oxygens bonded to chromium, with resonance distributing electron density across multiple CrŌĆōO bonds so that no simple alternating singleŌĆōdouble bond model accurately represents the ion. This extended polyatomic structure is a critical feature because it creates stability through resonance while simultaneously leaving chromium extremely electron-deficient. That electron deficiency underlies the powerful oxidizing ability of dichromate ions, which seek electrons from reducing agents in both organic and inorganic systems.
The sodium ions in sodium dichromate act as spectator cations that balance the total charge without participating directly in redox or structural shifts. Each NaŌü║ ion, formed by the loss of a single valence electron from elemental sodium, contributes to stabilizing the oxyanion electrostatically. In the solid state, NaŌü║ ions and CrŌééOŌéć┬▓Ōü╗ ions pack into a crystalline lattice held together by ionic attraction. This lattice architecture, rather than discrete NaŌééCrŌééOŌéć molecules, accounts for the hardness, brittleness and high melting point of sodium dichromate. When dissolved in water, hydration energy allows water molecules to break the ionic lattice apart. The sodium ions become freely solvated, while the dichromate ions remain intact but exist in equilibrium with chromate ions (CrOŌéä┬▓Ōü╗). This equilibrium illustrates a central educational principle: molecular identity in aqueous systems can depend on pH. At lower pH (more acidic environments), dichromate is the dominant species; at higher pH, chromate ions prevail. This transformation does not change the oxidation state of chromium but reorganizes oxygen bridges in response to proton concentration.
The oxidizing behaviour of sodium dichromate is one of its most defining features. Chromium in the +6 oxidation state has a strong thermodynamic tendency to gain electrons and be reduced to chromium(III), a far more stable oxidation state. This redox shift is the driving force behind many reactions of sodium dichromate. In acidic media, dichromate oxidizes a wide variety of substancesŌĆöincluding alcohols, aldehydes, organic fuels and certain metal ionsŌĆöwhile being reduced itself to Cr┬│Ōü║. The dramatic colour change accompanying this process, from orange dichromate to green chromium(III), visually reinforces electron transfer and redox chemistry. The redox behaviour of dichromate shows that oxidation and reduction are not random; they follow energetic pathways in which atoms adjust their electronic configuration toward greater stability.
Because of its oxidizing power, sodium dichromate is widely used industrially in metal finishing, pigment manufacture, corrosion control, wood preservation and organic synthesis. For example, dichromate ions convert alcohols to ketones or carboxylic acids through controlled oxidation, illustrating the capability of an inorganic compound to guide organic transformations through electron transfer. In anti-corrosion coatings, dichromate stabilizes metal surfaces, particularly zinc and aluminum, by forming protective chromium(III)-containing films that resist further oxidation. These roles highlight how single structural featuresŌĆöthe high oxidation state of chromium and the resonance-stabilized oxyanion frameworkŌĆötranslate into multiple technological uses.
In environmental and biological contexts, sodium dichromate requires careful handling due to the reactivity that makes it useful industrially. The same tendency of dichromate ions to accept electrons makes them reactive toward biological molecules. In living cells, the electron-transfer potential of chromium(VI) leads to oxidative damage and disruption of biochemical processes. This reinforces a fundamental scientific lesson: toxicity is linked not to the presence of an element alone but to its oxidation state and electron-accepting behaviour. Chromium(III) compounds are far less reactive and are considered micronutrients in trace amounts, whereas chromium(VI) compounds like dichromate are hazardous because of their aggressive electron-seeking nature.
The behaviour of sodium dichromate under heat also reflects structureŌĆōfunction relationships. Dichromate ions remain stable under mild heating, as the CrŌééOŌéć┬▓Ōü╗ unit is strongly bonded through resonance-stabilized oxygen networks. Under high heat or in the presence of reducing agents, however, the compound decomposes, releasing oxygen and forming lower oxidation state chromium compounds. This decomposition is again driven by the chromate-to-chromium(III) reduction process, showing that the ionic lattice and polyatomic structure are resilient until electronic reorganization becomes energetically favourable.
The educational significance of sodium dichromate lies in the many principles it illustrates simultaneously. It demonstrates resonance bonding and extended structures in polyatomic ions, the concept of oxidation states and how they determine reactivity, the formation and dissolution of ionic lattices, the pH-controlled equilibrium between chromate and dichromate ions, electron-transfer mechanisms in redox reactions, and the relationship between electron structure and biological and technological impact. In this one compound, students can observe how the invisible world of electrons governs the macroscopic visible world, from colour changes in the beaker to protective coatings on metals to environmental safety concerns.
Thus, sodium dichromate is not merely an industrial chemical; it is a powerful instructional model. Its vivid colour, dramatic redox shifts, pH-dependent equilibria and transition-metal oxyanion structure reveal the deep interconnections among bonding, electron distribution and reactivity. In studying NaŌééCrŌééOŌéć, learners discover that chemistry is the science of structure guiding behaviour, and that understanding a compoundŌĆÖs molecular and electronic identity allows us to interpret, predict and control the material world.