ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉīØÉ×ØÉŁØÉÜØÉøØÉóØɼØÉ«ØÉźØɤØÉóØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉłØÉ©ØɦØÉóØÉ£ŌĆōØÉéØÉ©ØÉ»ØÉÜØÉźØÉ×ØɦØÉŁ ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉæØÉ×ØÉØØÉ©ØÉ▒ ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ, ØÉÆØÉ©ØÉźØÉ«ØÉŁØÉóØÉ©Øɦ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉÜØɦØÉØ ØÉĆØÉØØÉ»ØÉÜØɦØÉ£ØÉ×ØÉØ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
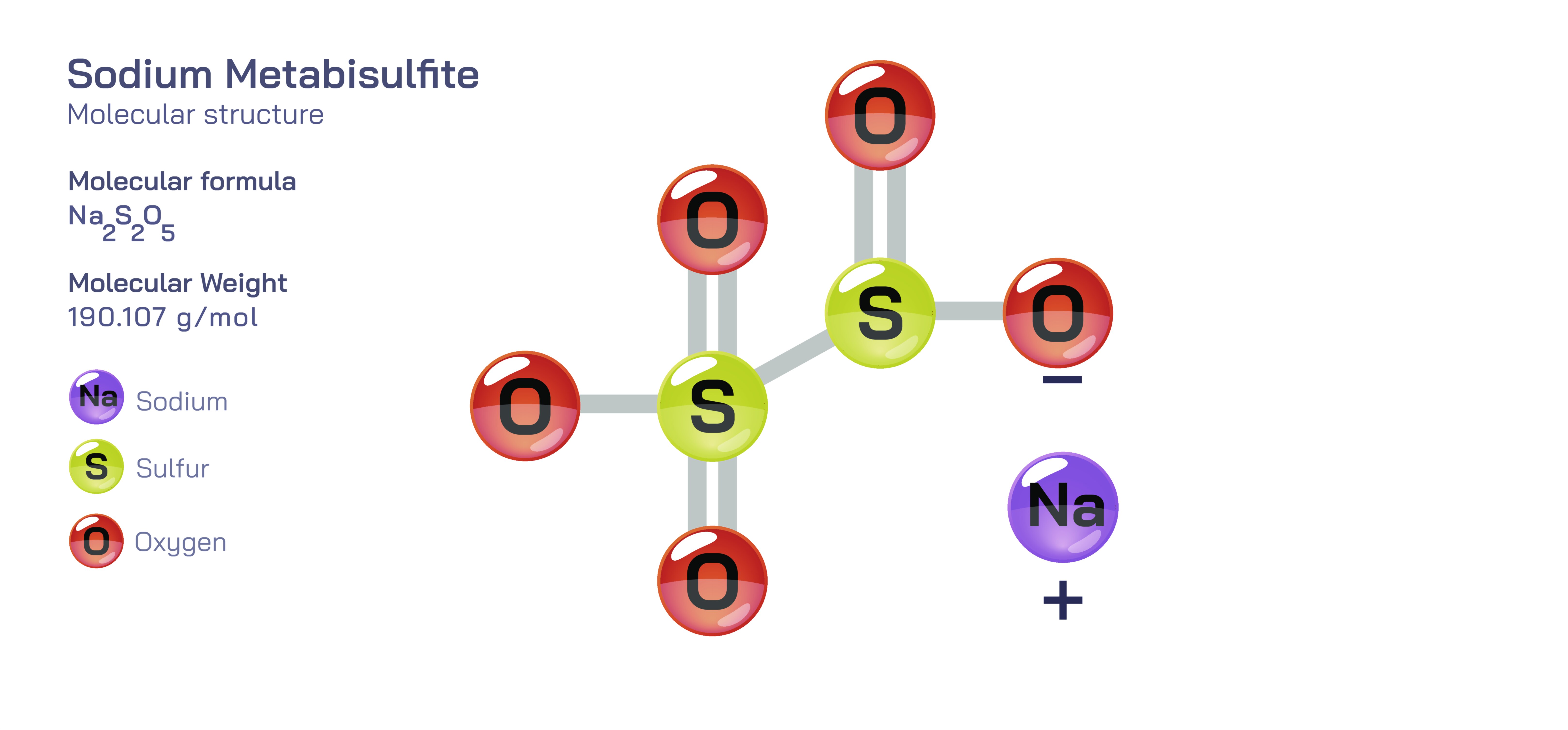
Sodium metabisulfite, chemically written as NaŌééSŌééOŌéģ, is an inorganic salt composed of two sodium ions and a polyatomic anion known as the metabisulfite ion (SŌééOŌéģ┬▓Ōü╗). Although often described as a simple preservative or antioxidant used in food, beverages and pharmaceuticals, its true importance emerges through its molecular architecture and redox chemistry. At the microscopic level, sodium metabisulfite contains a polyatomic anion that demonstrates a unique combination of covalent bonding between atoms inside the ion and ionic attraction in the solid. Its reactivity, particularly its ability to release sulfur dioxide (SOŌéé), is a direct consequence of the internal structure of the SŌééOŌéģ┬▓Ōü╗ ion. Studying sodium metabisulfite allows learners to explore acidŌĆōbase chemistry, redox reactions, resonance behaviour, equilibrium principles and solvation dynamics in aqueous systems.
The heart of sodium metabisulfite chemistry lies in the metabisulfite ion, a polyatomic anion containing two sulfur atoms bonded through a bridging oxygen atom, with the remaining oxygen atoms arranged in a bent geometry around each sulfur. One sulfur atom exists in a higher oxidation state than the other, a feature that drives redox behaviour and internal electron distribution within the ion. Each sulfur atom is covalently bonded to oxygen atoms, but the electron density is not confined to fixed bonds; instead, resonance distributes negative charge over the oxygen framework. The -2 charge of the anion is delocalized, giving stability to the polyatomic structure while still leaving it capable of reacting strongly with oxidizing agents and acidic environments.
The sodium ions in sodium metabisulfite are created when elemental sodium loses its single valence electron and becomes NaŌü║. These ions do not contribute to the reactive chemistry of the compound but balance the overall charge of the SŌééOŌéģ┬▓Ōü╗ anion. In the solid state, the compound forms an extended ionic lattice ŌĆö not isolated NaŌééSŌééOŌéģ molecules ŌĆö where sodium ions and metabisulfite ions alternate in a repeating array. The rigidity and high melting point of the solid are due to ionic bonding between NaŌü║ and SŌééOŌéģ┬▓Ōü╗, while the internal structure of the metabisulfite ion remains governed by covalent forces. This duality makes sodium metabisulfite a valuable teaching example showing that substances can contain both covalent and ionic bonding simultaneously, depending on whether interactions occur within ions or between them.
The behaviour of sodium metabisulfite transforms completely when it dissolves in water. Hydration energy from water molecules disrupts the ionic lattice and releases NaŌü║ and SŌééOŌéģ┬▓Ōü╗ into solution. However, the metabisulfite ion does not remain static in water; it undergoes equilibrium with bisulfite (HSOŌéāŌü╗) and sulfite (SOŌéā┬▓Ōü╗) ions, depending on the pH of the system. In acidic environments, the metabisulfite ion is protonated, forming bisulfite and releasing sulfur dioxide gas (SOŌéé), which is responsible for the preservative and antimicrobial properties of the compound. The reversible equilibrium between the dissolved anions and gaseous SOŌéé demonstrates how pH can control the identity and behaviour of chemical species. In neutral or alkaline conditions, metabisulfite tends to shift toward sulfite ions, while in strongly acidic conditions the equilibrium shifts toward SOŌéé formation.
The redox chemistry of sodium metabisulfite is one of its most defining features. The metabisulfite ion acts as a reducing agent, meaning it donates electrons to other substances during chemical reactions. This is due to the combination of sulfur oxidation states within the structure and the presence of electron-rich oxygen atoms capable of transferring electron density. During reduction, metabisulfite oxidizes to sulfate (SOŌéä┬▓Ōü╗), and this transformation reflects the overall tendency of the system to reach a more electronically stable configuration. This redox transition is widely used in industrial applications, including water treatment, food preservation, photographic processing, and chemical synthesis, where metabisulfite removes oxygen or prevents oxidation of sensitive molecules.
In food chemistry and fermentation science, sodium metabisulfite inhibits microbial growth not by acting as a ŌĆ£poisonŌĆØ but by interrupting metabolic oxidation pathways. SOŌéé generated in acidic solution reacts with key enzymes and cofactors in microorganisms, preventing them from accessing oxygen for respiration. The antimicrobial effect is therefore rooted in electron-transfer chemistry, not physical toxicity. In winemaking and beverage production, sodium metabisulfite prevents browning and spoilage by eliminating dissolved oxygen and neutralizing oxidizing compounds that would otherwise degrade flavour, aroma and colour. The chemical basis of preservation is simply controlled redox behaviour.
In analytical chemistry, sodium metabisulfite serves as a selective reducing agent used to quench reactions, destroy excess oxidizing compounds and regenerate reduced forms of chemical indicators. In polymer and textile industries, it participates in bleaching and dechlorination processes by neutralizing chlorine-based oxidants. In wastewater treatment, it removes residual chlorine from discharge streams because metabisulfite converts chlorine to chloride ions. In each case, the compound acts not through physical adsorption or dilution but through electron donation, demonstrating how reactivity arises from molecular structure.
The thermal behaviour of sodium metabisulfite also reflects its internal bonding. When heated without moisture, the compound decomposes into sodium sulfite (NaŌééSOŌéā) and sulfur dioxide gas. This process occurs because the bridging oxygen and sulfur arrangement within the metabisulfite ion is thermodynamically unstable at high temperatures and breaks into simpler ions and gaseous products. In the presence of sufficient heat and oxygen, complete oxidation yields sodium sulfate (NaŌééSOŌéä). These reactions show that the compound behaves differently depending on the surrounding atmosphere ŌĆö another illustration that chemical behaviour depends on available electron transfer partners.
Environmental and biological aspects of sodium metabisulfite stem directly from its chemical behaviour. In small, controlled quantities, it protects food and beverages through antioxidant activity, but in excessive or uncontrolled concentrations, the release of SOŌéé can irritate respiratory tissues. The compound itself is not persistent in the environment because it naturally oxidizes to sulfate, a stable and widely occurring ion in nature. This reinforces a principle that hazard depends on concentration and context: a compound that performs a protective function in one setting can be harmful in another if conditions change.
From an educational perspective, sodium metabisulfite is extremely valuable because it unites several pillars of chemistry in one compound: ionic bonding, covalent bonding, oxidation states, reducing behaviour, acidŌĆōbase equilibrium, solvation dynamics, equilibrium gas release and thermally driven decomposition. Its sodium ions demonstrate the stabilizing role of spectator cations. Its metabisulfite ion illustrates how electron distribution across a polyatomic structure determines reactivity. Its release of SOŌéé shows that gases can form through reversible ionic rearrangements rather than combustion or evaporation.
Ultimately, sodium metabisulfite demonstrates a central lesson of chemistry: electrons and molecular geometry determine function. The presence of two covalently bonded sulfur atoms, one bridging oxygen, multiple resonance-stabilized oxygen atoms and a ŌłÆ2 charge creates a structure capable of reducing oxidants, binding protons, shifting between ionic species and generating gas. These behaviours control major applications in food, medicine, water treatment and manufacturing. NaŌééSŌééOŌéģ is therefore not simply a preservative ŌĆö it is a vivid example of how atomic structure, electron distribution and bonding forces dictate every observable property of matter in both industrial systems and living environments.