𝐒𝐨𝐝𝐢𝐮𝐦 𝐈𝐨𝐝𝐢𝐝𝐞 — 𝐌𝐨𝐥𝐞𝐜𝐮𝐥𝐚𝐫 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞, 𝐈𝐨𝐧𝐢𝐜 𝐁𝐨𝐧𝐝𝐢𝐧𝐠, 𝐄𝐥𝐞𝐜𝐭𝐫𝐨𝐧𝐢𝐜 𝐁𝐞𝐡𝐚𝐯𝐢𝐨𝐮𝐫, 𝐑𝐞𝐚𝐜𝐭𝐢𝐯𝐢𝐭𝐲 𝐚𝐧𝐝 𝐃𝐞𝐞𝐩 𝐂𝐡𝐞𝐦𝐢𝐬𝐭𝐫𝐲 𝐄𝐝𝐮𝐜𝐚𝐭𝐢𝐨𝐧𝐚𝐥 𝐒𝐮𝐦𝐦𝐚𝐫𝐲.
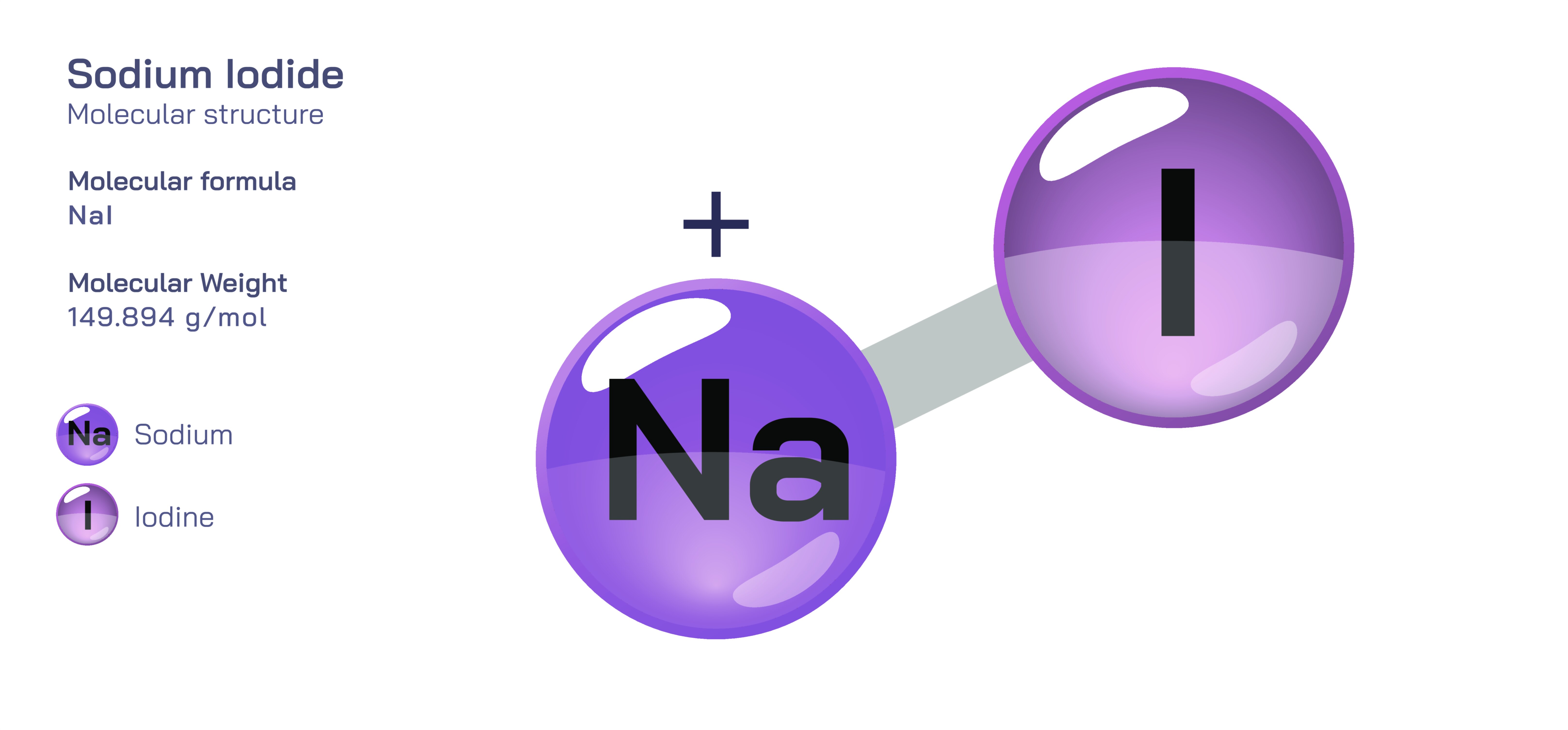
Sodium iodide, represented chemically as NaI, is an ionic inorganic compound best known for its use in medical imaging, nutrition, analytical chemistry and radiography. Although it appears externally as a simple white crystalline solid, its molecular and electronic architecture reveals a rich story about ionic bonding, periodic trends among halides, solvation dynamics in water, reactivity driven by redox potentials and the ability of ions to participate in biological and technological processes. Understanding sodium iodide means recognizing how electron transfer creates forces that shape the properties of matter, and how the identity of a halide ion determines the reactivity of salts that otherwise appear nearly identical.
At the structural level, sodium iodide consists of two ionic components: the sodium cation (Na⁺) and the iodide anion (I⁻). Sodium, an alkali metal from Group 1, has one valence electron that it readily loses in order to reach a stable noble-gas electron configuration. When this electron is released, the atom becomes a positively charged Na⁺ ion — electronically stable and therefore chemically non-aggressive. Iodine, a halogen from Group 17, has seven valence electrons and needs one more to complete its outer shell. When iodine accepts an electron, it becomes I⁻, an anion with a full octet and a large, diffuse electron cloud. This electron transfer from sodium to iodine forms the Na⁺ and I⁻ ions, setting the stage for ionic bonding. Unlike covalent bonding, where electrons are shared, ionic bonding stems from electrostatic attraction between ions of opposite charge, and this attraction is the foundational force that governs the structure of sodium iodide.
In the solid state, NaI does not exist as isolated “NaI molecules.” Instead, it forms a three-dimensional ionic lattice in which each sodium ion is surrounded by iodide ions and each iodide ion is surrounded by sodium ions. This lattice arrangement maximizes attraction between positive and negative ions while minimizing repulsion between ions of the same charge. The resulting structure accounts for the physical properties of sodium iodide: a definite crystalline shape, brittleness and a relatively high melting point. The lattice expands uniformly throughout the solid, emphasizing the idea that ionic compounds exist not as individual units but as extended networks of ions.
The behaviour of sodium iodide changes dramatically when the compound dissolves in water. Water molecules disrupt the ionic lattice through the process of hydration. The partially negative oxygen ends of water orient toward sodium ions, while the partially positive hydrogen ends orient toward iodide ions. This hydration releases enough energy to overcome ionic attractions in the solid, freeing Na⁺ and I⁻ into solution. Once dissolved, sodium iodide becomes a strong electrolyte capable of conducting electricity because the ions are mobile. This dissolution process illustrates several central principles of chemistry: solubility is governed by energy competition between ionic attraction and hydration, and electrical conductivity in solutions results from the movement of ions.
The iodide ion plays the dominant chemical role in the reactivity of sodium iodide. Iodide is a large, polarizable anion with a low electronegativity relative to other halides, meaning its electron cloud can deform easily and donate electrons readily in reactions. These characteristics make iodide a relatively strong reducing agent, capable of donating electrons to electron-poor species such as oxidizing agents or transition metals. During such reactions, iodide is converted to elemental iodine, I₂, which appears purple in organic solvents and reddish-brown in aqueous systems. This colour change provides one of the most dramatic and widely taught demonstrations of oxidation–reduction chemistry and reinforces the fundamental concept that redox processes involve the movement of electrons rather than the physical mixing of reactive substances.
The nucleophilic nature of iodide plays a central role in organic chemistry. Because the iodide ion donates electrons easily and forms bonds with carbon rapidly, it is used in substitution reactions, particularly in halogen exchange processes. When sodium iodide reacts with alkyl bromides or chlorides in polar aprotic solvents, iodide replaces the halogen in an SN2 mechanism, forming alkyl iodides. This transformation is driven not by sodium ions but by the iodide anion, which offers less steric hindrance than other nucleophiles and provides strong leaving-group stability once it departs from organic molecules. In this context, sodium iodide becomes a key synthetic tool that demonstrates how the microscopic characteristics of ions determine reaction speed and mechanism.
Sodium iodide also demonstrates the importance of periodic trends in halide chemistry. Moving down the halogen group, halide ionic radius increases while electronegativity decreases. Fluoride forms extremely stable ionic lattices but is a weak nucleophile; chloride is moderately nucleophilic; bromide is stronger; and iodide is the strongest nucleophile in polar aprotic systems. Sodium iodide therefore serves as a clear teaching example that chemical reactivity — even between compounds that differ only by a halogen — depends on atomic and electronic properties rather than on overall formula.
In biological and medical contexts, sodium iodide provides one of the clearest illustrations of how ions influence physiology. Iodide is an essential micronutrient that participates in the synthesis of thyroid hormones. Without sufficient iodide, the thyroid gland cannot produce hormones responsible for metabolic regulation, leading to enlargement of the gland (goiter) and metabolic disorders. Because the body cannot synthesize iodine on its own, iodide must be supplied through diet, and sodium iodide — as an easily absorbed ionic compound — has become a principal supplement form. The biological role of NaI therefore stems not from sodium, but from iodine, whose electronegativity and bonding behaviour enable the formation of life-critical organoiodine compounds.
The compound also plays a crucial role in radiation science, especially when enriched with radioisotopes of iodine. Sodium iodide activated with thallium (NaI:Tl) is widely used in gamma-ray detection because the ionic crystal lattice efficiently converts radiation energy into visible photons. This fluorescence property is another example of how electron behaviour at the atomic level determines the technological usefulness of a material. The ability of the iodide ion to stabilize various oxidation states and support crystal-lattice photophysics makes NaI one of the most important detectors in nuclear medicine and scientific instrumentation.
Even properties that seem unrelated — stability, solubility, melting point, or sensitivity to air — reflect structural chemistry. Sodium iodide is hygroscopic, meaning it absorbs water from the atmosphere. This behaviour arises because the ionic lattice readily hydrates, and the iodide ion forms strong hydrogen-bonding interactions with water molecules. In the presence of oxygen and moisture, iodide can slowly oxidize to iodine, giving aged samples a yellow or brown tint. This visual effect underscores that oxidation–reduction is a continuous competition for electrons between ions and the environment.
From an educational perspective, sodium iodide is much more than a laboratory reagent. It demonstrates that the gain and loss of a single electron transforms two neutral atoms into ions that create an organized solid, dissolve to form electrolytes, induce biological functions, drive industrial processes, and enable advanced scientific technologies. The sodium ion contributes structural stability and solubility but does not direct reactivity. The iodide ion, with its large radius, high polarizability, nucleophilic strength and redox activity, dictates nearly every significant property of the compound.
In the broadest sense, sodium iodide illustrates the core rule of chemistry: structure dictates behaviour. The movement of one electron from sodium to iodine — a seemingly tiny adjustment — gives rise to an entire world of macroscopic phenomena: bonding architecture, solubility, conductivity, redox reactivity, organic substitution pathways, nutritional biology and radiation detection. Through NaI, one sees that chemistry is not the study of formulas on paper but of electrons shaping matter, reactions and life itself.