ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉüØÉóØÉ£ØÉÜØɽØÉøØÉ©ØɦØÉÜØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉüØÉ×ØÉĪØÉÜØÉ»ØÉóØÉ©ØÉ«Øɽ ØÉÜØɦØÉØ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉéØÉ©ØɦØÉ£ØÉ×ØÉ®ØÉŁ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
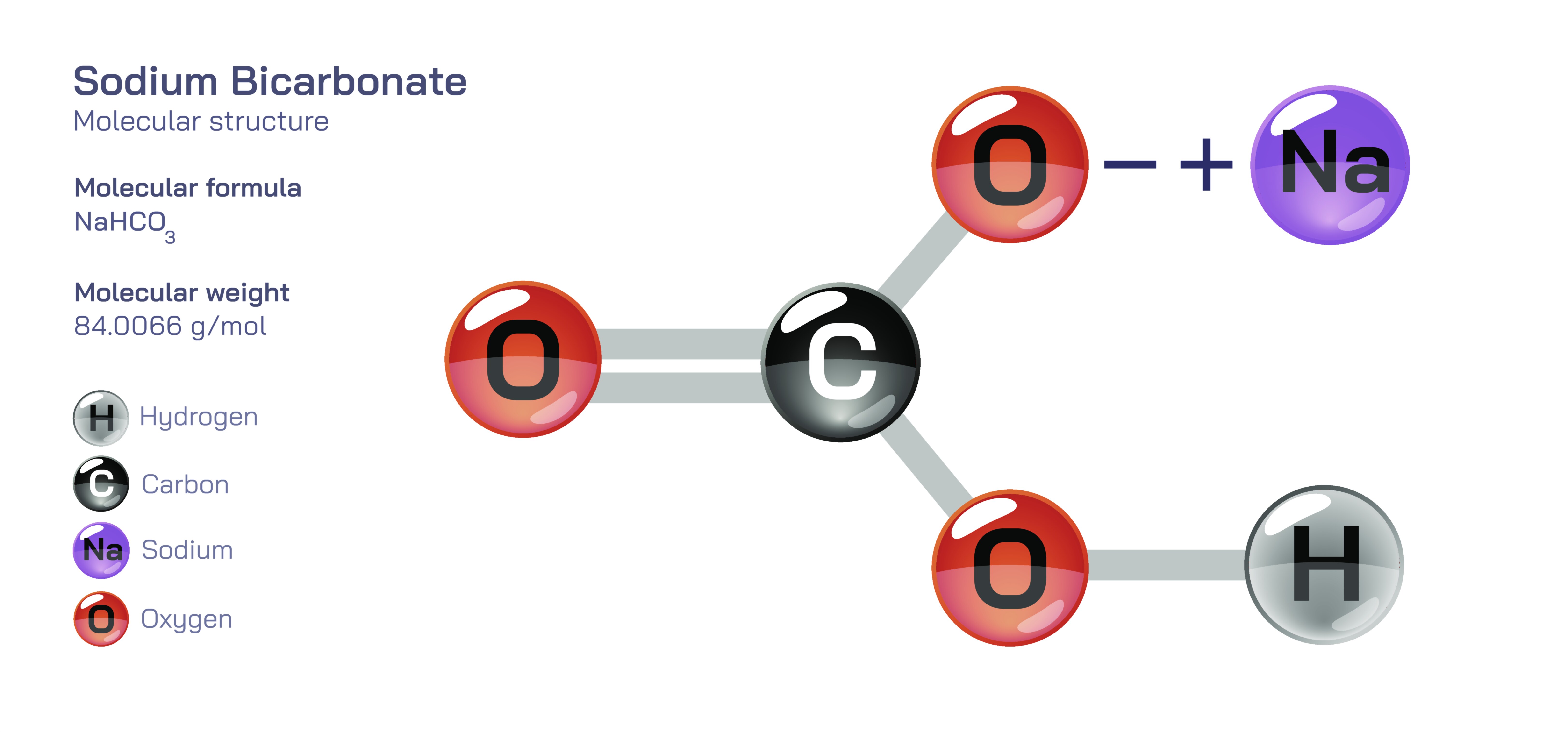
Sodium bicarbonate, commonly known as baking soda and represented by the chemical formula NaHCOŌéā, is a widely used inorganic compound with a distinctive molecular structure that gives rise to versatile chemical behaviour. Although it appears as a simple white crystalline powder, the compound demonstrates complex interactions in solution, in heat-driven reactions, in buffering systems and in biological environments. Understanding sodium bicarbonate means understanding how the bicarbonate ion behaves in water, how acidŌĆōbase equilibria influence its reactions and how the structure of the HCOŌéāŌü╗ ion determines everything from its mild alkalinity to its decomposition into carbon dioxide. Its molecular identity links everyday applicationsŌĆösuch as baking, cleaning and antacid therapyŌĆöto deep principles of chemistry including equilibria, solubility, thermodynamics and environmental cycles.
At the heart of sodium bicarbonate lies the bicarbonate ion (HCOŌéāŌü╗), which originates from carbonic acid (HŌééCOŌéā). Structurally, the bicarbonate ion contains a central carbon atom bonded to three oxygen atoms. One of those oxygen atoms is attached by a double bond, while the other two are single-bonded oxygensŌĆöone of which carries a hydrogen atom and the other carries the negative charge. However, this negative charge is not restricted to a single point; resonance distributes electron density across the oxygen atoms by shifting the double bond between them. This delocalized structure provides stability to the ion and explains its ability to act both as a weak base and as a weak acid, depending on chemical surroundings. When sodium ions pair with bicarbonate ions, an ionic compound forms through electrostatic attraction. In the solid lattice, NaŌü║ ions maintain charge neutrality with HCOŌéāŌü╗ ions, but when dissolved in water, the lattice breaks apart because water molecules solvate both species effectively. The sodium ion remains fully hydrated and unreactive, while the bicarbonate ion instantly enters equilibrium reactions that shape its behaviour.
The most defining property of sodium bicarbonate in solution is its amphoteric behaviourŌĆömeaning it can both donate and accept protons. The bicarbonate ion sits at the midpoint of the acidŌĆōbase spectrum within the carbonic acid system. In the presence of a strong acid, the bicarbonate ion accepts a proton to form carbonic acid (HŌééCOŌéā), which instantly decomposes into water and carbon dioxide gas. This well-known reaction explains baking sodaŌĆÖs vigorous bubbling when combined with vinegar or lemon juice. In the presence of a strong base, the bicarbonate ion donates a proton to form carbonate (COŌéā┬▓Ōü╗). These reversible transformations illustrate that bicarbonate forms part of a dynamic equilibrium system, switching between carbonic acid, bicarbonate and carbonate depending on pH. This behaviour is the reason sodium bicarbonate is an effective buffering agent, capable of resisting sudden changes in pH. In biological systems, this ion equilibrium is vital: bicarbonate is a major component of the bloodŌĆÖs buffering system, helping maintain the narrow pH range necessary for life.
The production of carbon dioxide gas in acidŌĆōbase reactions makes sodium bicarbonate an essential compound in baking and cooking. When baking soda is mixed into dough or batter and exposed to acidic ingredientsŌĆösuch as buttermilk, yogurt, lemon juice or cocoaŌĆöbicarbonate instantly reacts to form COŌéé gas. The gas becomes trapped in the stretchy protein network of dough and gradually expands as heat accelerates the reaction, causing baked goods to rise and achieve a soft, aerated texture. This culinary process reflects deeper chemical thermodynamics: as temperature increases, bicarbonate undergoes thermal decomposition even without added acids, forming sodium carbonate, water and carbon dioxide. This gas-releasing thermal breakdown explains why sodium bicarbonate also appears in fire extinguishers, where rapid release of COŌéé helps smother flames by displacing oxygen. The dual effectŌĆögas evolution from both acid contact and heatŌĆöshows how molecular structure determines multiple functions stemming from the same reaction pathways.
Sodium bicarbonate is widely used as a mild antacid due to its predictable reaction with stomach acid. When ingested in small amounts, it neutralizes hydrochloric acid (HCl) to form sodium chloride, water and carbon dioxide. The relief that follows is the direct result of the bicarbonate ionŌĆÖs buffering capacity. Unlike stronger alkalis such as sodium hydroxide, sodium bicarbonate does not create highly caustic conditions because the conjugate acid formedŌĆöcarbonic acidŌĆöis weak and transient. This selective behaviour makes sodium bicarbonate safe when used properly. In medicine and physiology, bicarbonate solutions are also employed intravenously to counter dangerous acidosis when blood pH falls too low. These clinical uses highlight how the same acidŌĆōbase equilibrium that lifts bread in baking can support life-critical biochemical regulation.
In cleaning and deodorizing applications, sodium bicarbonate displays properties linked to both its molecular charge distribution and mild alkalinity. Because the bicarbonate ion is a moderately strong proton acceptor, it neutralizes acidic odour molecules and allows them to convert into less volatile and less irritant forms. Additionally, the crystalline powder has gentle abrasiveness and dissolves gradually in water, helping remove grime and stains without scratching surfaces. In laundry applications, bicarbonate softens water by binding metal ions, improving soap effectiveness. These uses rely not on high chemical strength, but on well-controlled reactivityŌĆösoft alkalinity and equilibrium-based neutralization rather than aggressive corrosion.
Thermally induced decomposition of sodium bicarbonate provides insight into its structural transition behaviour. When heated, the bicarbonate ion does not melt into a liquid but instead chemically transforms, yielding sodium carbonate (NaŌééCOŌéā), water vapour and carbon dioxide gas. This decomposition is endothermic at first but becomes self-sustaining once initiated. The formation of sodium carbonate instead of leaving free sodium ions shows that the carbonate family maintains bonding even during transition. Industrially, this decomposition is controlled to manufacture soda ash (NaŌééCOŌéā), a major chemical used in glassmaking, detergents and metallurgy. Thus, a kitchen staple like baking soda becomes a precursor to versatile industrial raw materials, illustrating again that the behaviour of compounds reflects atomic connectivity.
The bicarbonate ion is also central to environmental chemistry. Oceans use carbonateŌĆōbicarbonate equilibria to moderate pH and dissolve carbon dioxide from the atmosphere. In water bodies, bicarbonate prevents rapid acidification and supports aquatic life by limiting extreme pH fluctuations. Soil chemistry also depends on bicarbonate levels, especially in agriculture where carbonateŌĆōbicarbonate systems influence nutrient availability. These natural cycles reveal that sodium bicarbonate belongs not only to human usage but to EarthŌĆÖs geochemical systems. Carbonate chemistry affects coral reef formation, atmospheric balance and long-term climate processes, proving that bicarbonate ionsŌĆöso familiar in the kitchenŌĆöplay a significant planetary role.
In biological systems, bicarbonate participates in respiration and metabolic regulation. In blood plasma, the reaction between carbon dioxide, carbonic acid, bicarbonate and hydrogen ions enables breathing to regulate pH. When COŌéé levels rise, blood becomes slightly more acidic and bicarbonate concentrations adjust to stabilize conditions. The kidneys and lungs work together to fine-tune this balance, excreting or retaining bicarbonate as needed. This physiological buffering is not incidental; it is a direct consequence of bicarbonateŌĆÖs intermediate proton affinity and resonance-stabilized structure.
Although sodium bicarbonate is generally safe, its effects depend on dosage and context. Ingesting excessive amounts can disrupt electrolyte balance or cause alkalosis due to increased systemic pH. Rapid formation of COŌéé gas in the stomach can lead to discomfort or, in extreme cases, internal pressure risk. Because bicarbonate decomposes to carbonate at high pH, overuse can produce undesirable alkalinity in soil or water treatment applications. These risks are not inherent dangers of the molecule but rather consequences of misuse or imbalance.
Ultimately, sodium bicarbonate exemplifies how the identity of a compound arises not from elemental composition alone but from molecular structure and equilibrium behaviour. The bicarbonate ion is neither strongly acidic nor strongly basic; its resonance-stabilized negative charge makes it responsive to environmental pH in a way that creates stability rather than extremes. This balance gives sodium bicarbonate the ability to release carbon dioxide when needed, resist pH changes when buffering is required, and gently neutralize acids rather than overwhelm them. It plays crucial roles in baking, medicine, cleaning, fire suppression, industry, physiology and the natural carbon cycle because the same molecular organization supports all these functions.
Sodium bicarbonate therefore stands as an educational model of how chemistry permeates everyday lifeŌĆönot through exotic substances but through the subtle power of structure, bonding and equilibria. A simple spoonful of baking soda illustrates acidŌĆōbase theory, solubility principles, thermodynamics, gas evolution, biological regulation and environmental chemistryŌĆöshowing clearly that the language of molecular structure explains both the smallest reactions in a beaker and the largest buffering processes on Earth.