ØÉÆØÉóØÉźØÉ»ØÉ×Øɽ ØÉÄØÉ▒ØÉóØÉØØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ, ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉÜØɦØÉØ ØÉģØÉ«ØɦØÉ£ØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÅØɽØÉ©ØÉ®ØÉ×ØɽØÉŁØÉóØÉ×Øɼ ŌĆö ØÉŗØÉ©ØɦØÉĀ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
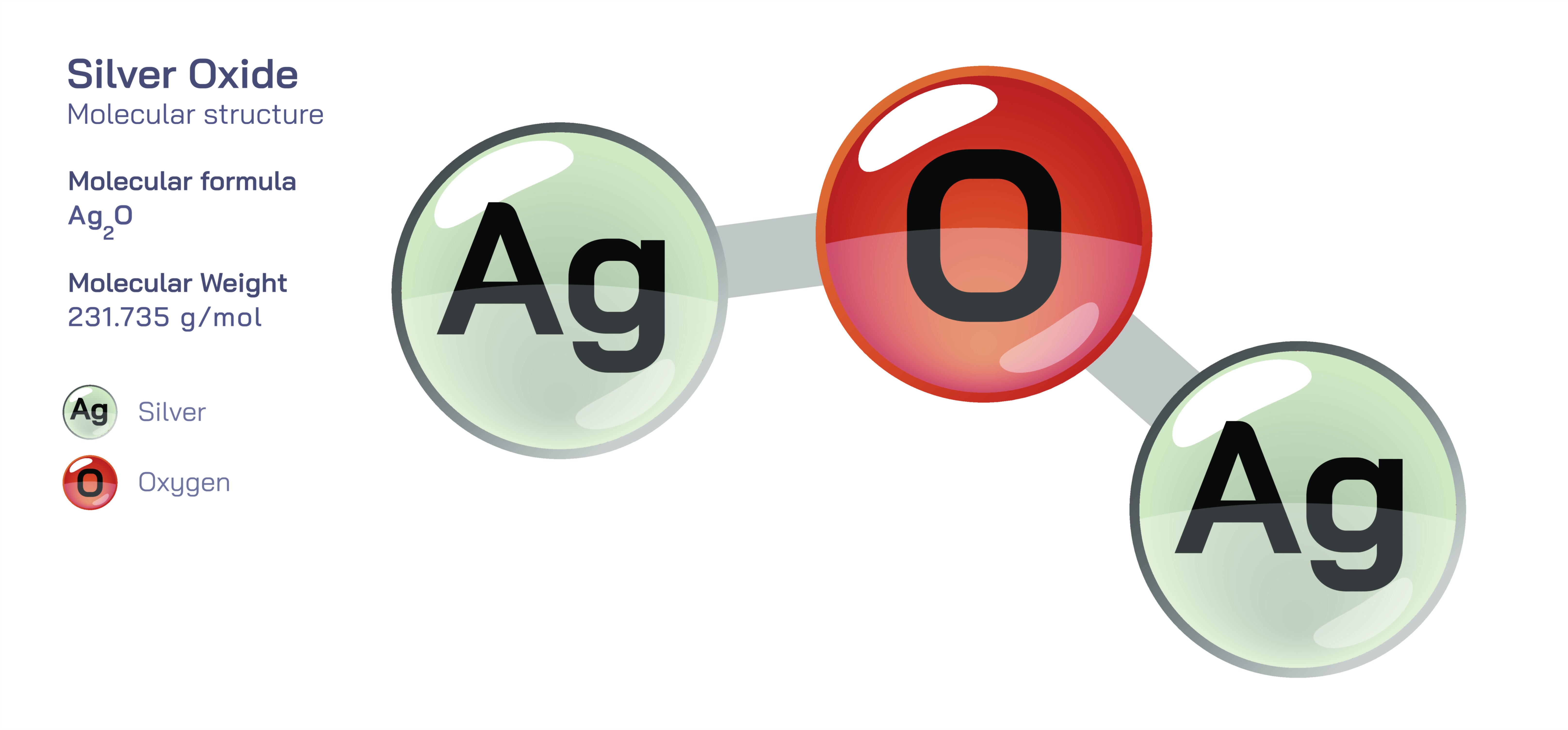
Silver oxide, typically represented by the chemical formula AgŌééO, is an inorganic compound composed of silver in its +1 oxidation state and oxygen in the form of oxide anions. Although it appears as a dark brown or almost black solid, the compoundŌĆÖs colour and reactivity reveal a complex relationship between electronic structure, ionic bonding and redox chemistry. Silver oxide is widely known in science and industry as a key oxidizing material, a component of high-energy batteries, a precursor to metallic silver, and a reagent in chemical synthesis. Its behaviour illustrates how even a compound composed of only two elementsŌĆösilver and oxygenŌĆöcan demonstrate broad applicability when the internal arrangement of atoms allows both structural stability and chemical responsiveness. To understand silver oxide deeply, one must begin with its lattice structure, which provides a foundation for all its physical, chemical and technological characteristics.
At the structural level, AgŌééO consists of AgŌü║ cations and O┬▓Ōü╗ anions arranged in a crystalline lattice. Unlike a covalent molecule that exists as a single discrete entity, silver oxide forms an ionic network throughout its solid phase. Each oxide ion in the lattice is coordinated by four silver ions, and each silver ion is bonded to two oxide ions, creating a repeating structure that maximizes electrostatic attraction. This arrangement gives the compound both stability in solid form and a distinct dark colour due to the way its electronic bands interact with visible light. Silver oxide is categorized as a basic oxide because the oxide ions possess a strong affinity for protons and react readily with acids and water. However, the behaviour of the compound cannot be understood simply by referring to the oxide ion; the electronic structure of the silver ion also plays an important role. Silver, with its filled d-electron configuration in the +1 oxidation state, creates a partially filled band pattern that enables unique transitions when exposed to light or chemical reducing agents. This is why silver oxide darkens rapidly upon decomposition into metallic silver and why it can serve as a bridge between ionic and metallic silver phases.
The reactivity of silver oxide with water provides one of its most fundamental transformations. When AgŌééO is introduced into water, rather than dissolving completely, it partially forms silver hydroxide, AgOH, a weak and unstable base. This product does not persist, however, and converts back into silver oxide and water unless excess hydroxide ions shift the equilibrium. The interaction between AgŌééO and water demonstrates how the compound maintains a balance between ionic dissolution and structural reassembly. In acidic solutions, the oxide ion binds readily to protons to form water, while the silver ions enter solution. This is why silver oxide dissolves in acids but not in pure water. In basic solutions, silver oxide reacts to form soluble silver complexes such as [Ag(OH)Ōéé]Ōü╗, explaining why strongly alkaline environments enhance its solubility. These acidŌĆōbase interactions highlight that the solid lattice can be broken under specific ionic conditions, showing once again that reactivity depends on the ability of the oxide ion to reorganize around hydrogen availability.
One of the most technologically significant applications of silver oxide lies in battery chemistry. Silver oxideŌĆōzinc batteries, commonly known as silver batteries, rely on the reduction of silver oxide to metallic silver during discharge. In these cells, AgŌééO acts as the cathode material, accepting electrons and transforming into silver metal while zinc at the anode undergoes oxidation to zinc oxide. This electron flow generates electrical energy. Batteries based on this system are valued because they deliver a stable voltage, high energy density and long shelf life compared to alkaline or leadŌĆōacid batteries. Their compact size and reliability make them ideal for precision electronics such as hearing aids, watches and military devices. The ability of AgŌééO to easily convert between ionic silver and metallic silver without structural breakdown makes the battery highly reversible in controlled environments, although commercial batteries are often single-use. This highlights a core principle: silver oxide functions not merely as an oxidant but as a material whose structure supports controlled electron transfer.
In chemical synthesis, silver oxide plays several important roles due to its gentle yet effective oxidizing power. It can convert aldehydes into carboxylic acids under controlled conditions and can promote selective transformations in organic reactions where stronger oxidizing agents would cause degradation. Silver oxide also acts as a reagent in halogen exchange and substitution reactions, particularly in contexts where silver ions can abstract halide ions and form highly insoluble silver halides. This selective formation of precipitates helps drive reactions forward. In addition, AgŌééO serves as a precursor in producing higher oxidation state silver compounds and silver salts. When heated or exposed to light or reducing agents, silver oxide decomposes into pure silver metal, making it an important material in metallurgical silver recovery. The conversion from oxide to metal demonstrates the relatively low energy barrier between the ionic and metallic phases of silver, which is one of the reasons silver chemistry is so rich.
Silver oxide also has antimicrobial significance due to the slow and controlled release of silver ions. Although AgŌééO is not highly soluble, the small concentration of AgŌü║ that enters aqueous environments displays strong antibacterial and antifungal properties by binding to proteins and nucleic acids in microbes, disrupting cellular function. This behaviour has been used in water treatment and some medical coatings, although usage must remain controlled because excessive silver ion levels can be toxic to humans and ecosystems. In material science, silver oxide nanoparticles have gained attention due to their enhanced catalytic and photocatalytic properties compared with bulk AgŌééO. At the nanoscale, large surface area and quantum confinement increase reactivity, making nanostructured silver oxide useful in hydrogen production, pollutant breakdown, electrode coatings and sensor materials.
Thermal decomposition of silver oxide reveals another layer of structural principles. When heated, the compound breaks down into metallic silver and oxygen gas without forming intermediate oxide species. This means the lattice simply reorganizes as silver ions gain electrons and oxygen ions release electrons, forming molecular oxygen. The simplicity and directness of this decomposition pathway have been important in the historical production of high-purity metallic silver. Because decomposition occurs at relatively moderate temperatures compared to many other metal oxides, AgŌééO can be a convenient source of silver metal in laboratory settings. Photochemical decomposition follows a similar path, except the energy input comes from light rather than heat. Ultraviolet light excites electrons, facilitating the reduction of AgŌü║ to AgŌü░, which causes the compound to darken as metallic silver particles form. This darkening phenomenon connects silver oxide with the broader history of light-sensitive silver chemistry, including photography.
Despite its valuable uses, silver oxide requires careful handling due to its oxidizing behaviour and the biological effects of silver ions. Although AgŌééO is not explosive or extremely aggressive as an oxidizer, it can intensify combustion when mixed with organic materials or reducing agents and heated. Environmentally, improper disposal of silver saltsŌĆöincluding silver oxideŌĆöcan release silver ions into water systems, where they pose risks to aquatic organisms. Therefore, industrial and laboratory waste must be treated to recover silver rather than discarded freely. Fortunately, silver recovery is economically favourable because metallic silver can be reclaimed from AgŌééO through thermal reduction, chemical reduction or electrochemical processes.
In summary, silver oxide is a powerful example of how the structure of an inorganic compound determines its function across chemistry, technology and biology. The ionic lattice of AgŌü║ and O┬▓Ōü╗ forms a solid that is stable yet ready to transform under the right conditions. Its ability to shift between oxide and metallic states allows it to function as a battery cathode, an oxidizing agent in synthesis, a precursor to silver salts and a disinfectant in controlled environments. Its photochemical and thermal decomposition teach core lessons about redox chemistry, while its solubility behaviour illustrates acidŌĆōbase equilibria and complex ion formation. Silver oxide demonstrates that even a seemingly simple compound hides structural sophistication that governs every aspect of its identity. It embodies the idea that chemistry is not only about composition, but also about the subtle interplay of charge, bonding, electron mobility and environmental context.