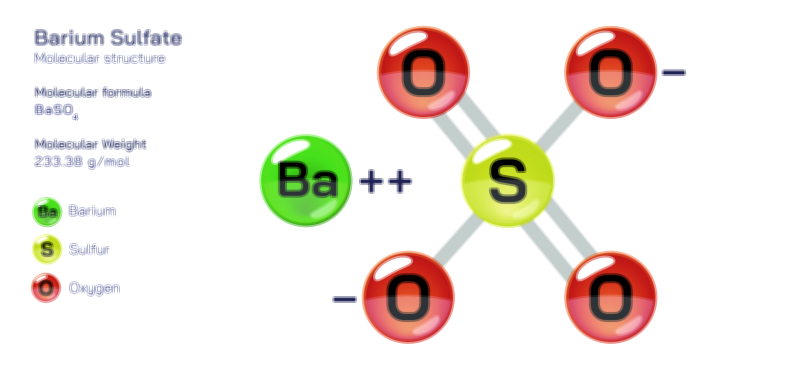
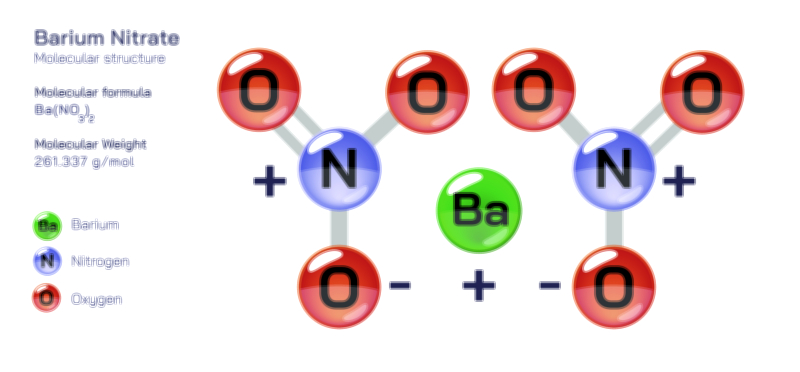
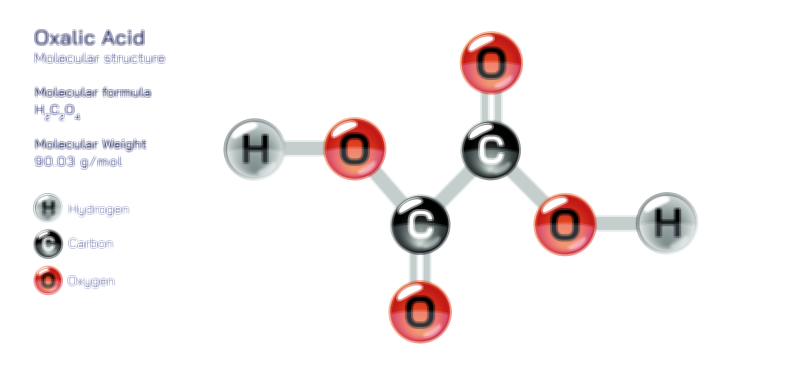
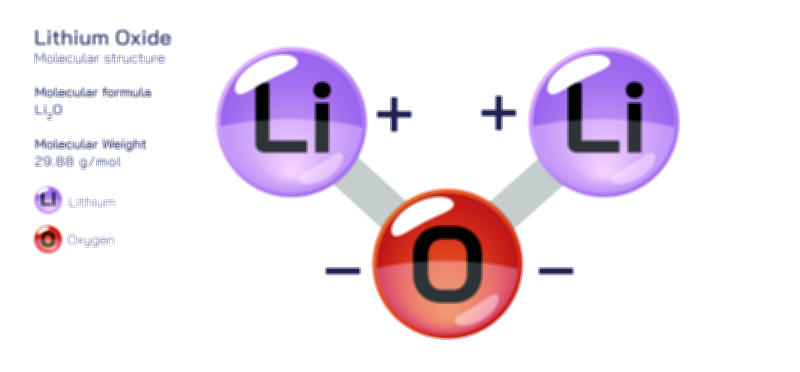
ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉōØÉĪØÉóØÉ©ØÉ£ØÉ▓ØÉÜØɦØÉÜØÉŁØÉ× ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×
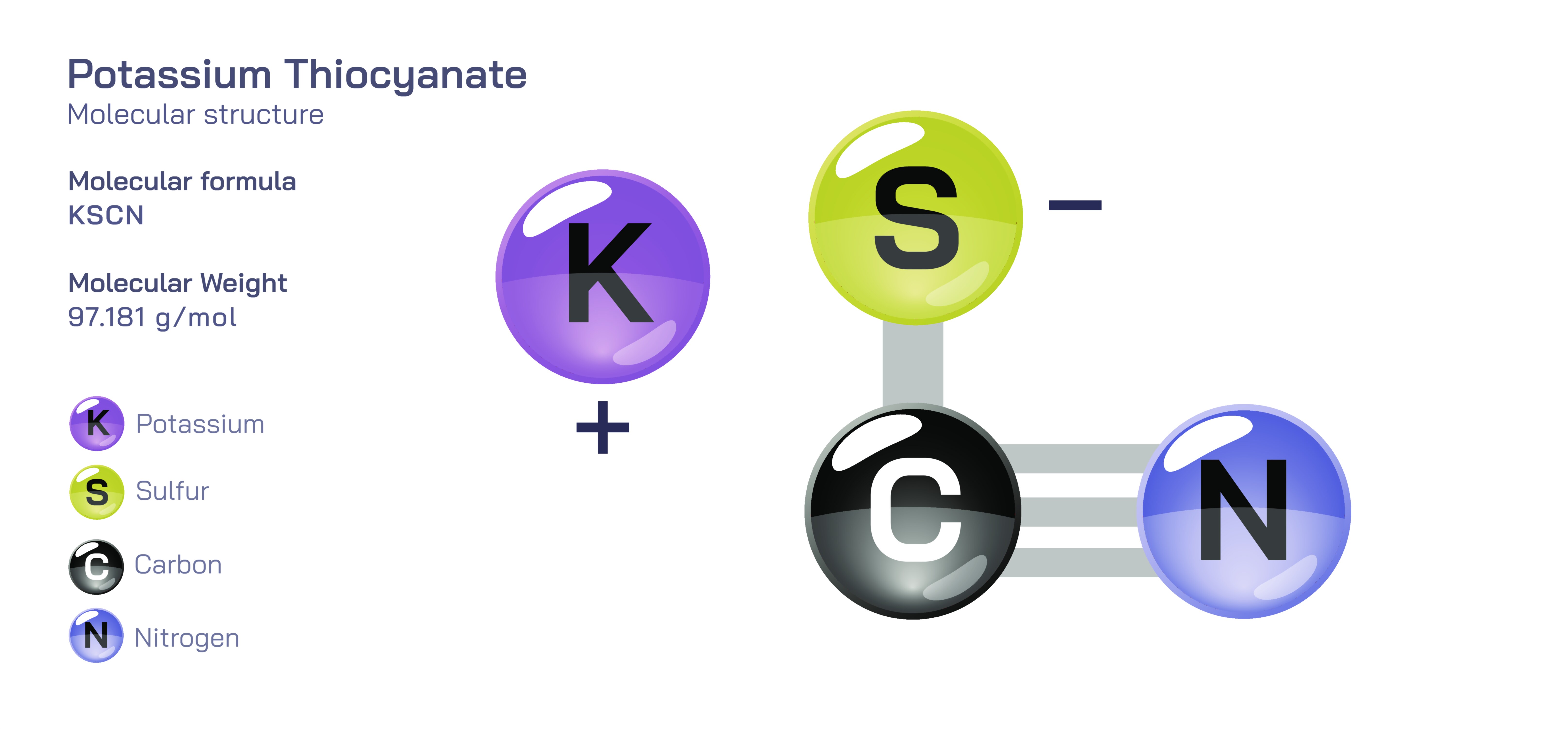
Potassium thiocyanate is an important inorganic compound consisting of potassium ions and thiocyanate ions, and its molecular structure explains the unique chemical behaviour, reactivity, and industrial usefulness that make it a valuable agent in analytical chemistry, coordination chemistry, biochemical research, and industrial synthesis. Although it appears externally as white, crystalline granules that dissolve easily in water and several polar solvents, its internal structure is built on a dynamic and highly interesting relationship between metal ions and an ambidentate ligand. The compound dissociates completely when dissolved, releasing potassium ions separately from thiocyanate ions, and this dissociation unlocks the behaviour of the thiocyanate group, which remains intact but changes the way it binds depending on the nature of its reaction partner. The simplicity of the empirical formula hides a sophisticated molecular identity, because the structure and bonding patterns of the SCNŌü╗ ion govern its biological, chemical, and analytical properties far more than the potassium ion itself. Understanding the molecular structure of potassium thiocyanate therefore provides scientific insight not only into how the compound behaves in solution but also into how the thiocyanate ligand participates in complex formation, sensing reactions, and chemical transformations.
The central feature of the compound is the thiocyanate ion, a linear group composed of sulfur, carbon, and nitrogen atoms arranged in a chain. The carbon atom is positioned between sulfur and nitrogen, and this arrangement is held together through alternating strong covalent bonding and electron sharing, giving the ion considerable internal stability. Despite being arranged in a straight line, the electron distribution across the sulfur and nitrogen ends is uneven, creating two distinct binding possibilities that make thiocyanate an ambidentate ligand. It can attach to metals either through sulfur or through nitrogen, and the preferred attachment depends on how the electronic environment of the metal interacts with the electron distribution in the thiocyanate ion. When a metal prefers binding with a softer, more polarizable atom, the sulfur end of the thiocyanate group becomes involved in coordination, forming thiocyanato complexes. However, when the metal has a stronger attraction to more electronegative atoms such as nitrogen, the thiocyanate group attaches through nitrogen instead, forming isothiocyanato complexes. This dual identity is the most characteristic and scientifically relevant property of the thiocyanate ion and explains why potassium thiocyanate is so widely used in complexometric analysis, ligand studies, material synthesis, and metal detection procedures.
The behaviour of potassium thiocyanate in aqueous solution demonstrates the interplay between ionic dissociation and ligand reactivity. Once dissolved, the potassium ion contributes mainly to ionic strength and conductivity, while the thiocyanate ion remains chemically active. In the absence of reactive species, the thiocyanate ion remains unchanged, maintaining its linear geometry that aligns sulfur, carbon, and nitrogen in direct sequence. However, the moment the solution encounters metal ions capable of forming coordination complexes, the thiocyanate ion begins acting as a ligand, and the way it attaches produces different colours, solubility patterns, and spectral behaviours depending on the metal centre. This makes potassium thiocyanate a classic reagent in inorganic qualitative analysis because the emergence of colour in solution provides rapid visual confirmation of the presence of certain metal ions. For example, ferric thiocyanate complexes are intensely red, and the formation of this colour has long been used in laboratories to detect or measure iron(III) ions. This transformation is not merely decorative but is rooted in a structural rearrangement that brings the nitrogen end of the thiocyanate ligand into contact with the ferric ion, creating a powerful chromophoric complex that absorbs light strongly and produces visible deep red coloration. This dramatic visual effect has been central to iron detection in water, soil, food, and environmental analysis, making potassium thiocyanate indispensable in analytical chemistry.
The structural adaptability of the thiocyanate group also makes potassium thiocyanate important in coordination chemistry research. Its ability to attach through either sulfur or nitrogen allows chemists to study the selectivity of metalŌĆōligand bonding, electronic configuration influences, and molecular geometry formation. The compoundŌĆÖs participation in bridging modes, where the thiocyanate ion connects two metal centres simultaneously, plays a significant role in the synthesis of coordination polymers, magnetic materials, and supramolecular structures. These extended architectures are vital to the design of new materials, and thiocyanate ligands contribute flexibility in both electronic and geometric effects. The existence of different coordination modes also contributes to the formation of complexes with distinct magnetic, catalytic, or spectroscopic behaviours, serving as building blocks for research on molecular switches, optical materials, and crystalline frameworks. Potassium thiocyanate therefore serves as a gateway to exploring principles of ligand field theory, metal complex structure, and molecular design.
In biological and biochemical environments, thiocyanate ions participate in subtle but meaningful interactions. Humans naturally generate small amounts of thiocyanate as a metabolite of cyanide detoxification, and the ion has been associated with physiological roles, particularly in the saliva and immune system. Potassium thiocyanate has been studied for its effects on haloperoxidase enzyme systems and mechanisms that influence antimicrobial activity. Although external supplementation must be handled with care due to potential interference with iodine uptake and thyroid function, the controlled and biologically relevant behaviour of thiocyanate underscores the importance of understanding its structural chemistry in biological contexts. The electron distribution within the SCN group plays a key role in determining how it interacts with proteins and enzymes, and these interactions are sensitive to the thiocyanate structure rather than the potassium ion.
In industrial chemistry, potassium thiocyanate supports organic transformations where thiocyanate groups act as building blocks for synthesizing pharmaceuticals, dyes, agrochemicals, and advanced intermediates. The molecular structure allows the ion to behave like a nucleophile under certain conditions and like an electrophile under others, enabling multiple reaction pathways. In surface science, potassium thiocyanate solutions can be used for etching, activation, or modification of metal surfaces because of the ligandŌĆÖs strong binding capability. In photography and textile dyeing, thiocyanate-based complexes play roles in colour enhancement, desilvering, and fixing processes. In polymer research, thiocyanate ions have been explored as structural modifiers for polymer electrolytes due to their ionic mobility and electronic effects.
While the structure of potassium thiocyanate enables wide scientific value, safety must be respected, as high concentrations can interfere biologically with the iodine cycle and thyroid activity. In industrial and chemical laboratory environments, the thiocyanate ion can release toxic gases if exposed to strong acids or rapidly oxidizing agents. These risks, however, do not diminish the compoundŌĆÖs relevance; rather, they emphasize the importance of responsible handling based on understanding the structure and reactive pathways.
The story of potassium thiocyanate demonstrates how the internal architecture of a molecule dictates its behaviour in countless chemical systems. The salt is held together ionically in solid form, yet internally the thiocyanate ligand is a tightly coordinated linear structure with two reactive binding ends. Dissolution does not break the SCN group apart but liberates it to behave independently as a ligand, chromophore, nucleophile, or structural unit depending on its environment. Its ambidentate nature creates the foundation for complex formation, its stability ensures survival under a wide range of chemical conditions, and its electron distribution allows reactivity that is both selective and versatile. This rich structural identity makes potassium thiocyanate a compound with scientific depth far beyond its simple appearance, proving again that in chemistry, the smallest changes in molecular arrangement can shape powerful transformations across analytical science, materials engineering, industrial synthesis, and biological chemistry.