ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉÆØÉ«ØÉźØɤØÉóØÉŁØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉäØÉ▒ØÉ®ØÉźØÉÜØÉóØɦØÉ×d.
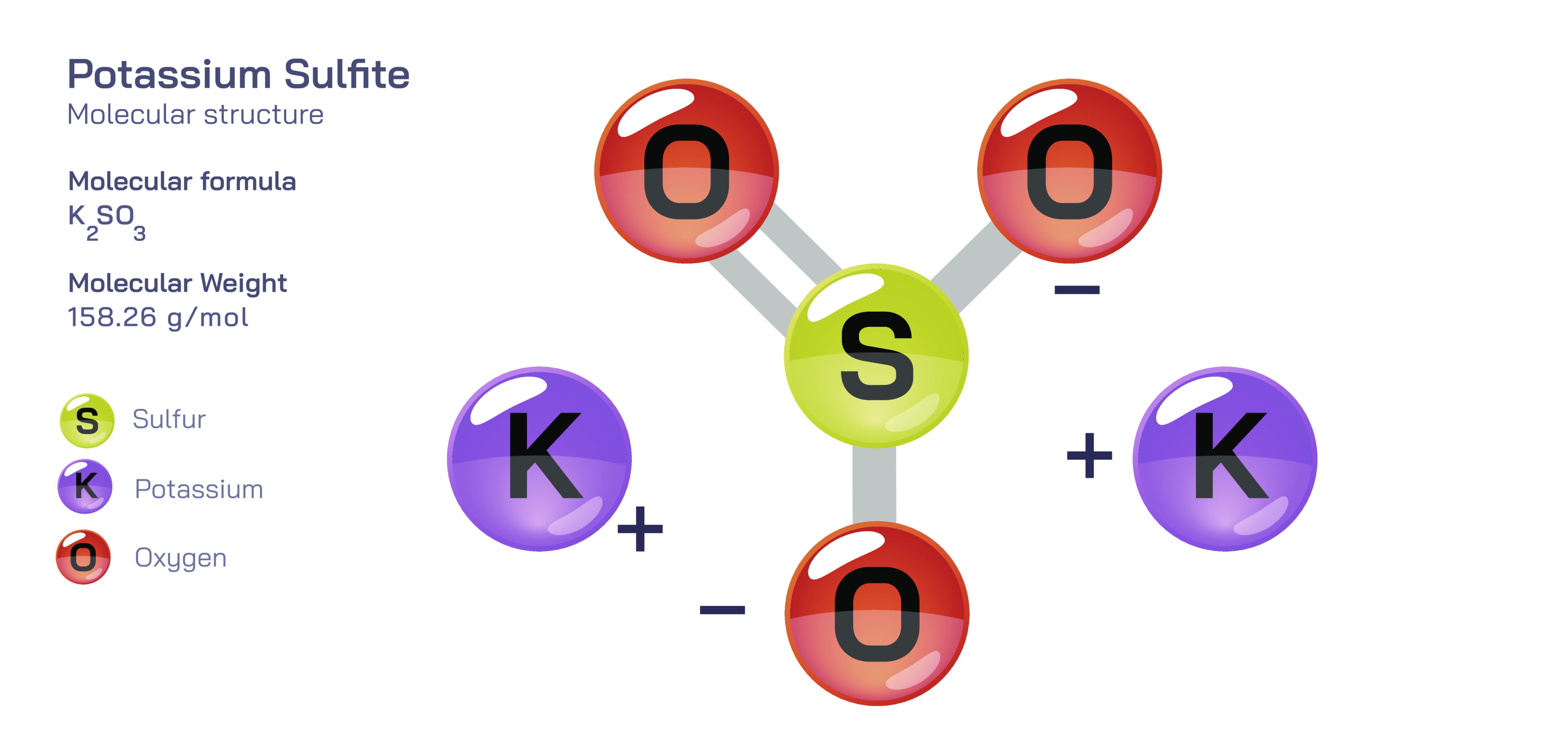
Potassium sulfite is an inorganic chemical compound recognized for its preservative, antioxidant and reducing properties, and its identity arises from the structural interaction between potassium ions and sulfite ions. Although its molecular formula KŌééSOŌéā appears simple, the behaviour of the compound is governed by the internal architecture of the sulfite ion and the electrostatic balance provided by potassium cations. In its solid crystalline form, potassium sulfite displays a salt-like appearance, signifying a stable ionic lattice in which each sulfite ion is surrounded by potassium ions in a repetitive arrangement that extends in three dimensions. The lattice exists because both potassium ions bear positive charges while the sulfite ion bears a double negative charge, and in order to maintain electrical neutrality and minimize repulsion, two potassium ions must associate with each sulfite unit. The potassium ions are nearly spherical and chemically calm, but the sulfite ion expresses the meaningful molecular behaviour. The sulfite structure contains one central sulfur atom bonded to three oxygen atoms in a trigonal pyramidal shape, with one lone electron pair on the sulfur that alters the geometry and produces a strong uneven distribution of negative charge. This lone pair, along with resonance among the sulfurŌĆōoxygen bonds, gives sulfite the ability to donate electrons, accept protons and bind temporarily with reactive substances, demonstrating that much of the compoundŌĆÖs reactivity arises from the chemical personality of the sulfite ion rather than the potassium ion.
When potassium sulfite dissolves in water, its solid ionic lattice breaks apart easily because the ions interact strongly with water molecules. The result of this dissolution is not a neutral molecule but fully separated potassium and sulfite ions. The potassium ion becomes solvated and simply stabilizes the electrical balance of the solution, while the sulfite ion retains its pyramidal geometry and becomes chemically active. In water, the sulfite ion behaves as a weak base because it can accept protons to form bisulfite ions; meanwhile, its spare electron pair and resonance-stabilized system make it behave as a reducing agent by donating electrons in redox reactions. This duality explains why potassium sulfite is both a preservative and an antioxidant: it prevents oxidation by lowering the oxidative stress within a system. When exposed to oxidative conditions or strong oxidizing substances, sulfite ions neutralize them by being oxidized themselves, converting gradually into sulfate. What makes this transformation scientifically meaningful is that sulfite acts as a redox buffer. This structure-driven ability to remove oxygen-based radicals or reactive oxygen species slows spoilage, discoloration and chemical breakdown in food and beverages. It does not sterilize the system but alters the chemical environment to discourage the reactions that lead to deterioration, proving that the potency of potassium sulfite lies in its electronic structure rather than in toxicity.
The industrial uses of potassium sulfite are primarily anchored in this antioxidant behaviour. In the food and wine industries, potassium sulfite prevents spoilage, colour loss and microbial degradation by reducing oxidative stress. Wine contains alcohol, sugars and organic acids that are sensitive to oxygen. During wine production and storage, even a small amount of oxidative reaction can change flavour and aroma compounds. Potassium sulfite counteracts this by interacting with dissolved oxygen and by converting unstable aldehydes into more stable compounds, protecting both the chemical structure and sensory quality of the beverage. The reducing action of sulfite prevents browning in juices, dried fruits and beverage concentrates. This browning is caused by enzymes that generate oxidative pigment molecules called melanins, and potassium sulfite interrupts this process by modifying intermediate compounds before they develop colour. The compound does not eliminate microbial growth at high levels but reduces the conditions that support spoilage, allowing other preservation systems to work more efficiently.
Chemical and industrial processing applications of potassium sulfite expand from the same structural principles. Because sulfite ions can trap oxygen and break down peroxides, the compound is useful in pulp and paper production, photography and boiler water treatment. In paper manufacturing, sulfite ions help soften wood chips and prevent oxidative degradation during pulping, maintaining cellulose strength and colour stability. In photography, sulfite solutions protect developing agents from oxidation, ensuring uniform image formation. In boiler systems, sulfite removes dissolved oxygen that would otherwise corrode steel surfaces. The basic principle across all these industries is identical: sulfite ions donate electrons to consuming oxygen or oxygen-derived species, stabilizing the system and protecting material integrity. These effects showcase structureŌĆōfunction alignment, where the lone electron pair on the sulfur atom allows sulfite to participate in reduction reactions without causing undesired side reactions under controlled conditions.
Potassium sulfite also appears in laboratory chemistry where its controlled reducing ability makes it a valuable reagent. It participates in redox reactions where gentle reduction is required rather than harsh reducing power. Because it can slowly oxidize to sulfate without inducing side reactions, it is preferred in situations where strong reductants would destroy sensitive compounds. This balance separates potassium sulfite from more aggressive reducing agents such as borohydrides, allowing specialists to reduce intermediates without damaging end products. The sulfite ion in solution can also act as a nucleophile, though this behaviour is limited compared to its reducing ability. It can add to electrophilic carbon centres under appropriate conditions, forming addition products that may undergo further transformations. These laboratory behaviors are not coincidental but arise from the localized negative charge distribution across the oxygen atoms of the sulfite ion, which makes attack on positively charged reaction centres possible under carefully selected environments.
The behaviour of potassium sulfite is also sensitive to pH because the sulfite ion exists in equilibrium with bisulfite and sulfurous acid depending on proton concentration. At lower pH, sulfite accepts hydrogen ions, forming bisulfite, which can then eventually convert into sulfurous acid. If acid concentration increases strongly or the temperature rises, the structure of sulfurous acid collapses and releases sulfur dioxide gas. This pH-dependent conversion demonstrates that the sulfite ion is not just a static species but a structural participant in a dynamic equilibrium involving protonation and gas evolution. This behaviour is responsible for both the utility and the hazards associated with the compound. In controlled food and wine production, limited release of sulfur dioxide from sulfites suppresses microbial activity and oxidation. However, in uncontrolled acidic environments, excessive release of sulfur dioxide can produce respiratory irritation. This means that safety in handling potassium sulfite depends on recognizing the structural pathways that lead to gas release and managing them appropriately.
In terms of safety, potassium sulfite is typically safe at regulated concentrations in food and beverages, but some individuals may show sensitivity to sulfites, especially asthmatic or allergic individuals. This sensitivity arises because sulfite can release sulfur dioxide within the respiratory system of susceptible individuals, causing inflammation or bronchoconstriction. In industrial and laboratory settings, handling requires awareness of the compoundŌĆÖs reactivity with acids and oxidizers. Contact with strong acids can rapidly generate sulfur dioxide gas, which is harmful when inhaled, and contact with powerful oxidizing agents can produce large quantities of heat during redox reactions. Potassium sulfite solutions at high concentrations can irritate the skin and eyes because the reducing and nucleophilic behaviour of sulfite can disrupt organic tissues upon prolonged contact. These risks do not undermine the compoundŌĆÖs usefulness but highlight that every reactive compound must be managed according to the structural transformations it can undergo.
Taken holistically, potassium sulfite is not just a stabilizing chemical or preservative but a prime example of how the structure of an ion determines wide-ranging chemical, industrial and biological impacts. The potassium ion maintains neutrality and solubility, allowing the sulfite ion to function freely in solution. The trigonal pyramidal geometry of sulfite and its resonance electrons equip the compound with controlled reducing power and antioxidant capacity. The tendency of sulfite to convert to sulfate under oxidative conditions explains its preservative properties. The equilibrium between sulfite, bisulfite and sulfurous acid explains both its beneficial regulation in food systems and its risks in acidic environments. Potassium sulfite embodies the principle that chemistry is not defined merely by formulas but by the interplay of structure, reactivity, electron distribution and environmental context. Through this lens, its role across winemaking, photography, food preservation, water treatment, material processing and laboratory science becomes a coherent story of structure enabling functionŌĆöhighlighting the way one ionic compound can shape multiple industrial and biological worlds when the architecture of atoms aligns with purposeful application.