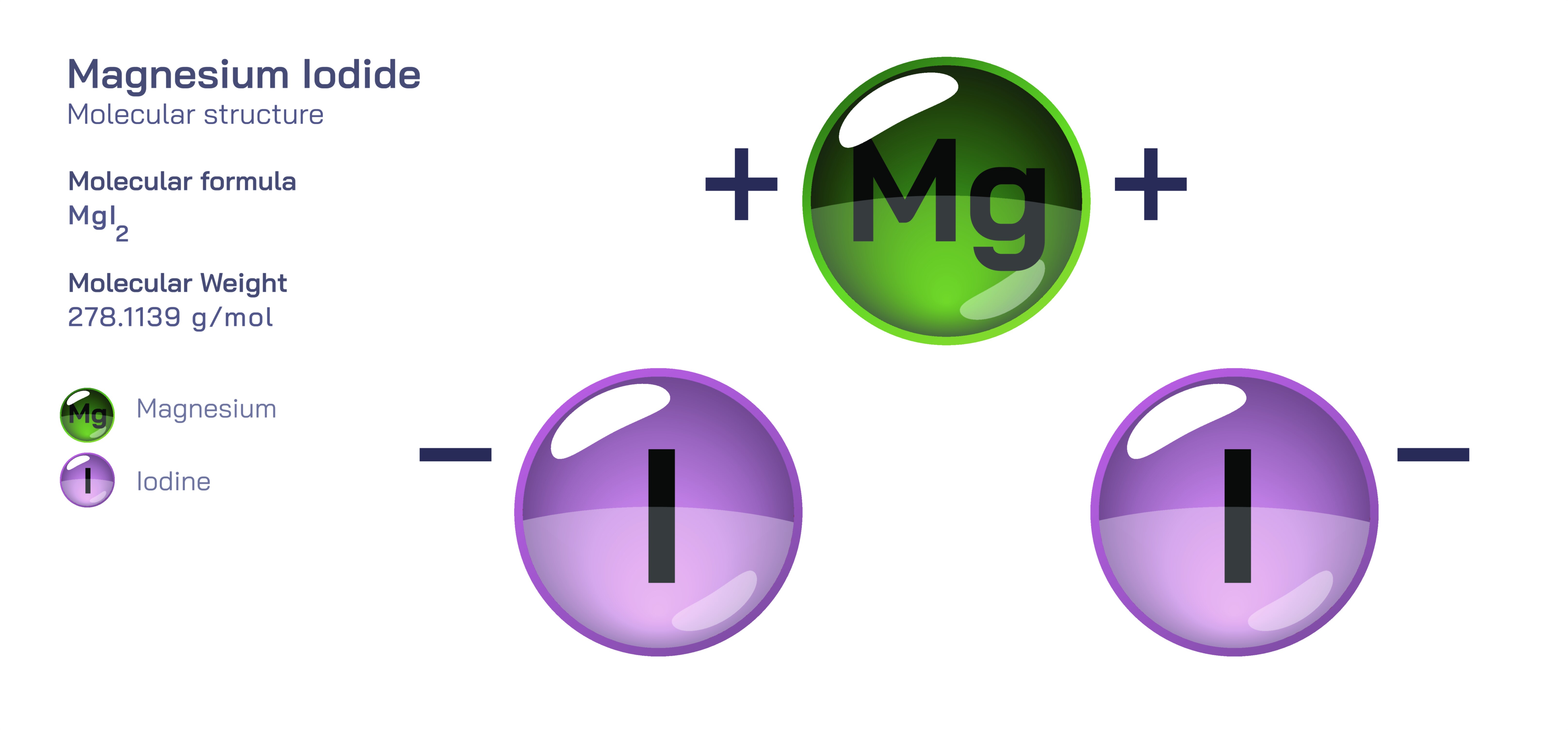Magnesium Iodide — Molecular Structure and Composition
Magnesium iodide is an inorganic chemical compound best represented by the formula MgI₂, and it stands out as a fascinating material because of the way its molecular structure, ionic arrangement, and compositional characteristics lead to diverse applications across chemistry, biology, materials science, and industrial processing. Within this compound, a magnesium atom carries a divalent positive charge and forms ionic bonds with two iodide ions, each with a monovalent negative charge, creating a stable unit that reflects electrostatic attraction between highly contrasting elements. Magnesium, a lightweight alkaline earth metal with strong electron-donating behavior, interacts with iodine, a large halogen atom known for its strong electron-accepting capability. This precise balance between magnesium’s tendency to lose electrons and iodine’s tendency to gain them results in the formation of a crystal lattice that is cohesive, dense, and electrically neutral. In solid form, magnesium iodide arranges itself in an organized pattern of repeating ionic units, but in aqueous solutions, the compound dissociates into free magnesium and iodide ions, a property that determines much of its reactivity, solubility, and biological significance. The compound also exists in both anhydrous and hydrated states, with the hydrated variant containing water molecules embedded within its structure, and this difference in water content affects physical behavior, stability, and suitability for various laboratory and manufacturing environments.
In biochemical contexts, magnesium iodide serves as a valuable source of both magnesium and iodine, two elements that are vital for living systems but function very differently within the body. Magnesium plays a fundamental role in energy production, neuromuscular signaling, enzyme function, and metabolic regulation, making it indispensable for cellular physiology. Iodine, on the other hand, is essential for the synthesis of thyroid hormones, which regulate growth, metabolism, and thermoregulation. When magnesium iodide dissolves in water, the release of these ions allows them to enter metabolic pathways that contribute to physiological balance; however, direct therapeutic use is approached cautiously because improper dosing of iodine can disrupt thyroid function. For this reason, magnesium iodide is not commonly used in general dietary supplementation even though, in theory, it can contribute valuable nutrients. Instead, it is primarily used in controlled laboratory or medical research settings where the concentration and ionic environment can be precisely monitored, particularly in studies exploring electrolyte behavior, halogen-metal interactions, and signaling pathways that depend on divalent cations.
From a chemical perspective, magnesium iodide is an excellent reagent for introducing iodide ions into organic and inorganic synthesis. Its strong dissociation in solution allows it to participate in nucleophilic substitution reactions, halogen exchange processes, pharmaceutical development, and formation of organometallic intermediates. In moisture-sensitive synthesis, the anhydrous form is preferred because water content can lead to undesirable side reactions or product degradation. The compound also plays a role in the preparation of alkyl iodides, which are highly useful intermediates in organic synthesis because iodide is a superior leaving group relative to chloride or bromide. This property makes magnesium iodide important for industries requiring efficient synthetic routes with fewer reaction steps and higher yields. In coordination chemistry, magnesium iodide contributes to the formation of metal complexes where ligand behavior, electronic distribution, and steric effects can be studied through controlled interaction with the magnesium center, advancing research into molecular bonding, catalysis, and material design.
Magnesium iodide also has relevance in electronics and optical science because of its hygroscopic nature and halogen composition. The compound readily absorbs moisture from the air and forms stable hydrates, which can be both beneficial and challenging. Its moisture-binding capacity allows controlled water removal in certain industrial and laboratory environments, but excessive water uptake can compromise purity and function in sensitive electronic or high-precision settings. In optical material development, halide compounds containing iodine are often studied for their ability to influence refractive properties, phonon behavior, and radiation response in crystal matrices. Magnesium iodide participates in ongoing research exploring halide materials for infrared optical systems, light-emitting components, and advanced sensors, though such studies remain specialized and typically require ultra-dry and meticulously managed conditions due to the compound’s sensitivity to air and humidity.
The thermal behavior of magnesium iodide further distinguishes it among halide salts. When subjected to heat under controlled conditions, it can decompose gradually into magnesium oxide and iodine vapor, and this reversible transformation has been studied for potential use in iodine recovery and closed-loop halogen-metal systems. Decomposition properties also make magnesium iodide useful in academic settings where thermodynamic cycles, lattice energy effects, and halogen-release kinetics are investigated. Because the iodide ion has a large electron cloud and occupies substantial molecular volume compared to other halogens, the melting point, solubility, and vapor characteristics of magnesium iodide differ significantly from those of magnesium chloride or magnesium bromide. These distinctions help researchers evaluate trends across the halogen group and map relationships between ionic radius, electronegativity, bond strength, and lattice configuration.
In manufacturing and specialized industries, magnesium iodide contributes to niche yet meaningful processes. It plays a role in catalysts for organic transformations, stabilizers for photographic materials in certain legacy imaging systems, and formulations for protective coatings where its halogen composition assists in controlled surface modification. Some research has explored magnesium iodide as a component in battery systems, using its ionic mobility and electronic properties to influence charge behavior in solid-state and hybrid electrolyte designs. While such applications are not widely commercialized, they represent a direction in which continued investigation may lead to next-generation technologies that benefit from halide-based ionic conduction.
Although magnesium iodide offers valuable chemical and material advantages, it must be handled with care, as iodine compounds can be reactive, sensitive to humidity, and capable of influencing biological systems at very small concentrations. Storage typically requires sealed containers, dry environments, and protection from light to minimize breakdown or water absorption. When used in scientific environments, magnesium iodide is appreciated for its predictability and purity, but these advantages depend on meticulous handling procedures that preserve its intended composition. The compound’s interaction with moisture is particularly significant because the transition from anhydrous to hydrated form alters weight, reactivity, and concentration, which can interfere with analytical accuracy if not accounted for.
Overall, magnesium iodide stands as a compelling example of how the structural characteristics of a molecular compound can influence its chemical behavior, compositional value, and real-world functionality. Its molecular architecture, based on the robust charge balance between magnesium and iodide ions, underpins a pattern of properties that bridge reversible ionic dissociation, strong halogen-metal interaction, and broad chemical utility. Whether supplying ions for metabolic studies, participating in synthetic chemistry as a halogen source, contributing to investigations in optical or electronic materials, or supporting the development of high-precision industrial processes, magnesium iodide illustrates the wide-ranging significance that can emerge from the interplay between atomic structure and molecular composition. As research continues to expand across fields that rely on halogen chemistry and sustainable material innovation, magnesium iodide will likely maintain and increase its importance as a versatile compound with deep scientific relevance and evolving technological potential.