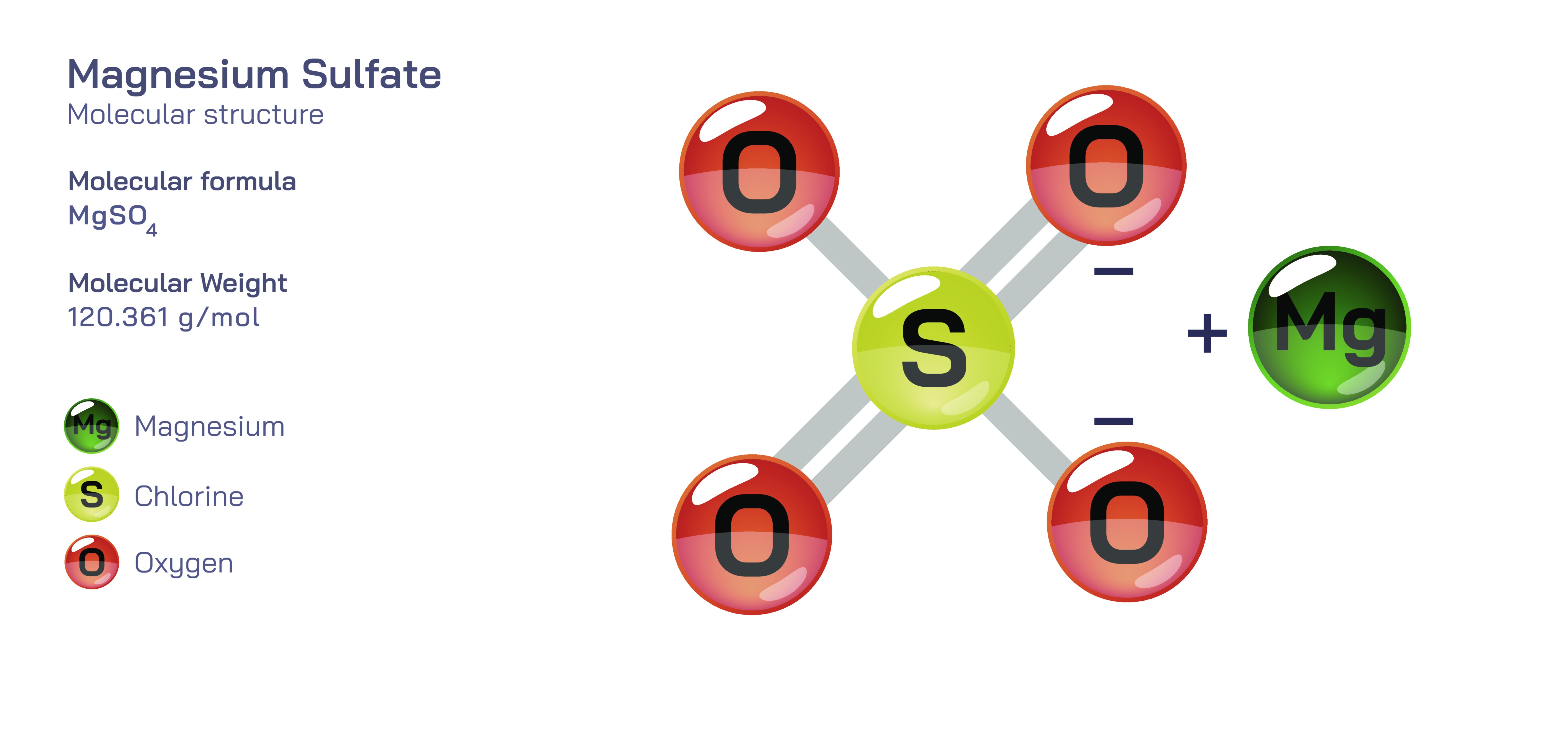
Magnesium Sulfate — Molecular Structure and Composition
Magnesium sulfate is an inorganic compound represented by the formula MgSO₄, and it occupies a remarkable position in both natural and applied science because of the way its molecular structure supports biological life, industrial technologies, environmental management, and medical treatment. At the core of the compound is a magnesium ion carrying a double positive charge that is electrostatically bonded to the sulfate group, a tetrahedral molecular unit in which a central sulfur atom is surrounded by four oxygen atoms. This sulfate structure distributes electron density evenly across its oxygen atoms through resonance, creating a highly stable anionic group that binds predictably to magnesium. The resulting ionic association forms crystalline units strong enough to maintain structural integrity in the solid state while being highly soluble in water, where the magnesium and sulfate ions dissociate completely and take part in independent chemical and physiological processes. Magnesium sulfate exists in multiple hydration states, with the most familiar form being magnesium sulfate heptahydrate, commonly known as Epsom salt, where seven water molecules integrate into the crystal lattice. The degree of hydration directly influences appearance, stability, melting behavior, solubility, and handling characteristics, which is why different versions of magnesium sulfate appear in agriculture, medicine, chemistry laboratories, and consumer products.
Inside living organisms, magnesium sulfate plays a direct and fundamental role because both magnesium and sulfate ions are essential to biological pathways that support life. Magnesium is a critical mineral required for enzyme activation, neuromuscular coordination, cardiovascular function, glucose metabolism, and the structural stability of DNA and ribosomes. Without magnesium, the biochemical machinery behind energy conversion, digestion, cell signaling, and nervous function cannot operate properly. The sulfate component participates in processes related to protein synthesis, detoxification, and structural integrity through sulfur-containing amino acids and glycosaminoglycans in connective tissues. When magnesium sulfate dissolves in water, it makes these ions available for absorption, allowing them to take part in the maintenance of electrolyte balance and metabolic homeostasis. In therapeutic contexts, magnesium sulfate administered orally or intravenously is used to relieve magnesium deficiency, normalize nerve and muscle functioning, regulate cardiac rhythm, and reduce neuromuscular excitability. In obstetric medicine, magnesium sulfate is a key intervention used to prevent seizures associated with preeclampsia and eclampsia because of its effect on neuromuscular transmission and vascular tone. Its ability to calm overactive neural conduction while improving tissue oxygenation has made it a life-saving mineral in emergency maternal care settings around the world.
Magnesium sulfate also has broad significance in pain management, physical recovery, and musculoskeletal health. When dissolved in warm water, as in a therapeutic bath, the magnesium ion from the dissociated solution can be absorbed through the skin and exert a relaxing effect on muscles, helping reduce soreness, inflammation, and tension caused by stress or physical exertion. The sulfate ion contributes to detoxification pathways that assist the body in breaking down metabolic waste products that accumulate within muscle tissue during fatigue. As a result, Epsom salt baths have been widely used in sports therapy, physiotherapy, and home wellness routines to reduce swelling, ease cramps, enhance circulation, and promote an overall sense of physical renewal after injury or exertion. The compound’s soothing properties extend beyond muscle relaxation to support sleep quality and the restoration of parasympathetic nervous system balance, especially for individuals who experience stress-induced tension.
In agriculture and horticulture, magnesium sulfate serves as an important fertilizer component because it supplies magnesium and sulfur — two nutrients that plants require for robust growth. Magnesium is an essential element in chlorophyll, the molecule responsible for capturing light energy during photosynthesis, and it also enables the movement of carbohydrates from leaves to developing tissues. Sulfur contributes to the formation of amino acids, enzymes, vitamins, and natural defense compounds that protect plants against stress. Because magnesium sulfate dissolves readily in water without changing soil pH, it provides immediate nutrient availability, making it particularly useful for correcting magnesium deficiencies that result in leaf yellowing, poor fruit formation, and reduced productivity. Crops grown in sandy soils, greenhouse operations, hydroponic setups, and magnesium-sensitive species such as tomatoes, peppers, roses, and citrus trees respond strongly to magnesium sulfate supplementation due to its rapid nutrient delivery and compatibility with other fertilizers. In large-scale farming, it helps balance soil nutrition profiles without introducing excess chloride or carbonate ions that might disturb delicate agricultural ecosystems.
Industrial applications of magnesium sulfate reflect its versatility as a chemical with both reactive and stabilizing functions. In dyeing and textile finishing, it serves as a conditioning agent that promotes even color absorption and improves binding between dyes and fabric fibers. In the production of paper and pulp, it contributes to improving fiber quality, minimizing bleaching damage, and enhancing brightness through controlled chemical interactions. In manufacturing processes involving cement, ceramics, and specialty coatings, magnesium sulfate can modify setting behavior, crystal formation, and moisture resilience, allowing specialized construction materials to perform under stress or exposure. It also finds a place in fireproofing, tanning, and water-treatment formulations, where its sulfate chemistry supports complex precipitation, pH regulation, and mineral balance requirements necessary for high-efficiency industrial operation.
Consumer and everyday-use products benefit from magnesium sulfate for its benign chemical profile and gentle performance characteristics. It is present in bath salts, mineral foot soaks, exfoliating scrubs, deodorants, and some skincare formulations because it absorbs excess moisture, mitigates irritation, and balances skin surface chemistry without blocking pores or causing harsh reactions. In home gardening, Epsom salt is used as a supplemental plant nutrient, especially by growers who cultivate magnesium-hungry crops and want to improve blossom production, fruiting outcomes, and foliage color without altering soil chemistry too aggressively. Food-grade magnesium sulfate also functions as a firming agent, stabilizer, and processing aid in select food manufacturing environments due to its safety and neutral flavor profile.
Although magnesium sulfate is considered safe and widely beneficial, proper handling is important to maintain its intended effects. Overconsumption — either orally or intravenously — can lead to electrolyte imbalances, particularly in individuals with impaired kidney function, because magnesium is cleared from the body through renal pathways. In agricultural contexts, adding more magnesium sulfate than needed does not improve plant growth and may disrupt soil nutrient ratios if used excessively. In industrial settings, storage conditions must prevent unnecessary moisture exposure, because hydration states influence weight, chemical strength, and dissolving behavior.
In every domain where it is applied, magnesium sulfate demonstrates how a compound’s usefulness is rooted in the harmony between its molecular structure and its functional behavior. The ionic partnership between magnesium and the sulfate group creates a mineral that is simultaneously soluble, bioavailable, physiologically compatible, and chemically stable. Whether it is assisting a muscle to relax after strenuous activity, saving a mother’s life during childbirth emergencies, nourishing a crop in depleted soil, stabilizing a chemical process inside a factory, improving the texture of a consumer product, or replenishing a biological system in need of essential minerals, magnesium sulfate reflects the profound power of mineral chemistry in shaping human health, environmental sustainability, and technological advancement.