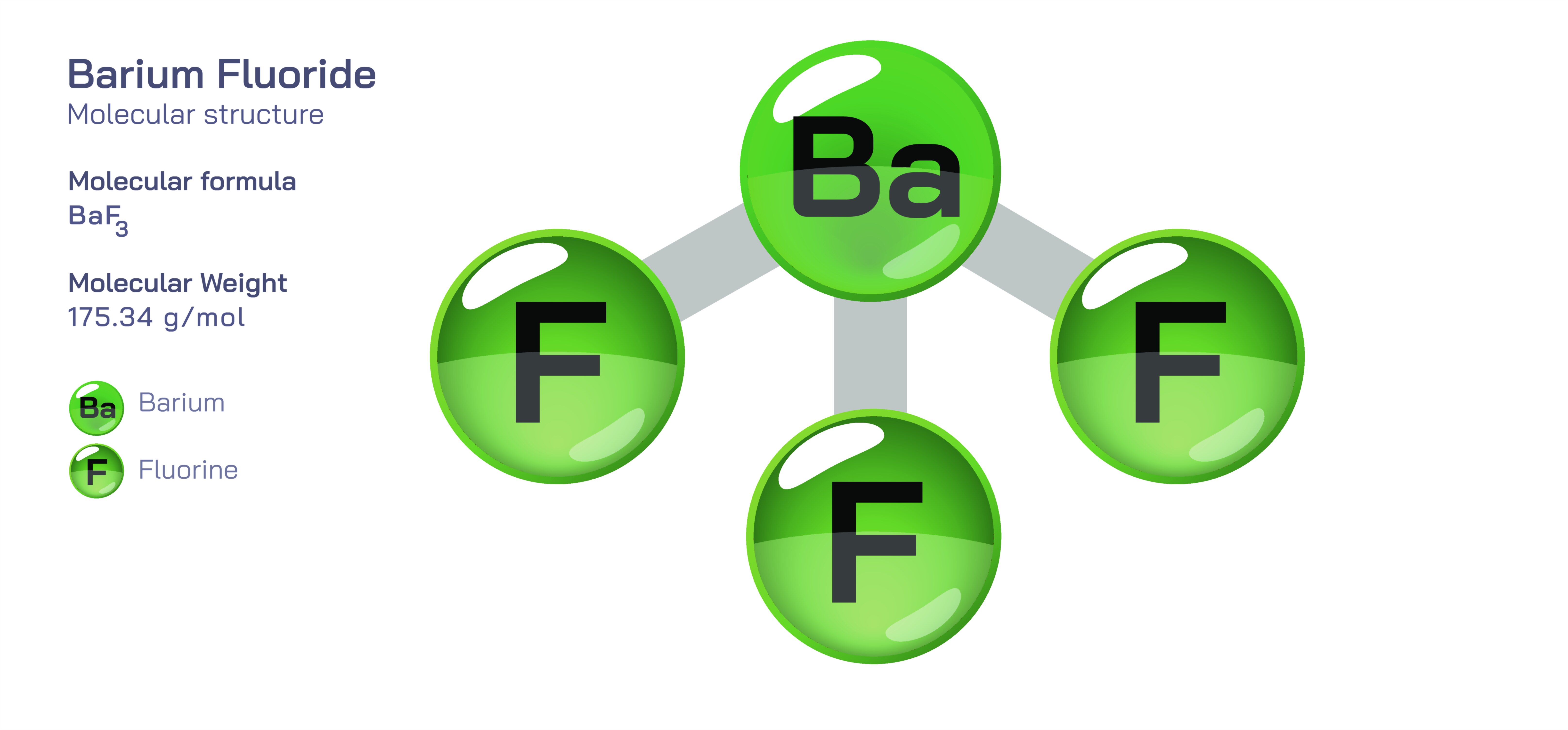
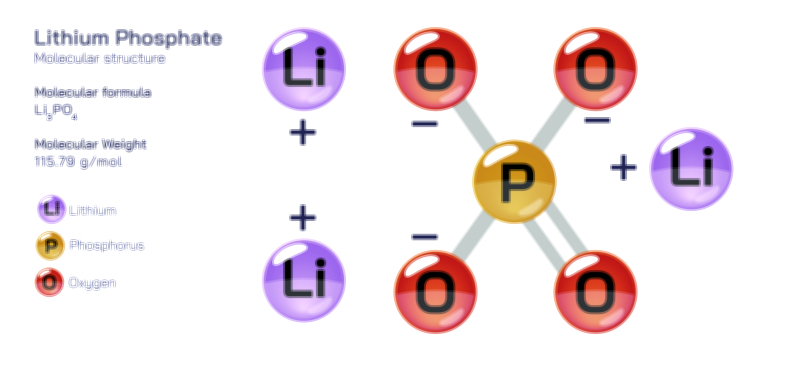
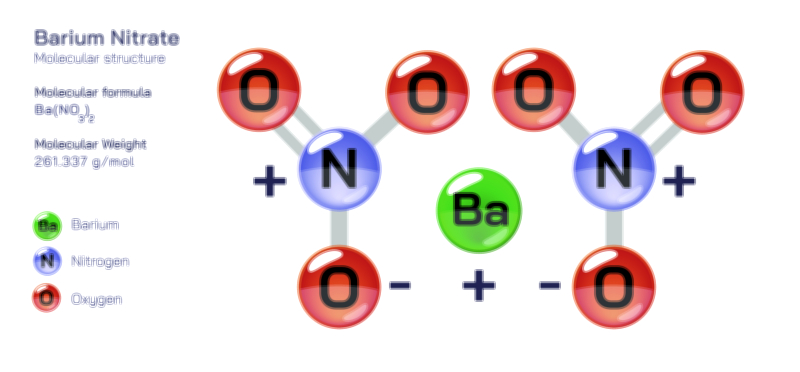
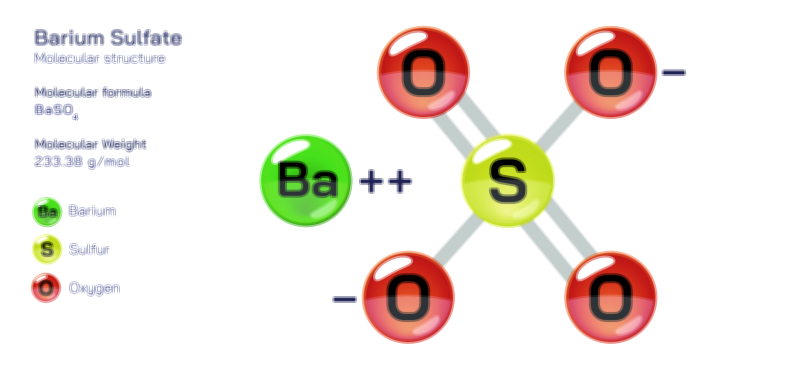
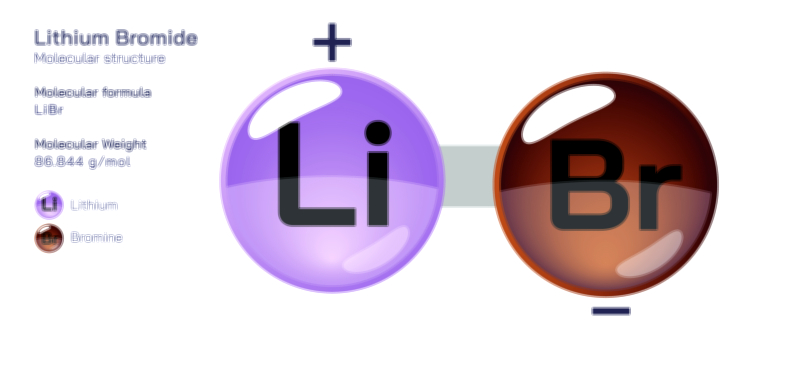
Barium Fluoride – Chemical Compound Structure, Properties, and Applications
Barium fluoride is a notable inorganic compound that holds a unique place in both the world of chemistry and the field of advanced industrial materials due to its combination of ionic bonding, optical capabilities, thermal stability, and versatile crystalline behavior. Represented chemically as BaF₂, it is formed from the union of barium cations and fluoride anions, creating a crystalline solid that is colorless, firm, and highly stable. In its structural essence, the compound reflects a strong ionic lattice, and this rigid geometric arrangement is responsible for many of its material characteristics, including high melting temperature, resistance to chemical reactivity in many environmental conditions, and the remarkable optical transparency that makes it suitable for both scientific and technological applications. When imagined as a vector illustration, the molecular depiction of BaF₂ is usually characterized by a large central barium atom coordinated symmetrically with surrounding fluoride ions to reflect the precise geometric balance of ionic charge. Such illustrations are used not only to communicate the atomic arrangement but also to bridge the gap between scientific theory and visual comprehension, allowing researchers, educators, and students to grasp the structural nature of the compound at a glance.
One of the most important characteristics of barium fluoride lies in its optical behavior. Unlike many inorganic salts that immediately lose transparency under ultraviolet radiation exposure, BaF₂ demonstrates outstanding transmission capabilities over a remarkably wide spectral range. It allows the passage of electromagnetic radiation from the deep ultraviolet region into the infrared spectrum, giving it a technological advantage in fields where components must remain transparent to different wavelengths. This property allows it to be used in lens systems, optical windows, and spectroscopic instruments that require durable materials that will not darken or degrade in demanding radiation conditions. The lattice structure that enables such optical clarity is also what gives barium fluoride the mechanical toughness required to withstand harsh operating environments without rapid wear, warping, or chemical breakdown.
Another dimension of the compound’s industrial significance lies in its thermal properties. BaF₂ has a high melting point and retains its structural strength and optical clarity under considerable temperature changes. This makes it ideal for applications in thermal imaging systems, aerospace optical modules, and scientific instrumentation that must operate within fluctuating high-temperature environments. Instead of distorting or suffering from thermal scattering effects, barium fluoride continues performing faithfully and consistently. Industrial processing of the compound takes advantage of this reliability, because it allows the manufacture of components that do not require excessive replacement or maintenance even when subjected to regular heating or intense light exposure.
Alongside its optical and thermal roles, barium fluoride also serves as a crucial material in the chemical industry. It is used as a precursor in the production of specialty glasses and ceramics, where its presence influences the structural rigidity and behavior of the final product. In high-performance glasses, for instance, it helps reduce reflection, improve transparency, and enhance resistance to radiation. BaF₂ also appears in the manufacture of molten salt mixtures that require predictable ionic chemistry and stability. Its use in such mixtures reflects a very different side of the compound compared with its optical applications, yet both underline the same fundamental truth: the stable ionic identity of barium fluoride forms the basis of its technological versatility.
Scientific research also benefits from the unique characteristics of BaF₂, particularly in the study of scintillation — the process by which materials emit light when exposed to ionizing radiation. Barium fluoride scintillators display extremely fast response times, allowing them to play a role in detecting high-energy particles and supporting radiation measurement systems. Experiments in nuclear physics, astrophysics, and even medical imaging take advantage of this compound’s ability to convert high-energy interactions into measurable light signals. Although other scintillating materials exist, BaF₂ remains valuable because its rapid emission response provides data that slower materials cannot capture with the same precision.
The environmental and chemical stability of the compound also contributes to its continued popularity. In many ambient conditions, barium fluoride remains inert and does not react aggressively with moisture or common atmospheric elements. While it can dissolve in strong acids or slowly degrade in prolonged exposure to water, it is generally durable and long-lived wherever it is used. Its low chemical activity in normal conditions helps prevent contamination and unwanted interactions, which is particularly important in optical and spectroscopic setups where chemical deterioration could interfere with instrumentation accuracy.
Yet despite its stability and utility, barium fluoride must be handled with care due to the toxicity associated with soluble barium compounds. When BaF₂ is intact as a solid crystal or installed in equipment, it presents no hazard, but ingestion or improper handling during manufacturing processes can introduce health concerns because the fluoride and barium ions can be harmful in biological systems. As a result, responsible industrial protocols rely on proper protective equipment, sealed component design, and careful waste management when working with the material in powdered or reactive form. These safety measures ensure that the compound can be used effectively without compromising the health of workers or the environment.
When expressed visually as a vector illustration, barium fluoride is often depicted through a variety of styles that, while artistic, are rooted in molecular accuracy. The central metal ion is shown coordinated with surrounding fluoride atoms in a symmetric arrangement that reflects the electrostatic attraction between oppositely charged ions. In educational contexts, such illustrations help learners understand the lattice nature of ionic solids, using BaF₂ as a model to explain how charge balance dictates molecular structure. In professional settings, a vector illustration can support product documentation, research presentations, and industrial diagrams by offering a clean, infinitely scalable image that communicates the compound’s structure without loss of clarity.
Taken together, barium fluoride embodies a striking combination of chemical simplicity and technological sophistication. It is structurally straightforward, consisting only of barium and fluoride ions arranged in a repeating lattice, yet this simplicity gives birth to properties that have made the compound indispensable across optics, scientific experimentation, nuclear detection, and advanced manufacturing. Every field that utilizes BaF₂ is ultimately relying on the invisible power of ionic bonding, crystal regularity, and atomic clarity that the compound naturally provides. The vector illustration of barium fluoride is therefore much more than a scientific graphic — it is a symbolic bridge connecting the microscopic world of atomic structure with the macroscopic world of engineering, research, and industry. It translates the abstract into the visible, helping both experts and learners appreciate how a solid based purely on electrostatic attraction can become a foundational material for some of the most demanding technologies in the modern world.