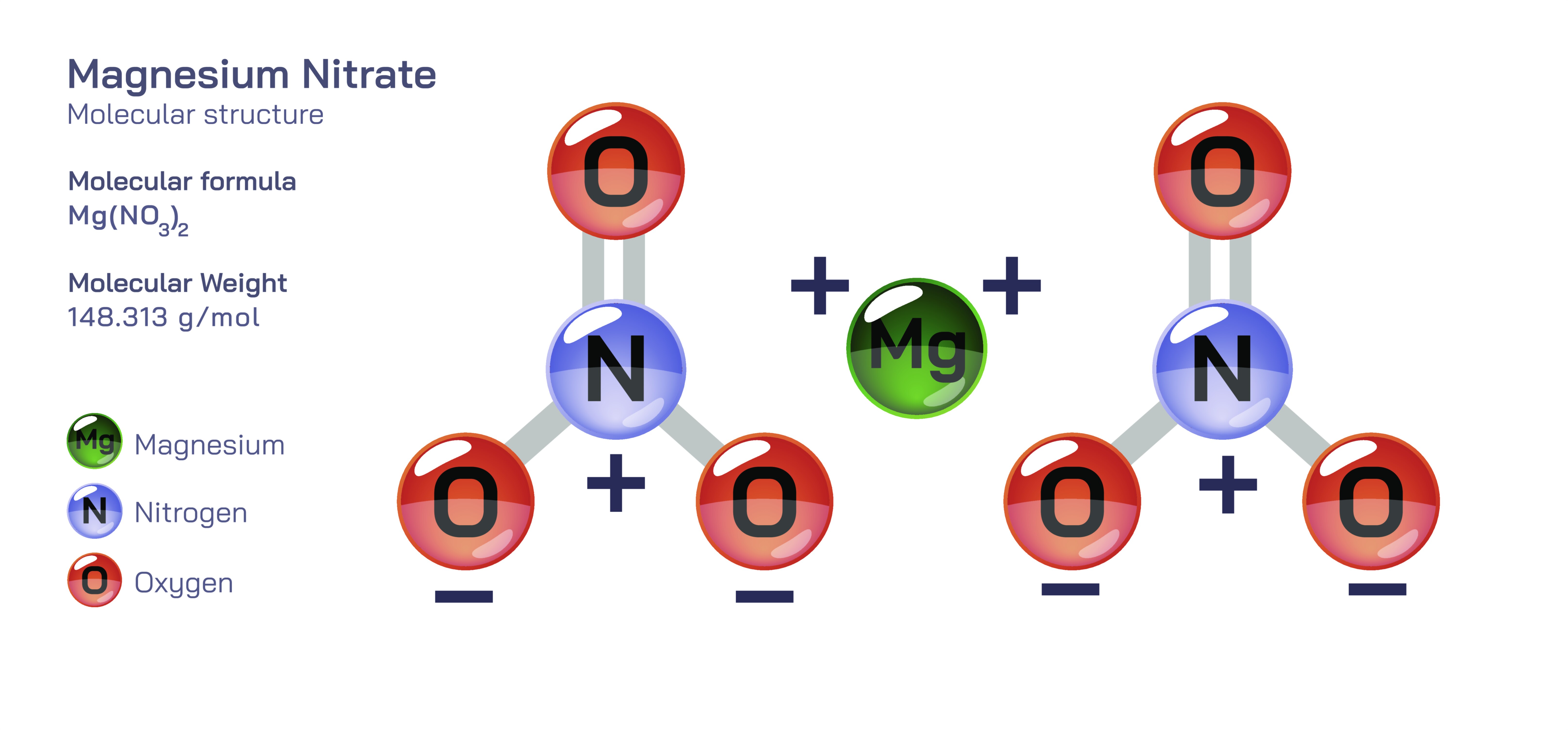
Magnesium Nitrate — Chemical Structure and Composition
Magnesium nitrate is an inorganic compound represented by the chemical formula Mg(NO₃)₂, and it stands as an important example of how the structural arrangement of ions determines the physical behavior, chemical reactivity, and practical uses of a substance across science, agriculture, industry, and environmental management. At its core, the compound is built from a divalent magnesium cation bound through ionic attraction to two nitrate anions, each composed of one nitrogen atom surrounded by three oxygen atoms. The geometry of the nitrate ion, which is planar and exhibits resonance among its oxygen atoms, creates a highly stable and evenly distributed arrangement of electron density. When two of these nitrate units interact electrostatically with the positively charged magnesium ion, the result is a crystalline lattice that dissolves readily in water and dissociates into free ions without requiring extreme conditions. This exceptional solubility is one of the most defining aspects of magnesium nitrate, making it a powerful supplier of magnesium and nitrogen — two of the most essential nutrients that support plant development, biological chemistry, and industrial reactions. In nature, magnesium nitrate can occur in limited concentrations in mineral deposits and cave environments, but commercial use largely relies on synthesized forms produced by reacting magnesium compounds with nitric acid under controlled conditions to ensure purity, consistency, and safety.
In agricultural systems, magnesium nitrate plays an important role as a dual nutrient fertilizer that supplies both magnesium and nitrogen in forms that are immediately available to plants. The nitrate portion contributes nitrogen that plants rapidly absorb through root systems to synthesize proteins, chlorophyll, and enzymes needed for healthy metabolism and vigorous growth. The magnesium ion, on the other hand, is central to the structure of chlorophyll, the light-absorbing pigment responsible for photosynthesis, and it helps regulate carbohydrate transport, enzyme activation, and energy transfer inside plant tissues. Because the nitrogen component exists in the nitrate form rather than the ammonium form, it supports fast nutrient assimilation without acidifying the soil, which helps maintain a stable root environment for crops that are sensitive to changes in pH. High-value plants such as fruit orchards, vegetables, greenhouse crops, and hydroponic systems particularly benefit from magnesium nitrate due to its excellent solubility and ability to prevent magnesium deficiency symptoms such as leaf yellowing, poor fruit formation, and reduced sugar production. Unlike some mineral fertilizers that require microbial conversion before being usable, magnesium nitrate delivers nutrients directly, enabling farmers and agronomists to fine-tune plant feeding programs with precise timing and efficiency.
Magnesium nitrate is also deeply relevant in industrial and chemical manufacturing processes where its molecular composition and thermal behavior contribute to both transformative reactions and specialized applications. When heated, magnesium nitrate undergoes controlled decomposition that produces magnesium oxide while releasing nitrogen oxides and oxygen, a reaction pathway that has made the compound valuable in pyrotechnics, energetics, and oxidation-based chemical processes. The oxygen-releasing capability of nitrates is essential in manufacturing formulations requiring strong oxidizing agents, and magnesium nitrate is known for producing high-purity oxidation environments with predictable thermal profiles. The resulting magnesium oxide formed during decomposition also serves industrial needs, particularly in ceramics, refractories, electronics, specialty coatings, and environmentally friendly construction materials. Production lines requiring uniform metal-oxide conversion appreciate magnesium nitrate because its breakdown produces fine particles with consistent distribution and controlled surface area, properties vital for performance in catalysis, adsorbent materials, and advanced composite systems.
Water treatment and environmental engineering represent another sphere where magnesium nitrate demonstrates notable usefulness. Wastewater and landfill gas treatment systems apply magnesium nitrate to biologically reduce the formation of hydrogen sulfide, a highly corrosive and dangerous gas that results from the decomposition of organic matter under oxygen-deficient conditions. By providing a nitrate source that supports sulfur-oxidizing microbial pathways, magnesium nitrate helps redirect microbial metabolism toward environmentally safer byproducts instead of sulfide gas production. In doing so, it also minimizes odor pollution, infrastructure corrosion, and health hazards within sewage facilities and industrial treatment plants. The magnesium component additionally contributes to alkalinity regulation and buffering capacity in water systems, helping stabilize pH during biological treatment cycles. These properties collectively place magnesium nitrate at the intersection of safety, operational reliability, and ecological management, which is increasingly important as industries shift toward regulatory compliance and sustainability-driven wastewater solutions.
Because of its strong hydrophilic nature, magnesium nitrate exists commercially in hydrated forms, most commonly magnesium nitrate hexahydrate, in which six water molecules are incorporated into the crystalline matrix. This hydration dramatically influences melting behavior, appearance, storage, and reactivity. Hydrated magnesium nitrate melts to form a clear, viscous liquid at relatively low temperatures, and its high water affinity enables it to act as a powerful desiccating agent in certain controlled-humidity environments. At the same time, because the compound pulls moisture from the air so efficiently, it must be stored in tightly sealed containers under dry conditions to prevent unwanted liquefaction or structural breakdown. In laboratory work, this strong moisture interaction is particularly relevant because the difference between anhydrous and hydrated forms affects mass calculations, molarity, and reaction stoichiometry. Researchers and analysts therefore emphasize strict control of environmental exposure to ensure consistent and accurate use of magnesium nitrate in synthesis, titrations, analytical tests, and solution preparation.
Magnesium nitrate also plays a role in chemical synthesis, metallurgical processing, and materials engineering where nitrate ions participate in complex transformations involving redox reactions, coordination chemistry, and surface modification. In catalyst production, magnesium nitrate is used to impregnate support materials in preparation for calcination, allowing uniform distribution of magnesium that later enhances catalytic selectivity, stability, or mechanical strength. In the development of advanced ceramics, the uniform release of magnesium and nitrogen-oxide gases during thermal processing helps fabricate microstructures with controlled porosity, density, and grain morphology, which improves performance in high-temperature applications, heat sinks, and electronic insulators. In organic chemistry, magnesium nitrate participates in nitrate exchange and oxidation mechanisms that contribute to the synthesis of specialized compounds used in pharmaceuticals, coatings, and energy materials. Each of these applications highlights the compound’s versatility in both structural formation and chemical transformation.
Despite its usefulness, magnesium nitrate requires careful handling because, like all nitrate salts, it possesses strong oxidizing properties that can intensify the combustion of flammable materials if stored or used improperly. Safety guidelines stress avoiding contact with organic fuels, reducing agents, or high heat sources outside controlled industrial or laboratory processes. Its high solubility also means that spill management must include measures to prevent uncontrolled nutrient loading into natural water systems, since nitrate release in aquatic environments can lead to eutrophication and ecological imbalance. With proper containment, ventilation, and personal protection practices, magnesium nitrate is considered safe and dependable, and its long record of industrial use has produced a clear understanding of its storage and operational requirements.
Magnesium nitrate embodies the connection between molecular design and practical benefit. Its ionic arrangement supports a wide range of functions — nourishing crops with precision, aiding the reduction of hazardous gases, supporting catalytic and ceramic technologies, enabling efficient oxidation processes, and contributing to nuanced research in both analytical and applied chemistry. Each property can be traced back to the fundamental structure of the compound: a magnesium ion surrounded by two resonating nitrate groups that create a system both soluble and reactive, stable yet transformable under heat, and predictable across a multitude of physical and chemical environments. For these reasons, magnesium nitrate continues to serve as an essential resource for agriculture, industry, and scientific exploration, and as technologies evolve toward higher productivity and environmental responsibility, its significance is expected to grow even further.