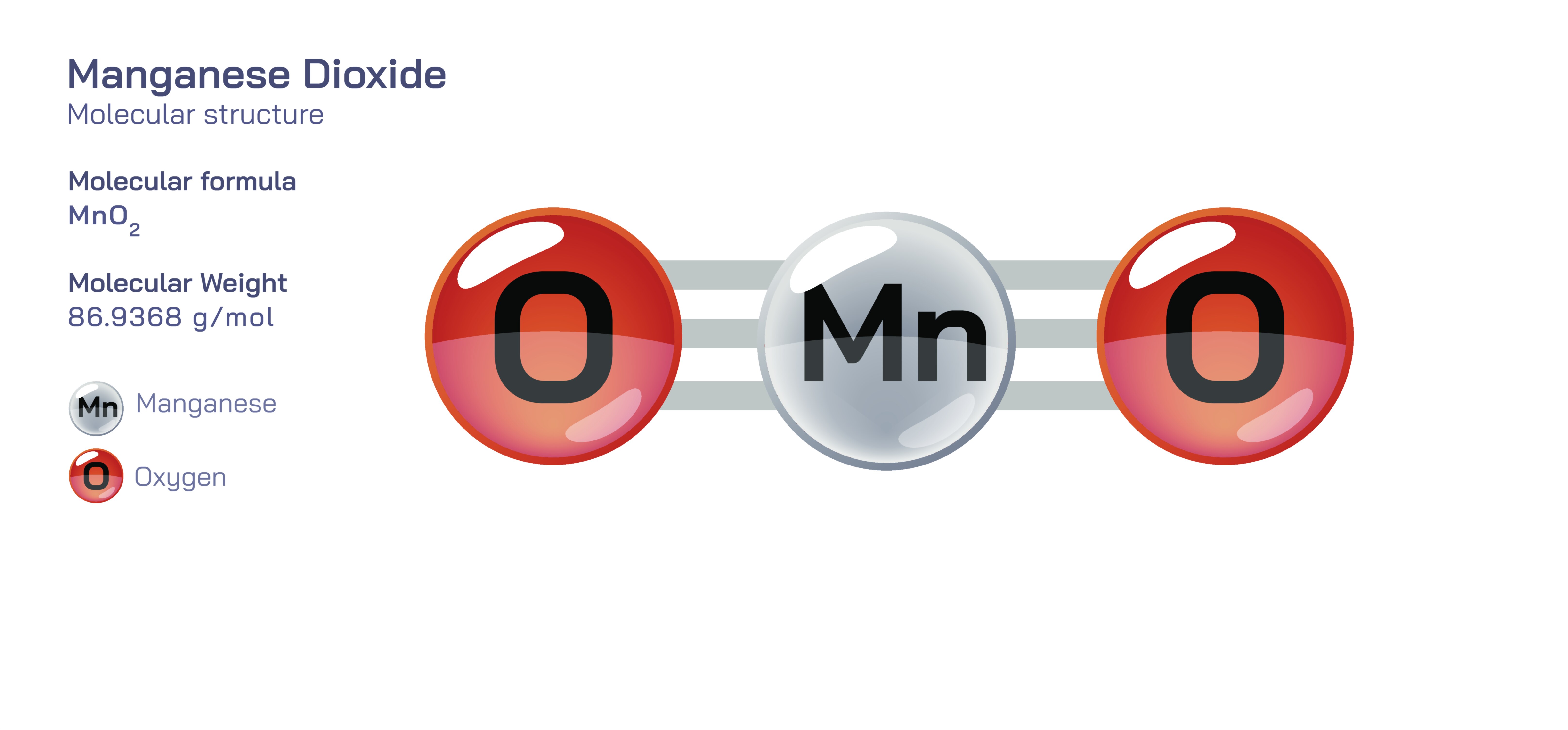
Manganese Dioxide — Molecular Structure and Composition
Manganese dioxide is an inorganic compound of exceptional chemical, industrial, geological, and technological importance, and its significance is inseparable from the structural organization of its atoms, the oxidation state of manganese, and the layered architecture of its crystalline lattice. Represented by the chemical formula MnO₂, this compound consists of manganese in the +4 oxidation state bonded to oxygen atoms through a three-dimensional network of metal–oxygen octahedra. Although the formula appears simple, manganese dioxide exists in several polymorphic forms—with the most common being the naturally occurring mineral pyrolusite—where MnO₆ octahedra share edges and corners to form tunnel-like and layered structures. These intricate atomic arrangements determine electron mobility, catalytic behavior, ion diffusion capacity, magnetic properties, and thermal stability, which is why manganese dioxide is simultaneously a geological mineral, a catalyst, a battery material, and a component of chemical synthesis. The ability of MnO₂ to reversibly participate in electron transfer reactions without collapsing its structural framework lies at the foundation of its technological value in energy-storage systems, oxidation reactions, and purification processes. Its lattice is rigid enough to maintain stability under extreme physical and chemical environments, yet versatile enough to allow ions to migrate through its microscopic tunnels—an important characteristic that represents the molecular logic behind its practical applications across modern scientific fields.
From a chemical standpoint, manganese dioxide is best known for its strong oxidizing properties, which originate from the high oxidation state of manganese and the compound’s ability to accept electrons during chemical reactions. When MnO₂ encounters reducing agents, the manganese center can shift from its +4 state to lower oxidation states, such as +2 or +3, allowing it to participate in transformations that break down organic molecules, decompose unstable compounds, or convert unwanted substances into inert products. For more than a century, manganese dioxide has served as a reliable catalyst in laboratory and industrial oxidation reactions, helping accelerate processes without itself being permanently consumed. One of its classic roles is in the catalytic decomposition of hydrogen peroxide to release oxygen, a reaction that demonstrates how MnO₂ lowers activation energy while promoting the rapid breakdown of reactive oxygen species. In organic chemistry, finely powdered manganese dioxide selectively oxidizes allylic and benzylic alcohols into carbonyl compounds, doing so with a level of precision that avoids over-oxidation or unwanted reaction with other functional groups. This characteristic selectivity shows that manganese dioxide’s reactivity is not only strong but controlled, stemming directly from the geometry of the oxygen-rich manganese surface and the arrangement of open catalytic sites.
The role of manganese dioxide in energy storage is one of the most influential illustrations of how its atomic lattice drives technological progress. MnO₂ has been the cathode material in traditional alkaline batteries and dry-cell batteries for decades, converting chemical energy to electrical energy through reversible redox reactions involving the manganese center. During discharge, Mn⁴⁺ is partially reduced to Mn³⁺ as electrons move through the external circuit to power devices. The robustness of the MnO₂ crystal structure allows this electron transfer to occur without immediate lattice collapse, making the material durable and widely affordable. As renewable energy and portable electronics continue to expand, manganese dioxide remains critical in next-generation battery designs—especially in rechargeable zinc–manganese and lithium–manganese systems—because the layered structure can be engineered to improve ion diffusion, electrical conductivity, and cycling stability. By manipulating tunnel dimensions, defect concentrations, and particle morphology on the nanoscale, scientists can significantly enhance energy density, power output, and service life, demonstrating how the molecular composition of MnO₂ becomes a toolkit for solving modern energy challenges.
Manganese dioxide also stands at the crossroads between chemistry and environmental management because of its capacity to adsorb, neutralize, and transform contaminants. In water treatment processes, MnO₂ removes dissolved metals such as iron and manganese by catalyzing their oxidation into insoluble forms that can be filtered out, restoring water clarity and safety. It also plays a role in removing hydrogen sulfide and other sulfur-based impurities that cause odor and corrosion in groundwater supplies. In soil and geological environments, naturally occurring manganese dioxide contributes to the cycling of organic carbon and toxic trace elements by interacting with microbial metabolism and redox gradients. These environmental processes demonstrate that MnO₂ is not only a technological material but a natural engine of geochemical transformation, controlling the distribution of metals and nutrients across ecosystems. The mineral’s ability to cycle between oxidation states is central to soil fertility, river chemistry, and the long-term processing of organic matter that decomposes in terrestrial and aquatic environments.
In manufacturing and materials science, manganese dioxide contributes properties that enhance the performance of glass, ceramics, pigments, and metallurgical processes. When introduced into molten glass, MnO₂ acts as a decolorizing agent by oxidizing iron impurities responsible for greenish tints and converting them into forms that neutralize coloration. In ceramics, it serves as a pigment that produces rich brown, black, or purplish colors, depending on firing conditions and composition. In steel production, manganese dioxide participates indirectly through its transformation into compounds that remove unwanted sulfur, improving the strength, ductility, and processing characteristics of the final metal. Its thermal stability and electronic behavior also support the production of catalysts, welding fluxes, and friction materials. These applications reflect how the structure and composition of manganese dioxide become an invisible yet essential contributor to everyday products and industrial infrastructure.
In the field of medicine and biotechnology, manganese dioxide has drawn interest because of its antioxidant, oxygen-release, and catalytic behavior in microenvironments where oxidative stress is a concern. Researchers are exploring MnO₂ nanoparticles as smart therapeutic agents capable of regulating hydrogen peroxide levels in diseased tissues, helping neutralize free radicals and improve oxygenation in tumors, wounds, or inflammatory regions. Manganese dioxide can act as a carrier for drugs, biomolecules, or enzymes, degrading gradually under biological conditions and releasing manganese ions that are naturally processed by the body. Although clinical applications remain in development and require careful dosage control to avoid toxicity, the molecular properties of MnO₂—particularly its ability to catalyze the decomposition of reactive oxygen species—suggest a promising future in biomedical engineering and targeted therapeutic strategies.
Beyond its scientific and industrial dimensions, manganese dioxide also reflects a deeper lesson about how atomic structure defines real-world function. A single metal atom surrounded by oxygen atoms in a geometric arrangement can become a catalyst, a battery material, a water filter, a glass modifier, a geological regulator, a pigment, a biomedical nanomaterial, and a part of consumer electronics. The unifying principle behind these functions is the adaptability of the manganese oxidation state and the stability of the MnO₆ octahedral network, which allow the material to trade electrons, interact with ions, withstand extreme conditions, and yet maintain coherence. Changing the size of MnO₂ particles, tuning crystallinity, introducing vacancies, or modifying surfaces does not alter the elemental composition but dramatically reshapes conductivity, reactivity, adsorption capacity, and energy-storage performance. Thus, manganese dioxide stands as a vivid example of how the most impactful properties in chemistry arise not only from what elements are present, but from how atoms are arranged and how electrons move through the resulting structure.
In every setting—from natural rock formations to cutting-edge battery electrodes—manganese dioxide demonstrates that molecular architecture is destiny. The octahedral lattice that binds manganese and oxygen is not just a pattern; it is a blueprint for behavior, enabling the compound to support biological cycles, industrial reactions, technological innovation, and environmental sustainability simultaneously. Whether powering a portable device, purifying groundwater, shaping the hue of ceramics, participating in the evolution of planetary minerals, or inspiring new catalytic and biomedical technologies, manganese dioxide continues to prove that the structure of matter determines what is possible in science and in the world around us.