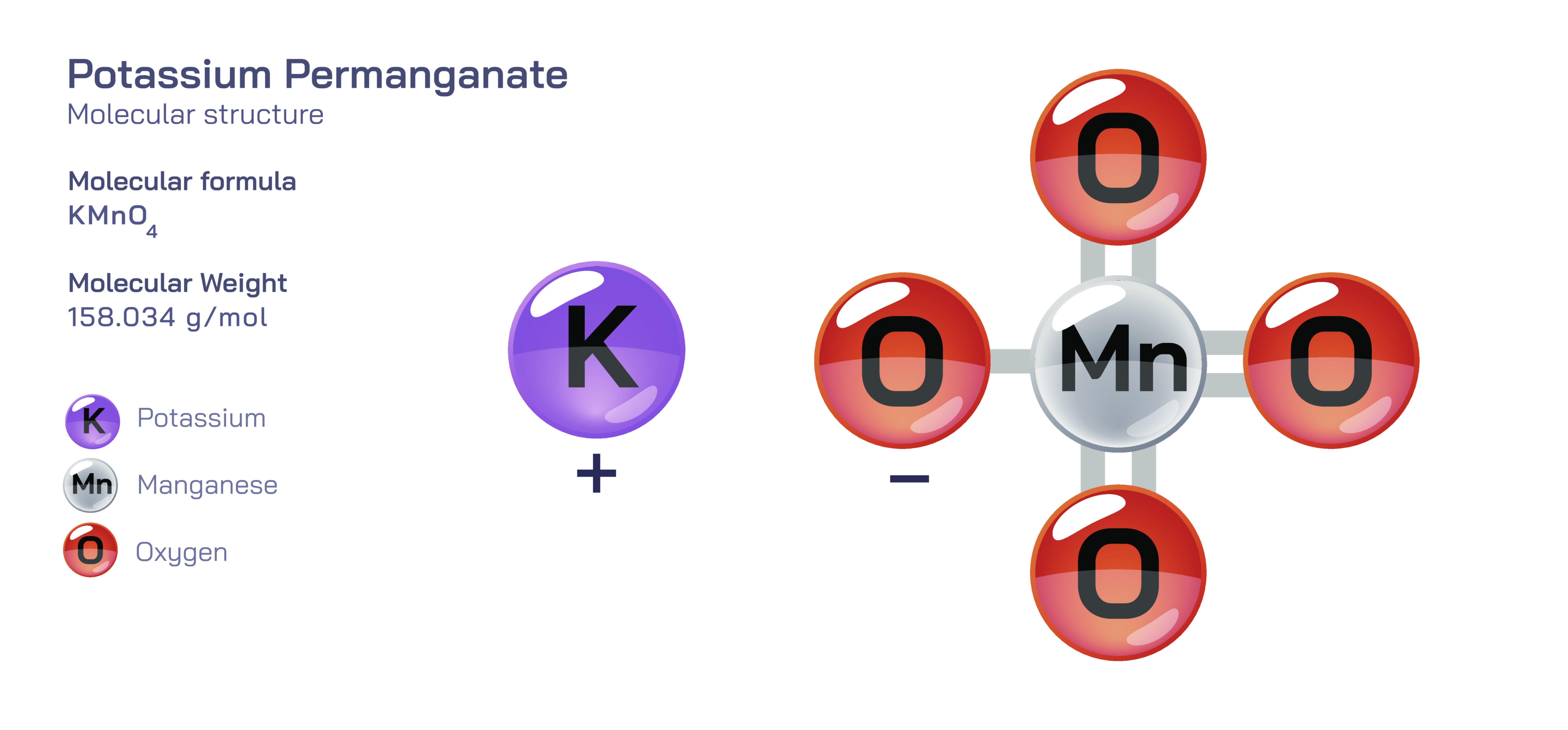
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐏𝐞𝐫𝐦𝐚𝐧𝐠𝐚𝐧𝐚𝐭𝐞 (𝐊𝐌𝐧𝐎₄) 𝐎𝐱𝐢𝐝𝐢𝐳𝐞𝐫, 𝐔𝐬𝐞𝐬 𝐚𝐧𝐝 𝐒𝐚𝐟𝐞𝐭𝐲.
Potassium permanganate is one of the most distinguished inorganic oxidizing agents in chemistry, recognizable instantly by its deep purple crystalline appearance and its intense ability to accept electrons from other substances, driving chemical reactions forward with a powerful oxidative force. The compound consists of potassium ions and permanganate ions, but its real strength lies within the structure of the permanganate ion itself. A single manganese atom sits at the core surrounded tetrahedrally by four oxygen atoms, forming a strongly bonded, symmetrical unit that places manganese in a very high oxidation state. Because of this oxidation state, manganese has a strong tendency to withdraw electrons from other chemical species in an attempt to reduce itself to a more stable form. This electron-seeking behaviour is the foundation of potassium permanganate’s role as one of the most reliable oxidizers across medical, industrial, environmental and laboratory applications. The manganese atom’s drive to achieve a lower oxidation state makes permanganate capable of triggering deep transformations in organic molecules, killing microbes by destroying cellular structures and eliminating contaminants by oxidizing harmful substances into inert forms. Its physical stability in solid form and dramatic responsiveness in solution make it an exceptional example of how the structural design of a molecule determines its usefulness.
When potassium permanganate dissolves in water, the permanganate ion becomes free and expresses its full chemical personality. The deep purple colour visible in dilute solutions is not simply aesthetic; it is the optical result of electron transitions occurring inside the permanganate structure. The manganese atom holds electrons in orbitals that are sensitive to visible light, and the absorption of certain wavelengths leaves only the purple hue for the human eye to perceive. This colour becomes an indicator of concentration and chemical reaction progress. When permanganate encounters substances capable of donating electrons, it shifts colour as it is reduced to manganese dioxide or manganese ions, producing brown or colourless products depending on the environment. This transition is not just a visual change but a structural shift inside the molecule. As soon as manganese receives electrons, the saturation of the oxygen coordination network relaxes and restructures, and the purple colour fades. The change in colour therefore allows scientists, doctors, engineers and water treatment specialists to monitor reactions and detect chemical changes in real time without needing sophisticated instruments. The disappearance of the purple tint marks the completion of oxidizing action, illustrating once again that structure communicates behaviour directly.
As an oxidizer, potassium permanganate has a long and influential scientific history. In laboratory chemistry, it acts as a classical reagent capable of oxidizing alcohols to aldehydes or acids, breaking carbon–carbon double bonds, generating epoxides, and converting organic contaminants into harmless products. It has been used for more than a century in titrations known as redox volumetric analysis because it behaves predictably and visibly during electron transfer. These titrations require no external indicator because the permanganate colour acts as its own visual endpoint, proving that structural transitions inside the permanganate ion align with macroscopic tracking. Even in strictly controlled analytical setups, the value of potassium permanganate lies in the precision of the permanganate redox system: it reacts cleanly, completions are easy to observe, and the reduction products are stable when conditions are properly maintained.
Potassium permanganate’s oxidizing power gives it an important medical and hygienic role as well. In medical applications, dilute solutions are used externally as antiseptics to treat infected wounds, ulcers, dermatitis, athlete’s foot and fungal infections. The oxidizing behaviour enables the permanganate ion to destroy microbial membranes, denature harmful proteins and deactivate biological pathogens. Unlike antibiotics, which affect only certain metabolic pathways, potassium permanganate works through non-selective oxidative destruction and therefore does not lead to microbial resistance when used appropriately. Because the permanganate ion does not penetrate deeply through intact skin and does not persist biologically, it is physiologically safe when used in mild concentrations. At controlled dilution, it supports wound healing by reducing harmful bacterial populations and drying excessive secretions. In dermatology, physicians rely on potassium permanganate baths to treat eczema, blistering skin conditions and weeping wounds because oxidizing action reduces inflammation and limits bacterial presence on the surface. These therapeutic applications stem directly from the structural ability of permanganate to transfer oxygen equivalents to organic systems, altering harmful compounds without affecting deeper tissues when properly diluted.
Environmental purification is another field deeply dependent on potassium permanganate. In water treatment systems, the compound is used to eliminate iron, manganese and hydrogen sulfide impurities by oxidizing them into insoluble forms that can be filtered away. This is not coincidental but structurally predictable: the permanganate ion is more capable of accepting electrons than the dissolved contaminants are of retaining them, leading to a controlled conversion. Municipal water treatment, industrial wastewater cleanup and groundwater remediation use potassium permanganate to degrade organic pollutants, pesticides and industrial solvents that have contaminated soil and aquifers. Its ability to destroy toxins without creating heavy-metal residue or long-lasting chemical by-products anchors its environmental relevance. When the reaction is complete, manganese is left in an insoluble and less reactive form, and this end product settles rather than persist in water, which is why potassium permanganate is preferred over harsher oxidizers in ecological applications.
Industrial applications of potassium permanganate follow from the same structural principles. It is used in air purification units to remove ethylene gas in produce storage rooms, slowing down fruit ripening and preventing spoilage. It oxidizes odorous organic gases into non-odorous forms, promoting hygiene in waste handling facilities and sewage treatment systems. In metallurgy, it supports surface cleaning, passivation and metal preparation by oxidizing surface residues that interfere with bonding or plating. In textile manufacturing, potassium permanganate is used to bleach and fade denim fabric because it breaks down colour molecules in a controlled manner. The versatility of these processes comes from the fact that permanganate does not burn chemically like a fuel oxidizer but instead performs precision electron capture reactions that restructure molecules without excessive heat release.
Although potassium permanganate is chemically versatile, its powerful oxidizing ability requires strict safety consideration. If mishandled, it can react dangerously with organic materials, concentrated acids, glycerol, powdered metals and reducing agents. Because the permanganate ion seeks electrons aggressively, contact with easily oxidized substances can cause combustion or decomposition reactions that release heat rapidly. For this reason, storage recommendations require the compound to be kept away from fuels, solvents, greases, wood dust and other combustible materials. The solid form must be stored in dry conditions, protected from contamination, and handled carefully because even trace contact with reducing residues can trigger undesired reactions. Skin or mucous membrane contact with concentrated crystals or solutions can lead to burns because the oxidizing action does not distinguish between laboratory samples and biological material. Inhalation of dust should be avoided because permanganate can irritate or damage respiratory tissues. These risks do not negate the usefulness of potassium permanganate but instead illustrate that any compound with strong chemical power must be controlled with awareness of its structural reactivity.
The purple colour of potassium permanganate therefore represents more than pigmentation—it marks a compound in a highly energetic oxidation state, ready to participate in electron-transfer chemistry that can cure infection, disinfect water, detoxify environments, support laboratory reactions, enable industrial purification and transform manufacturing processes. Its structure dictates its behaviour: the high oxidation state of manganese drives reactivity, the tetrahedral oxygen coordination maintains stability until reaction occurs, the colour changes signal reduction completion, and the insoluble manganese products that form afterward make the compound environmentally manageable. Every use—medical, agricultural, industrial, environmental and laboratory—flows directly from the interaction of the permanganate ion with surrounding molecules.
Potassium permanganate is therefore a one-of-a-kind oxidizer whose molecular design has allowed it to remain relevant for more than a century. Its usefulness depends on the harmony between structural stability and reactivity: stable enough to store safely in the right conditions, powerful enough to oxidize contaminants in water, strong enough to neutralize microbes, and predictable enough to serve as a scientific indicator. When handled with respect, it becomes a tool of precision chemistry and public health. When mishandled, its oxidative power becomes a potential hazard. Understanding its oxidizing nature, uses and safety does not simply catalogue a list of applications—it reveals the direct link between molecular structure and the material world, showing once again that chemical behaviour is written in the architecture of atoms long before it reaches laboratories, industries, hospitals or environmental systems.