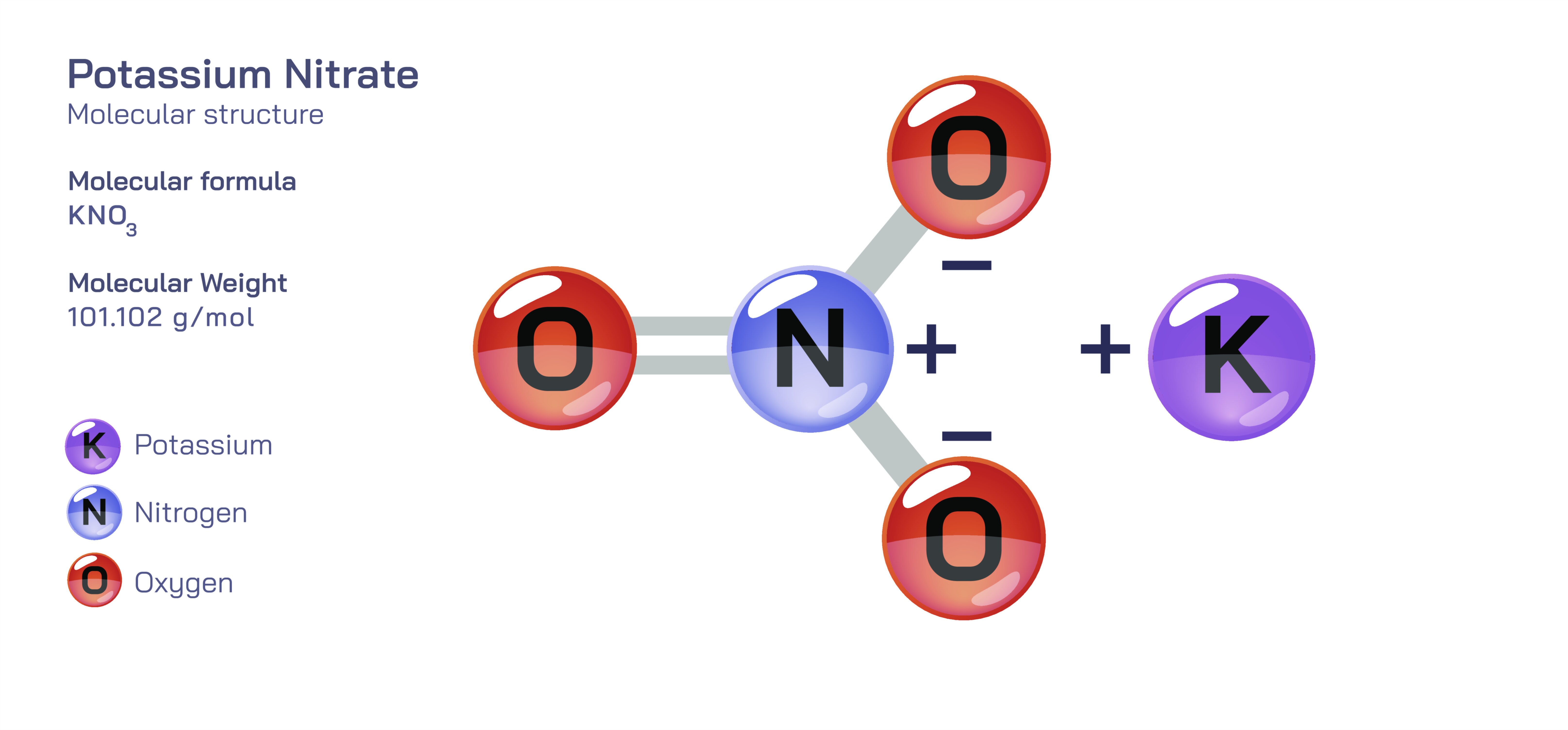
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐍𝐢𝐭𝐫𝐚𝐭𝐞 (𝐊𝐍𝐎₃) 𝐔𝐬𝐞𝐬.
Potassium nitrate is a widely used inorganic compound that has played an influential role in chemistry, agriculture, medicine, food preservation, industry and even historical advancements in warfare. Although it appears as a simple crystalline salt composed of potassium, nitrogen and oxygen, its usefulness arises from the remarkable balance between its chemical reactivity and physical stability. Potassium nitrate’s nitrate ion provides a strong oxidizing property, while the potassium ion helps maintain solubility and electrolyte balance. This dual nature allows the compound to release oxygen thermally or chemically when required, yet remain safe enough for long-term storage under controlled conditions. Throughout different fields, potassium nitrate has become valued for improving plant growth, enhancing manufacturing processes, accelerating chemical reactions, maintaining food quality, enabling controlled combustion and assisting in medical and dental applications. It is one of the rare minerals whose usefulness spans ancient history and modern science, confirming that its applications are deeply rooted in its chemical identity.
One of the most extensive uses of potassium nitrate lies in agriculture as a fertilizer. Plants require potassium and nitrogen as major nutrients, and potassium nitrate provides both of them together in a highly soluble form that roots can absorb efficiently. The nitrate portion stimulates rapid vegetative growth by supporting protein synthesis and metabolism in plant cells, while the potassium ion strengthens stems, improves water regulation, increases resistance to drought stress and enhances overall plant immunity. Farmers and horticulturists often prefer potassium nitrate in fertigation systems and greenhouse cultivation because its chloride-free nature protects sensitive crops such as strawberries, tomatoes, grapes and leafy vegetables from salt toxicity. It also improves flowering and fruiting quality in high-value crops, making it a preferred nutrient source for precision agriculture. Slow and sustained nutrient availability from potassium nitrate helps avoid the destructive impact of nutrient imbalances and enables predictable plant development. Across commercial farming, hydroponics and nursery production, it is considered a scientifically efficient fertilizer that supports yield, crop health and post-harvest quality.
Industrial applications of potassium nitrate are equally important and can be traced back centuries to when it became a key component in gunpowder. When potassium nitrate is heated or combined with combustible materials, the nitrate supplies oxygen that accelerates burning. This ability to support oxidizing combustion made it essential in propellants, fireworks, blasting powders and pyrotechnics. Firework manufacturers rely on potassium nitrate for producing brightly coloured flame effects because its oxygen-releasing behaviour enables fuels and metal salts to burn at high temperatures. In this context, the compound does not merely serve as an additive; it is the structural driver of ignition efficiency and flame stability. Modern industrial pyrotechnics refine this same principle for airbag propellants, emergency flares and safety signalling devices. Potassium nitrate is not explosive on its own, but its controlled oxygen delivery transforms fuel mixtures into reliable energy systems, explaining why it has maintained a central position in combustion technologies from historical warfare to contemporary engineering.
Beyond pyrotechnics, potassium nitrate holds important value in metallurgy and ceramics. In metallurgical heat treatment, molten potassium nitrate baths are used for surface hardening processes such as case hardening and quenching, where the salt bath transfers heat evenly and reduces surface oxidation. This uniform heat exchange improves strength and durability in steels and alloy components. In glass and ceramic manufacturing, potassium nitrate helps control thermal expansion, glaze properties and optical clarity. It provides the oxygen-rich atmosphere required to stabilize high-temperature processes and supports smooth surface finishing in specialty glass, optical lenses and ceramic tiles. The compound’s compatibility with industrial scaling and its predictable chemical behaviour allow manufacturers to maintain product consistency and production efficiency.
The food industry also benefits from the chemical characteristics of potassium nitrate. Historically, it served as a curing and preserving agent for meats because it inhibits the growth of bacteria, particularly those responsible for spoilage and botulism. Although regulations now limit nitrate use in many processed foods, potassium nitrate still appears in traditional cured meats, certain cheese recipes, brined products and artisanal preservation techniques. Its mode of action is tied to slowing bacterial metabolism, reducing oxygen demand inside food tissues and maintaining colour and flavour stability. Even where direct food use has decreased, potassium nitrate continues to support safe preservation indirectly through the production of sodium nitrite and potassium nitrite, essential curing salts that originate from nitrate conversion. The antimicrobial behaviour of the nitrate ion ensures food safety without requiring artificial preservatives in traditional preservation systems.
Medical and dental applications of potassium nitrate are less widely known to the general public but scientifically significant. In medicine, potassium nitrate has mild vasodilating and diuretic effects and has been used historically to treat respiratory woes, urinary discomfort and circulatory disorders. Although modern pharmaceuticals have replaced many of these functions, potassium nitrate still appears in topical treatments and medicinal preparations. In dentistry, its use has increased dramatically because of its effectiveness in treating tooth sensitivity. Potassium nitrate reduces dental hypersensitivity by calming the nerve endings inside dentinal tubules. The potassium ion blocks nerve signal transmission and reduces overreaction to temperature or pressure changes, which is why potassium nitrate is one of the main active ingredients in many desensitizing toothpaste formulas. Unlike surface-coating agents, potassium nitrate addresses sensitivity from within the tooth structure, gradually restoring comfort without damaging enamel or soft tissue. Its safety in oral care and long-term compatibility with bio-mineral surfaces make it an ideal therapeutic component in dental science.
Potassium nitrate also plays a role in environmental and laboratory chemistry. In wastewater treatment and aquaculture, nitrate acts as a controlled nutrient source for beneficial microorganisms, balancing nitrogen cycles without triggering ammonia toxicity. It enhances biofiltration efficiency and supports ecological stability in recirculating aquatic systems. In analytical chemistry, potassium nitrate solutions serve as ionic strength adjusters and electrolyte standards for studying electrochemical behaviour. Because it dissolves completely and does not form complex ions readily, its presence improves accuracy in potentiometric and conductivity measurements. In laboratories it is also used as a mild oxidizer in various inorganic syntheses and reaction pathways, where controlled oxidation rather than aggressive reactivity is required.
Even in everyday consumer goods, potassium nitrate appears more often than many realize. It is used in heat packs that rely on exothermic crystallization, in antifreeze blends for heat-transfer fluids and in specialized fertilizers for household gardening. Combinations of potassium nitrate with other plant nutrients are marketed widely as performance fertilizers because of their ability to support both rapid growth and crop strength. Animal feed supplementation may also incorporate potassium nitrate under controlled conditions to stabilize nitrogen balance and support selective rumen microbial activity in livestock.
Every use of potassium nitrate is directly connected to its chemistry. Its oxidizing nature explains its inclusion in pyrotechnics, heat treatments and industrial combustion. Its nutrient composition and solubility explain its agricultural importance. Its ability to inhibit bacterial growth explains its historical role in food preservation. Its bioelectrical regulatory function explains its dental and medical applications. Its chemical neutrality and strong dissociation explain why it remains useful in laboratories and environmental systems. Few compounds demonstrate such scientific versatility with behaviour rooted in such a simple ionic formula.
The story of potassium nitrate shows that usefulness in chemistry does not require structural complexity. Instead, it requires the precise alignment of chemical traits with functional needs. The oxidizing capability of the nitrate ion, the electrolyte stability of potassium, the solubility of the compound and the controlled reactivity under heat or redox stimulation have together created a material that has shaped industries, advanced technologies, supported agriculture, contributed to medicine and even influenced world history. From ancient black powder to modern precision agriculture and clinical dentistry, potassium nitrate remains an example of how the properties of a single compound can sustain relevance across centuries of scientific progress, proving that true chemical value endures when structure and function operate in harmony.