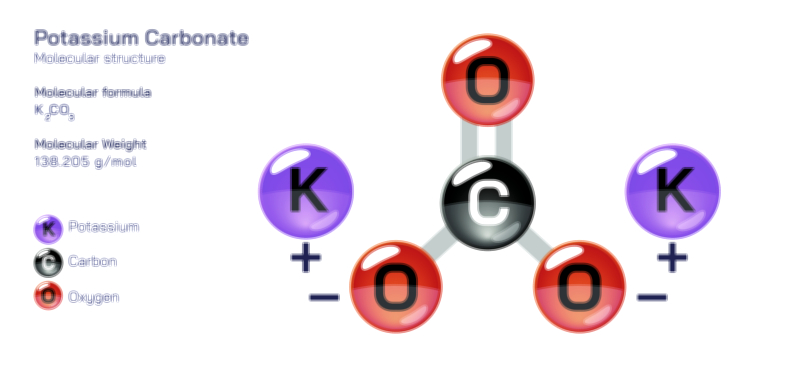
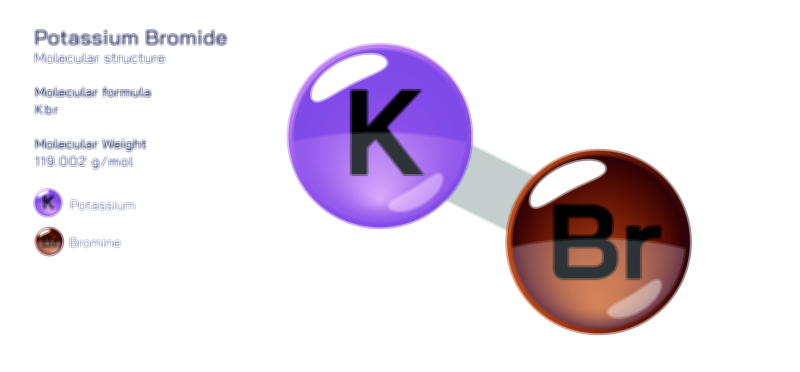
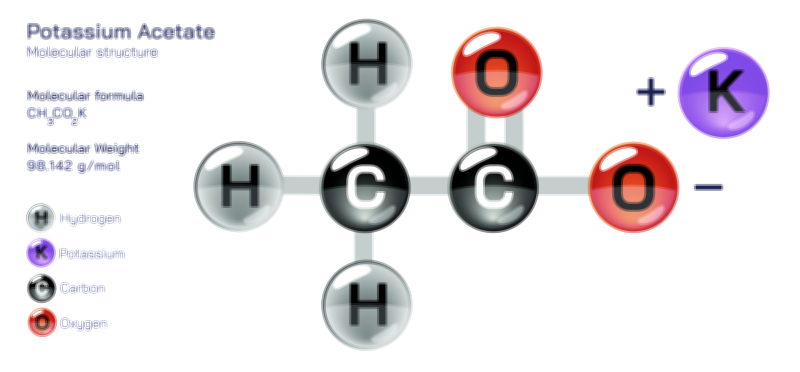
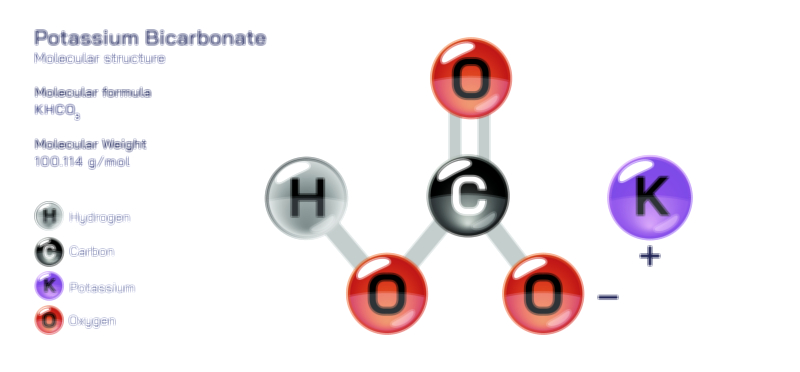
ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉćØÉ▓ØÉ®ØÉ©ØÉ£ØÉĪØÉźØÉ©ØɽØÉóØÉŁØÉ× ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×
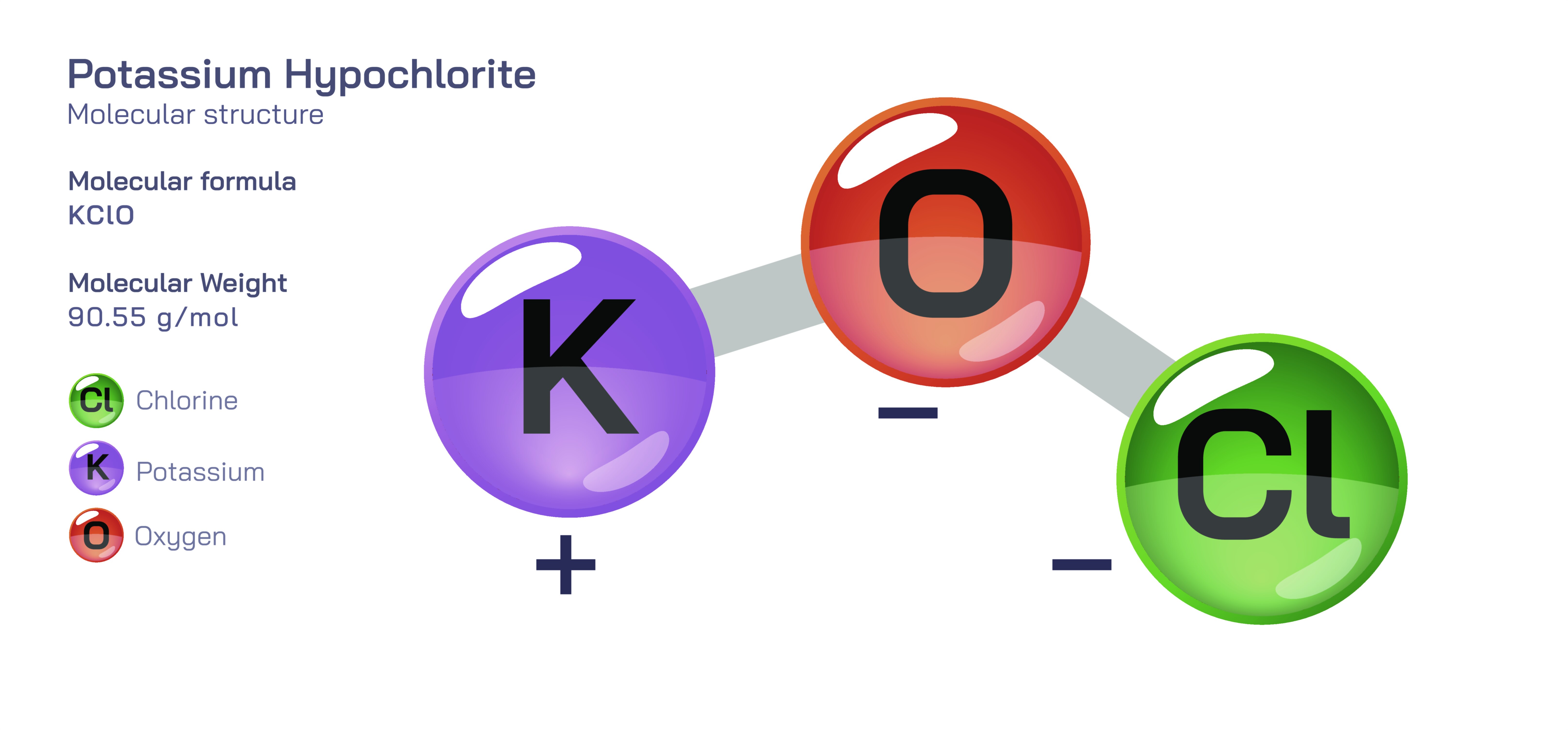
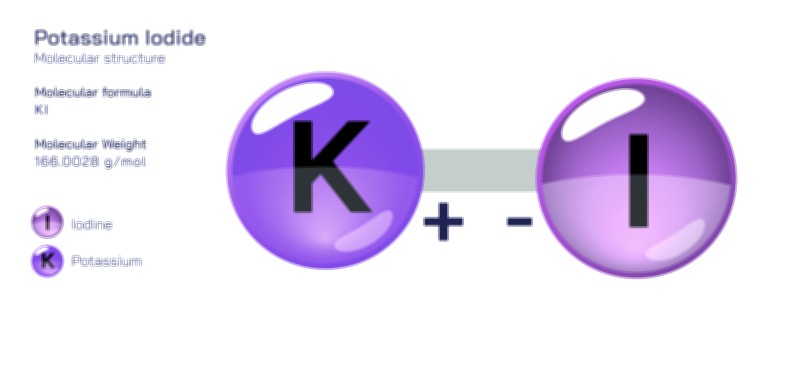
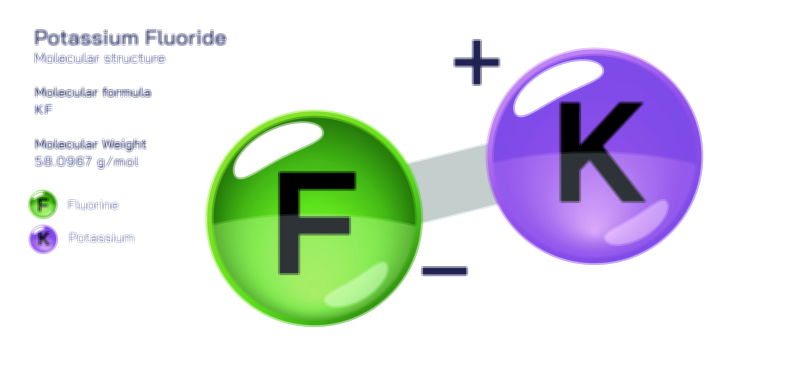
Potassium hypochlorite is an inorganic compound whose molecular structure reflects a characteristic relationship between metal ions and oxyhalogen ions. At the centre of its identity is an ionic pairing between a potassium ion and a hypochlorite ion. The potassium ion arises after losing one electron to attain a stable positive charge, while the hypochlorite ion consists of a chlorine atom bonded to an oxygen atom and carrying a negative charge. This hypochlorite unit forms through an internal covalent bond between chlorine and oxygen, creating a polar arrangement that distributes electron density unequally across the molecule. The oxygen atom retains more electron density, and chlorine carries a higher oxidation state, giving the ion strong oxidizing tendencies. When the hypochlorite ion and potassium ion assemble, they form the compound known as potassium hypochlorite, whose crystal lattice is held together by electrostatic attraction rather than directional covalent forces. The compound does not express molecular geometry in the traditional single-molecule sense because it does not exist as a discrete neutral molecule; instead, its structure consists of independent ions in an ordered lattice.
The structure becomes more expressive when potassium hypochlorite dissolves in water. The lattice breaks apart immediately because both potassium and hypochlorite ions interact strongly with water molecules. The potassium ion becomes fully solvated and remains comparatively inactive in solution, acting only as a counter-ion to balance charge. The hypochlorite ion, however, maintains its internal covalent structure while moving freely in solution, and this is where the chemistry of the compound manifests. At the molecular level, the hypochlorite ion is defined by the chlorine atom bonded to oxygen in such a way that the oxygen holds a pair of electrons available for accepting protons and engaging in hydrogen bonding, while the chlorine atom remains electron deficient and highly reactive. This asymmetry is responsible for the ionŌĆÖs powerful disinfecting and bleaching properties. The structural arrangement places chlorine in a high oxidation state that is energetically inclined to accept electrons during reactions. As soon as potassium hypochlorite enters an aqueous environment, this structural behaviour becomes dominant, allowing hypochlorite ions to interact with microbial and organic molecules, breaking down cellular components and oxidizing contaminants.
Another important aspect of the structure lies in the dynamic equilibrium behaviour of the hypochlorite ion in solution. Although the ionic form remains intact under alkaline conditions, the hypochlorite ion becomes less stable when acidity increases. Structural rearrangements can produce chlorine gas, chlorate ions, or other oxychlorine species depending on temperature and pH. This means the molecular architecture is highly sensitive to its environment rather than static. Under strongly alkaline conditions, the oxygenŌĆōchlorine covalent bond in the hypochlorite ion stays secure and the compound behaves predictably as a disinfecting oxidizer. In weakly acidic surroundings, however, the equilibrium shifts and the hypochlorite ion begins to transform. These structural tendencies explain why potassium hypochlorite must always be stored in alkaline solutions to maintain stability and why industrial and laboratory formulations are carefully controlled to preserve its useful ionic identity and prevent unwanted decomposition.
The hypochlorite ionŌĆÖs structure also dictates its role in oxidation reactions. Because chlorine holds an elevated oxidation state in the OŌĆōCl arrangement, it has the capability to accept electrons during chemical transformations, leading to a reduction process that often converts hypochlorite into chloride. This conversion marks the end point of a typical disinfecting or bleaching reaction where chlorine transitions from an aggressive oxidizing form to a more stable reduced ion. The oxygen atom in the ion complements this behaviour by providing electron density that helps stabilize chlorine during reaction transitions. Every step of this transformation is driven by the internal architecture of the ion rather than by potassium, which remains chemically quiet in the background. This division of molecular labour is characteristic of hypochlorite salts: the alkali metal merely stabilizes the ionic network, while the oxychlorine species defines the chemistry.
In solid form, potassium hypochlorite is less commonly encountered because it is most stable as an aqueous solution. When crystallization occurs, the compound forms an ionic lattice with potassium ions and hypochlorite ions arranged in an orderly pattern. The lattice offers temporary physical stability but is highly hygroscopic and sensitive to heat and light, reflecting the reactive nature of the hypochlorite component. This sensitivity originates from the structural tension within the OŌĆōCl bond, which is strong enough for ionic function yet weak enough to undergo redox-driven decomposition under stress. The compoundŌĆÖs reactivity has practical value in bleaching, sanitizing, and deodorizing applications but also demands careful handling to prevent breakdown and release of gaseous chlorine.
The molecular structure of potassium hypochlorite therefore defines every aspect of its identity. The ionic relationship between potassium and hypochlorite explains its solubility, lattice integrity, and dissociation in solution. The internal OŌĆōCl covalent configuration explains its oxidizing capacity, disinfecting properties, and chemical instability under acidic or heated conditions. The high oxidation state of chlorine within the hypochlorite ion explains both its effectiveness and its sensitivity, making structural awareness essential for proper use and storage. Despite appearing simple in composition, potassium hypochlorite demonstrates how a small shift in atomic arrangement and electron distribution can produce a compound with significant technological usefulness and high biological activity, illustrating once again that structure lies at the heart of chemical function.