Ammonium Persulfate — Chemical Structure, Properties, and Functional Behavior
Understanding the Composition and Structural Identity of Ammonium Persulfate
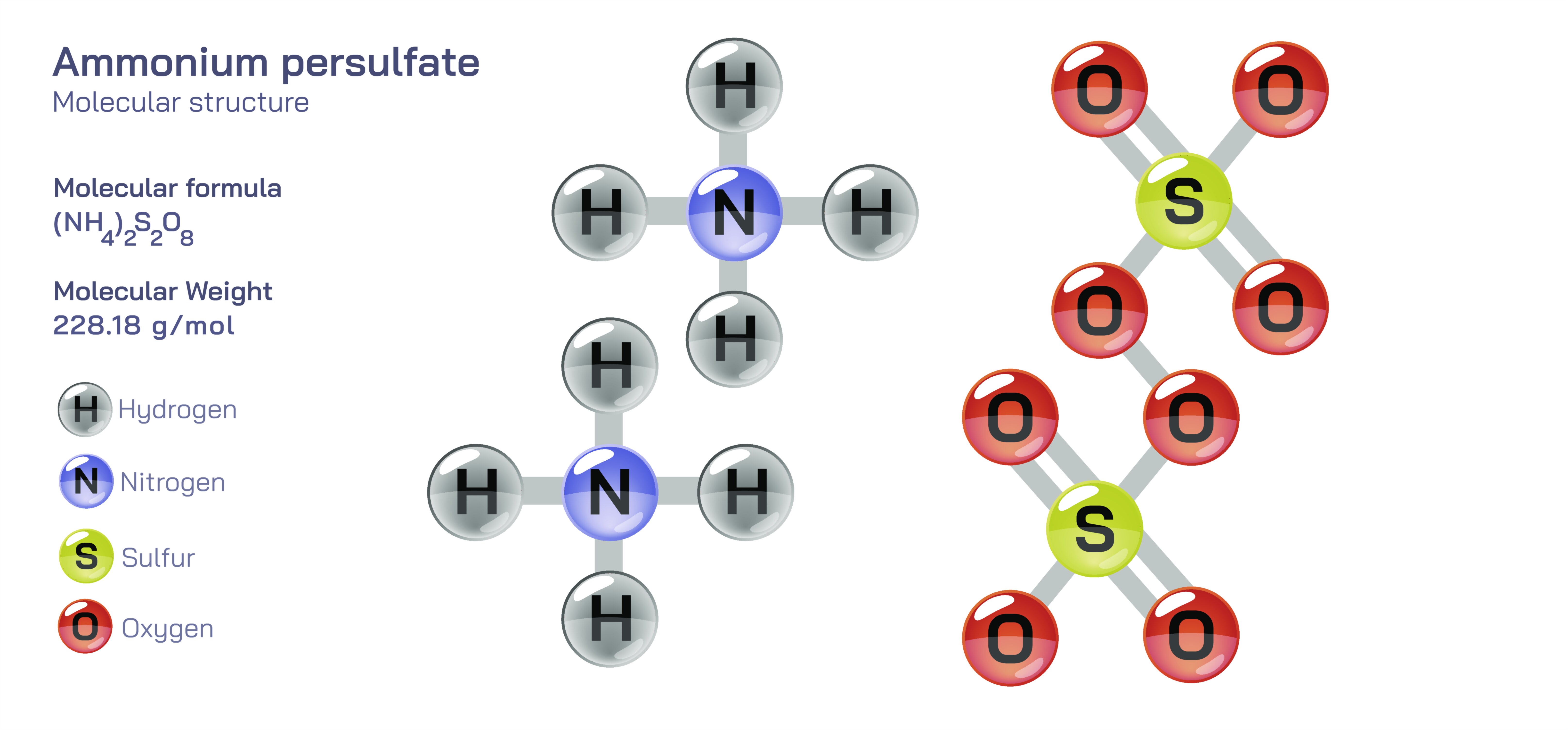
Ammonium persulfate, chemically represented as
(
𝑁
𝐻
4
)
2
𝑆
2
𝑂
8
(NH
4
)
2
S
2
O
8
, is a powerful oxidizing agent widely used in industrial chemistry, polymer science, analytical processes, and laboratory applications. Its structure consists of two ammonium ions balancing a persulfate dianion, which contains a unique peroxy bond linking two sulfate groups. This internal peroxide linkage is the defining feature of the molecule, giving it strong oxidizing power and the ability to participate in radical-generating reactions. When depicted in a vector illustration, ammonium persulfate is typically shown with two tetrahedral sulfate groups joined by an oxygen-oxygen single bond, a structure that visually communicates the high-energy nature of the persulfate species. The ammonium ions, often displayed as tetrahedral arrangements around a central nitrogen, complete the ionic lattice. This clear structural visualization helps explain why the compound behaves so vigorously in chemical environments where it decomposes to generate sulfate radicals—species that drive oxidation, polymer initiation, and surface cleaning. The contrasting roles of the ammonium cations and the reactive persulfate anion create a compound that is both stable in solid form yet highly reactive when dissolved or heated.
Key Physical and Chemical Properties That Define Its Behavior
Ammonium persulfate forms white crystalline salts that dissolve readily in water, producing a clear solution capable of strong oxidizing reactions. Its high solubility ensures that the persulfate ion becomes chemically active quickly once introduced into aqueous systems. The compound is stable under normal dry conditions but decomposes in the presence of moisture, heat, or impurities, releasing oxygen and sulfate radicals. These radicals are short-lived but extremely reactive, enabling the compound to break down organic materials, initiate polymerizations, and oxidize various substrates. Ammonium persulfate is also non-volatile and relatively easy to handle in solid form, although it requires careful storage in cool, dry conditions to prevent premature decomposition. The oxidizing nature of the persulfate ion makes it incompatible with reducing agents and combustible materials. In vector illustrations, its decomposition pathway is often represented by radiating arrows indicating the cleavage of the O–O bond, symbolizing the formation of active radicals. This visual representation reinforces the idea that the structure of ammonium persulfate directly dictates its chemical potency and reactivity profile.
Industrial Roles and Applications in Polymerization Processes
One of the most important uses of ammonium persulfate is as a free-radical initiator in polymer manufacturing. In systems such as acrylamide polymerization, vinyl acetate polymer formation, or styrene-based resin production, ammonium persulfate generates sulfate radicals upon decomposition. These radicals attack monomer molecules and initiate the chain reactions that lead to polymer growth. Because its decomposition occurs in a controlled and predictable manner, ammonium persulfate offers precise timing in polymerization reactions, which is essential for achieving consistent molecular weights, viscosity levels, and product clarity. Water-soluble polymers used in wastewater treatment, paper manufacturing, and gel electrophoresis often rely on persulfate initiation. In vector illustrations, polymer chains are often shown branching from radical sites produced by the persulfate anion, visually capturing the compound’s role as the spark that gives rise to long molecular chains.
Analytical and Laboratory Uses in Oxidation and Sample Preparation
In analytical chemistry, ammonium persulfate is a key reagent for oxidative digestion, sample preparation, and redox-based analytical techniques. It is commonly used to break down organic contaminants, decompose interfering substances, and oxidize compounds to more measurable forms. In laboratories, it also finds frequent use in gel electrophoresis, especially when forming polyacrylamide gels for protein and DNA separation. The compound initiates gel polymerization when paired with a secondary catalyst, enabling the formation of transparent gels with precisely controlled pore sizes. This application is central to molecular biology, where separation accuracy determines the success of experiments. The radical-generating ability of ammonium persulfate also makes it valuable for surface treatment of metals, textiles, and plastics, where it removes organic residues and activates surfaces for bonding or coating. A vector illustration of these uses often includes laboratory flasks, polymer gel matrices, or surface-cleaning schematics to visually represent how the compound functions in research and analytical settings.
Environmental and Cleaning Applications Driven by Oxidation Power
Ammonium persulfate also has environmental applications, especially in wastewater treatment and soil remediation. When introduced into contaminated environments, it generates reactive species that oxidize organic pollutants, breaking them down into simpler, less harmful compounds. This process, known as advanced oxidation, is particularly effective for petroleum hydrocarbons, chlorinated solvents, and other persistent organic pollutants. In cleaning applications, ammonium persulfate is used to remove tarnish, bleach materials, or strip impurities from industrial surfaces. It is favored for these tasks because it produces little residue and decomposes into non-toxic sulfate once its reactive oxygen has been expended. Vector illustrations often depict pollutant molecules being broken apart by reactive oxygen species, using arrows and fragmentation symbols to communicate the compound’s environmental utility.
Safety Considerations and Handling Practices
Although ammonium persulfate is extremely useful, it must be handled with caution due to its strong oxidizing nature. The compound can cause skin and respiratory irritation, particularly in powdered form, and may accelerate combustion if it contacts organic materials. Its decomposition is exothermic, meaning that improper storage or exposure to heat can lead to rapid breakdown or hazardous reactions. Industrial users must store it in cool, dry environments away from incompatible substances, and laboratory personnel typically work with it using gloves, masks, and safety ventilation. Understanding these safety characteristics is essential because the very properties that make ammonium persulfate valuable—the ability to generate radicals and oxidize substances—also create potential hazards. Vector illustrations frequently include warning symbols, storage icons, or decomposition diagrams to highlight best practices in handling.
Visualizing Ammonium Persulfate Structure Through Vector Graphics
A well-designed vector illustration of ammonium persulfate typically emphasizes the central persulfate dianion with its distinctive O–O bond, flanked by ammonium ions that balance the charge. The tetrahedral geometry of the sulfate groups is rendered with clear bond angles and atomic labels, while arrows or glow effects often highlight the peroxy linkage. Additional diagrams may depict the dissociation of ions in solution or the radical formation that occurs during decomposition. These visuals help bridge the gap between structural chemistry and functional behavior, making the compound’s reactivity easier to conceptualize.