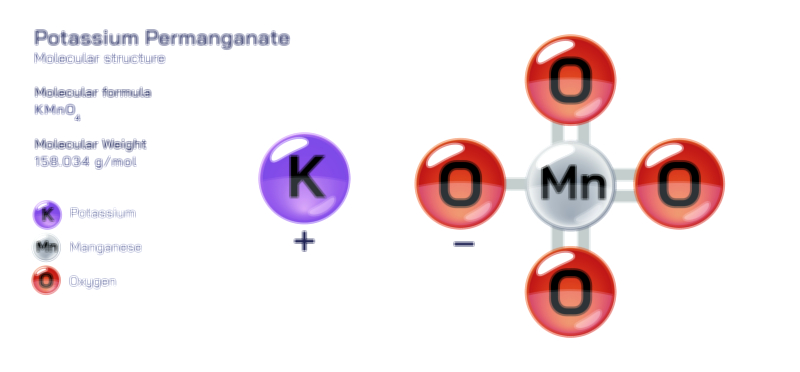
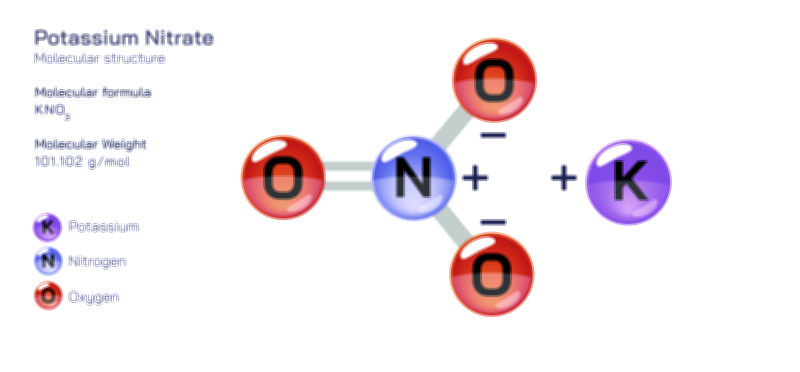
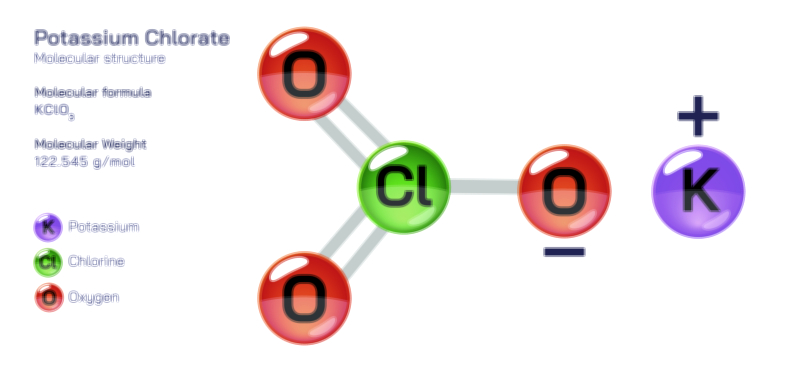
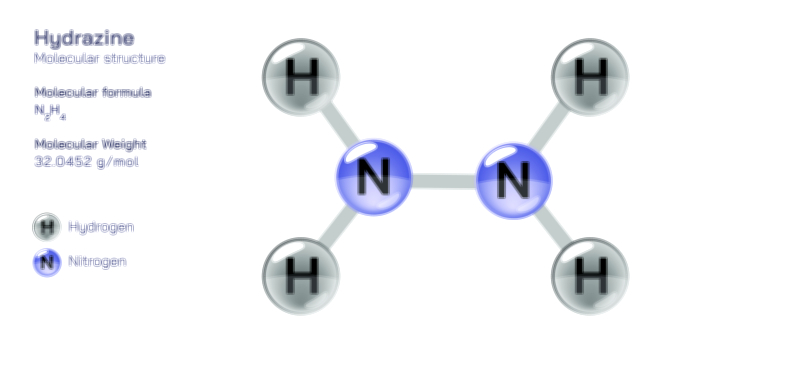
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐍𝐢𝐭𝐫𝐢𝐭𝐞 (𝐊𝐍𝐎₂) 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞.
Potassium nitrite is an inorganic compound whose identity and behaviour are rooted in the structural interaction between potassium ions and nitrite ions, forming a crystalline material with a distinctive internal architecture and highly influential chemical properties. The structure of potassium nitrite is defined by the way its ions arrange themselves in the solid state and how they separate and function in aqueous environments. On a basic chemical level, potassium nitrite consists of a potassium ion that carries a positive charge and a nitrite ion that carries a negative charge. This pairing is not based on electron sharing, as found in covalent molecules, but rather on complete electron transfer from potassium to the nitrite unit, resulting in a strong electrostatic attraction that holds the salt together. When observed in a single formula unit, this relationship appears simple, but the true structural complexity emerges when these ions assemble into a large-scale crystalline lattice in which alternating potassium and nitrite ions repeat in an ordered three-dimensional pattern. The regularity of this lattice provides the solid with its stability and external salt-like appearance, yet within this extended framework, the nitrite ion preserves its own internal geometry that ultimately determines the chemical behaviour of the compound.
To understand the structure of potassium nitrite, it is crucial to examine the molecular architecture of the nitrite ion itself, because that is where the complexity resides. The nitrite ion is not a simple straight-line arrangement of atoms; rather, it adopts a bent geometry with a nitrogen atom positioned between two oxygen atoms in a shape that distributes electron density unevenly. The nitrogen–oxygen bonding network contains both covalent and resonance-stabilized character, meaning that electrons within the ion are continuously shared among the two nitrogen–oxygen bonds, preventing one bond from being permanently stronger or weaker than the other. This internal distribution of charge gives the nitrite ion a planar triangular shape and a polarized electron cloud, with one side of the ion holding slightly more negative character than the other. These structural attributes give nitrite a strong tendency to engage in redox chemistry and substitution reactions and to coordinate with metal ions in highly specific ways. In the crystal lattice of potassium nitrite, each nitrite ion aligns according to its charge distribution, and the potassium ions arrange themselves to minimize repulsion and maximize ionic attraction. While the nitrite ion retains a covalently bonded internal structure, the potassium ion remains spherical and electronically inert in comparison, functioning primarily as a counter-ion that stabilizes the lattice without influencing the internal bond network of nitrite.
When potassium nitrite dissolves in water, the ionic lattice collapses almost instantly because the attraction between the ions and water molecules becomes stronger than the attraction between the ions themselves. The aqueous form of the compound consists of free potassium ions dispersed in solution and nitrite ions that retain their bent molecular geometry and resonance-averaged electron distribution. It is in this dissolved form that the chemical identity of potassium nitrite becomes fully active, because the nitrite ion expresses the behaviour that results from its structural arrangement. The oxygen atoms of the nitrite ion possess lone pairs of electrons that allow the ion to attract positively charged species, act as a ligand toward certain metals, or participate in proton transfer reactions under acidic conditions. In addition, the nitrogen center inside the nitrite ion plays a key role in oxidation–reduction chemistry because of its intermediate oxidation state, which makes the ion capable of transitioning to either nitrate in an oxidation reaction or nitric oxide in a reduction pathway. These transformations illustrate how the internal structure of the nitrite ion is designed to allow flexibility while retaining its geometric foundation: the triangular arrangement remains, but electron density can shift to allow the ion to support multiple functional changes.
The structural attributes of potassium nitrite also explain its sensitivity to environmental conditions. While the compound remains relatively stable under neutral or alkaline conditions, acidic environments trigger a dramatic transformation because hydrogen ions attack the electron-rich oxygen atoms of the nitrite ion. As the structure accepts protons, the internal distribution of charge shifts, and the nitrite ion converts into nitrous acid, which then rapidly decomposes to produce gases such as nitric oxide and nitrogen dioxide. This sequence reveals how the geometry of the nitrite ion is closely tied to its chemical stability; the bent nitrogen–oxygen framework is stable when deprotonated, but when the structure is forced into protonation, its internal electron system collapses and rearranges to form new species. Because nitrite ions release nitrogen-based gases during decomposition, potassium nitrite cannot be stored in acidic conditions or mixed with strong acids without safety controls. In industrial and laboratory contexts, this structural pathway is not only a hazard but also a deliberate tool that allows nitrite to serve as a controlled source of nitrogen oxide compounds when used properly.
A deeper structural consequence emerges when nitrite interacts with biological molecules, especially proteins that contain iron centers. The nitrite ion has the capacity to bind to metal ions through either nitrogen or oxygen, a behaviour known as ambidentate coordination. This dual bonding capability arises directly from the resonance character and electron distribution within the nitrite structure, and it allows the ion to associate with metal proteins in biological systems. In hemoglobin and other metalloproteins, nitrite can attach to metal centers and participate in electron-transfer cycles that convert hemoglobin derivatives into nitric oxide carriers under certain conditions. These subtle but structurally dictated interactions are the reason potassium nitrite has pharmacological relevance and why it has been used in medicine and physiology to understand vascular regulation and oxygen transport. The structure of the nitrite ion therefore influences not only inorganic reactivity but also biological signalling pathways in which nitric oxide plays a central role.
In the crystalline form of potassium nitrite, the orientation of the nitrite ion controls how the lattice grows and clears imperfections. Because the ion is not spherical but asymmetric and planar, the angles at which the ions pack affect how the crystal fractures or responds to temperature changes. The bent geometry makes the solid material moderately hygroscopic, meaning it can absorb moisture from the atmosphere, since water interacts strongly with the polarized oxygen atoms of the nitrite ion. This sensitivity does not destroy the internal structure of nitrite but affects how the crystal holds together physically, demonstrating the relationship between microscopic charge distribution and macroscopic stability.
Throughout all phases of matter, potassium nitrite behaves according to its structural identity. The potassium ion stabilizes the system electrically, remaining chemically quiet and structurally spherical. The nitrite ion, however, acts as a reactive functional unit whose bent geometry, delocalized electrons and multi-centred charge distribution allow it to serve as a reducing agent, an oxidizing agent, a ligand, a proton acceptor and, under specific conditions, a precursor to gaseous nitrogen compounds. The structure of the nitrite ion determines its chemical behaviour entirely, and the lattice arrangement of potassium nitrite ensures that this functional ion is held in a form that is easy to handle, store, dissolve and apply.
Taken together, the structure of potassium nitrite illustrates how a compound with a simple formula can possess deep internal complexity and wide scientific impact. Its crystalline lattice explains its solid-state stability. Its dissociation in water explains its behaviour as an electrolyte and reactive anion. The geometric and resonance-stabilized nature of the nitrite ion explains its multifaceted chemistry. The structural sensitivity to pH explains its decomposition into nitrogen-based gases. The ability to coordinate metals explains its biochemical significance. Through structure and function bound together, potassium nitrite stands as an example of how even a single cation–anion pair can support a network of chemical, physical and biological behaviours when the underlying molecular architecture is sophisticated enough to accommodate both stability and transformation.