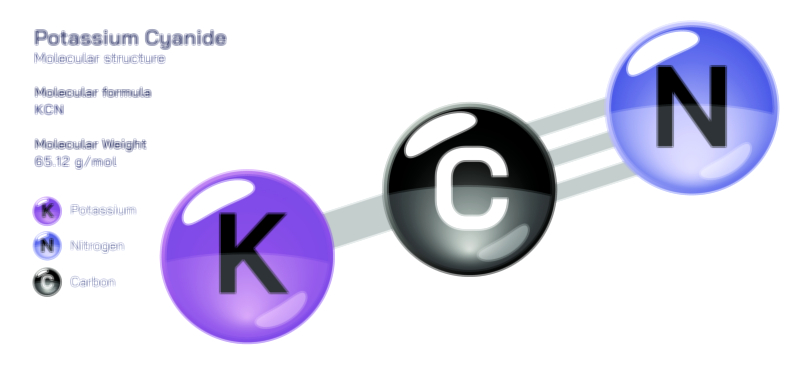
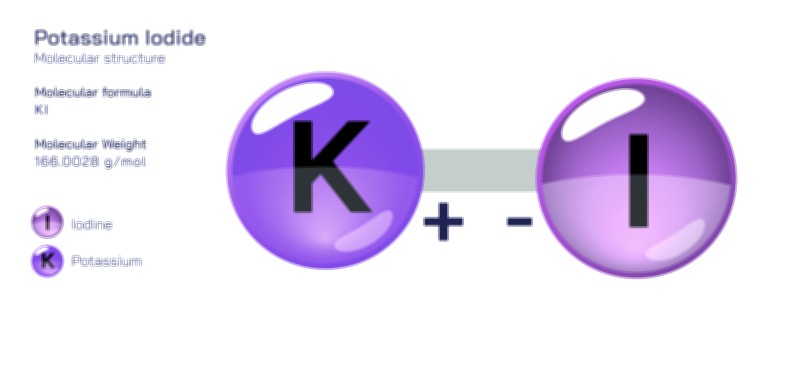
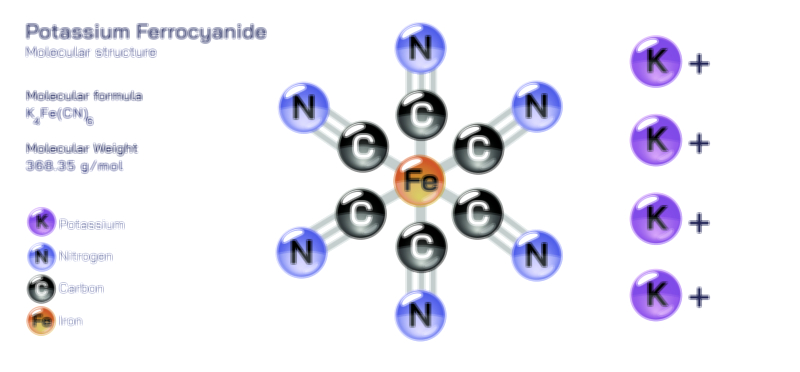
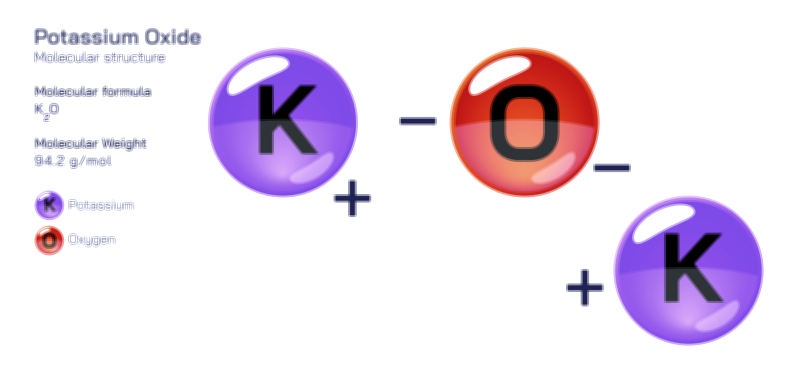ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉāØÉóØÉ£ØÉĪØɽØÉ©ØÉ”ØÉÜØÉŁØÉ× ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉÜØɦØÉØ ØÉÆØÉÜØɤØÉ×ØÉŁØÉ▓
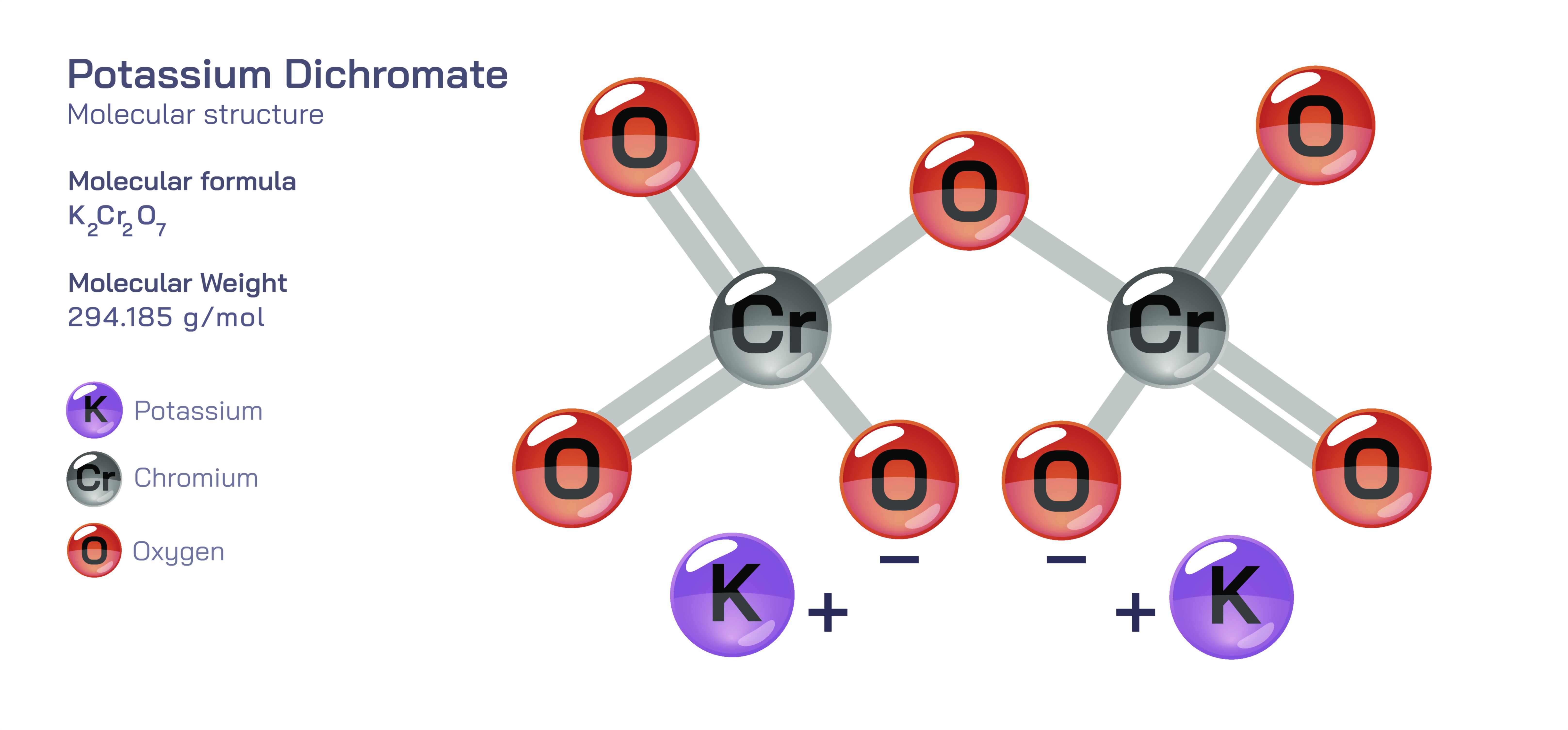
Potassium dichromate is a highly distinctive inorganic compound, known for its deep orange crystalline appearance and strong oxidizing behaviour, and its molecular structure is the core reason behind its physical properties, reactivity, laboratory uses, and safety concerns. The compound is made of potassium ions and dichromate ions held together in an ionic lattice, but the real complexity lies inside the dichromate ion itself. The dichromate ion contains two chromium atoms connected through a shared oxygen atom and surrounded by additional oxygen atoms arranged in a specific geometry that stabilizes chromium in a very high oxidation state. Inside the ion, each chromium atom exists in the +6 oxidation state, which means the chromium strongly attracts electrons from other substances it reacts with. This oxidation state not only gives the compound its vivid orange colour but also governs the aggressive reactivity that has made potassium dichromate useful in industrial processes, chemical analysis, and laboratory techniques. Despite its high reactivity, the internal structure remains stable and does not break apart easily; rather, it behaves predictably under controlled conditions, maintaining its dichromate identity until the environment alters the balance between dichromate and chromate species.
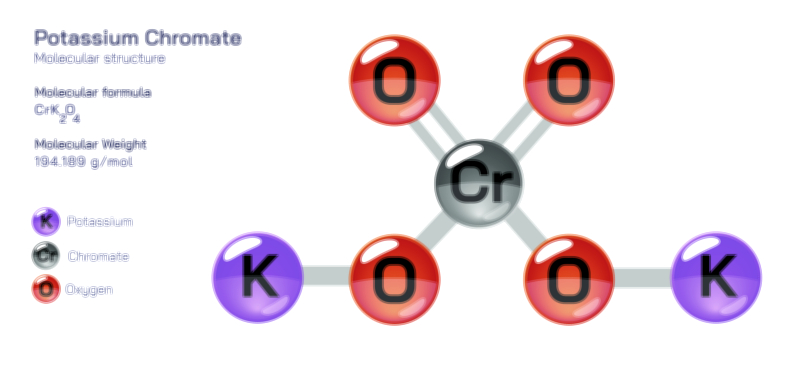
The dichromate molecular structure is best understood as two tetrahedrally coordinated chromate units linked together by a shared oxygen atom. Each chromium atom is surrounded by four oxygen atoms, three of which remain terminal while the fourth forms the bridge connecting the two chromium centres. This CrŌĆōOŌĆōCr bridging arrangement gives the dichromate ion its characteristic bent geometry rather than a linear form and allows the electron clouds around both chromium atoms to interact through oxygen, distributing charge across the ion in a stable but reactive pattern. The potassium ions do not share electrons directly with the dichromate unit; instead, they balance its negative charge electrostatically within the crystal lattice. When dissolved in water, the potassium ions separate and allow the dichromate ion to stay intact, although the balance between dichromate and chromate shifts depending on the acidity of the solution. In acidic conditions, orange dichromate ions are dominant, while in alkaline or basic conditions, yellow chromate ions become more abundant, showing a visual transformation that directly reflects molecular rearrangement. This reversible interaction between dichromate and chromate is one of the most important aspects of chromium chemistry and is widely used in analytical techniques to track pH changes, oxidationŌĆōreduction reactions, and equilibrium systems in laboratories and academic settings.
The strong oxidizing ability of potassium dichromate comes from the high oxidation state of chromium and the way oxygen atoms are arranged around it. Chromium(VI) inside the dichromate ion is energetically inclined to gain electrons and be reduced to a lower oxidation state. When the compound encounters materials that can donate electrons, such as organic compounds, reducing agents, metals, or biological molecules, it readily oxidizes them while chromium itself is reduced, often to chromium(III). This change is accompanied by a colour transformation from orange to green, which is why potassium dichromate has been used as a visual indicator of oxidation in chemical education. The structural need of chromium to achieve electron stability is what drives these reactions, and this inherent tendency explains why the compound has been historically valued in applications such as cleaning laboratory glassware, metal surface treatment, photographic processing, wood preservation, colour pigments, and organic oxidation reactions. However, this same molecular power is also the reason the compound must be handled with extreme caution, because the oxidizing strength that is beneficial in controlled chemical settings becomes hazardous in uncontrolled biological environments.
Safety concerns surrounding potassium dichromate are directly linked to its molecular structure and the chromium(VI) oxidation state. In biological tissues, the dichromate ion is not simply inert; it interacts aggressively with proteins, DNA, and cell membranes because it acts as a strong electron acceptor. When chromium(VI) enters a living system, it is reduced stepwise inside cells, and these reduction steps generate reactive intermediates that damage cellular components. The chromium that results after reduction may bind to proteins and nucleic acids, disrupting normal functions and triggering oxidative stress. For this reason, potassium dichromate is considered toxic, corrosive, carcinogenic, and environmentally hazardous. Even short-term exposure can cause severe irritation to the eyes, skin, and respiratory system, and long-term exposureŌĆöespecially through inhalation of dust or aerosolsŌĆömay lead to chronic health effects. In laboratories or industrial environments, the compound must be kept away from the skin and respiratory pathways, requiring protective gloves, eye protection, lab coats, fume hoods, and strict handling protocols. Its oxidizing properties also mean it should never be stored near flammable or easily oxidizable materials, reducing agents, organic fuels, or acids that could trigger violent chemical reactions.
Potassium dichromate also has environmental implications because chromium(VI) compounds are toxic to plants, aquatic organisms, and soil microorganisms. If released into the environment without proper treatment, the compound can contaminate water systems and enter biological cycles where it accumulates rather than dissipates. Modern industrial safety regulations therefore require careful disposal and treatment that converts chromium(VI) to the less harmful chromium(III) state before release. The reduction from dichromate to chromium(III) renders the chromium significantly less reactive and far less likely to cause oxidative damage in living systems, though it still must be managed responsibly. Many countries now restrict or prohibit the use of potassium dichromate in consumer products and wood preservatives and encourage safer alternatives in manufacturing and metal finishing. However, the compound remains in use in specific industries and scientific applications where its particular oxidizing structure cannot be replaced without compromising precision or effectiveness.
The molecular structure of potassium dichromate therefore defines both its utility and its hazard. Its crystalline ionic lattice explains its stability in solid form and its solubility in water. The tetrahedral oxygen coordination around chromium explains both its vivid colour and its strong oxidizing power. The bridging oxygen between chromium atoms explains its equilibrium relationship with chromate species and its role in analytical chemistry. And the high oxidation state of chromium explains why it can be dangerous to biological systems and requires serious safety measures wherever it is handled. Understanding the structure of potassium dichromate provides clarity about why it behaves the way it does in chemical systems and why its benefits in industrial and laboratory applications must be balanced with responsible safety procedures to protect health and the environment.