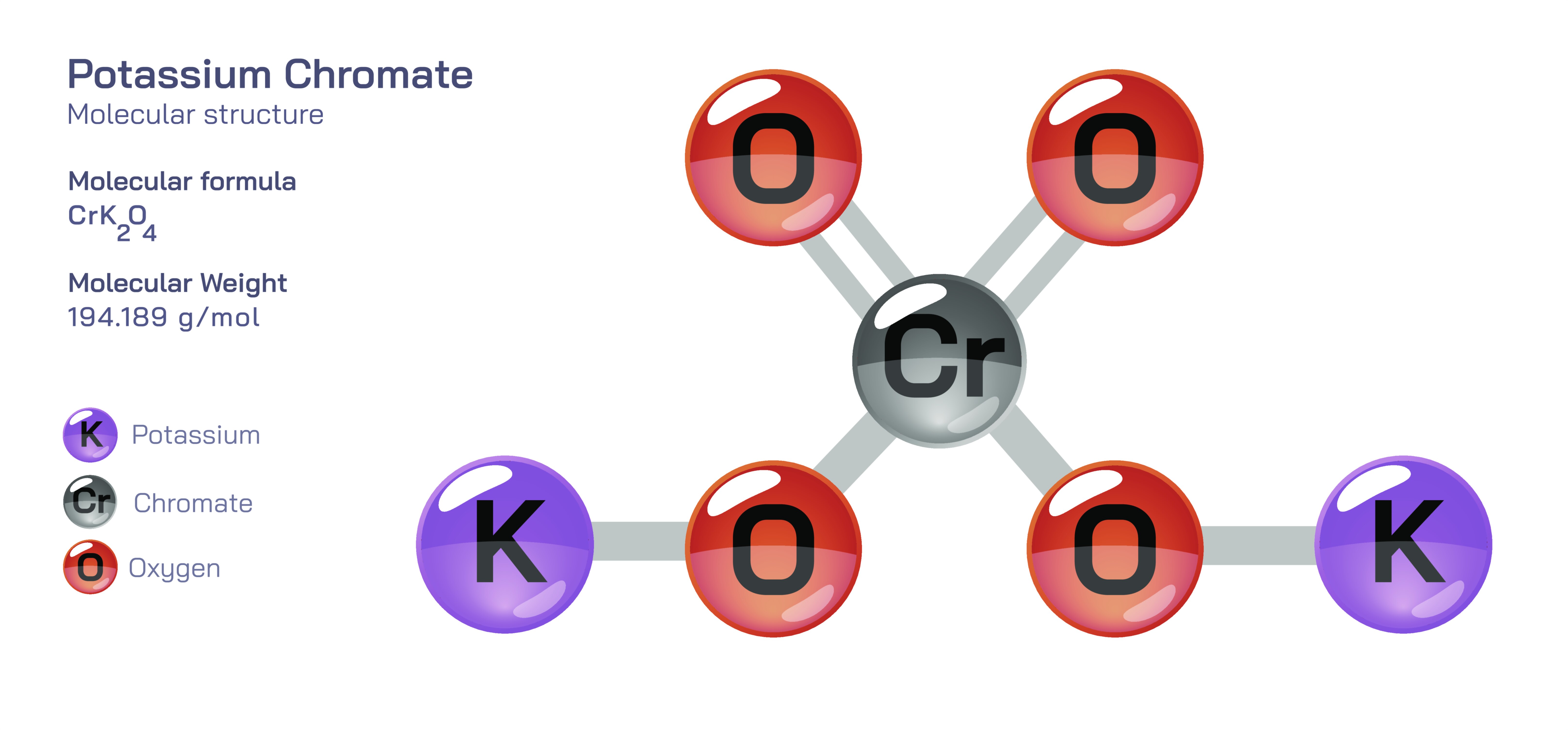
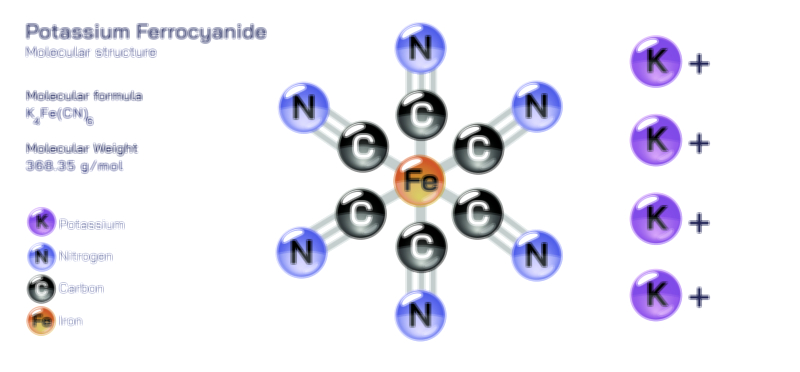
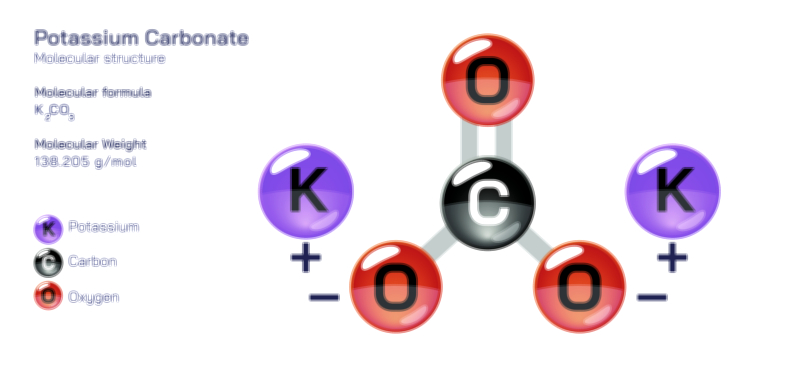
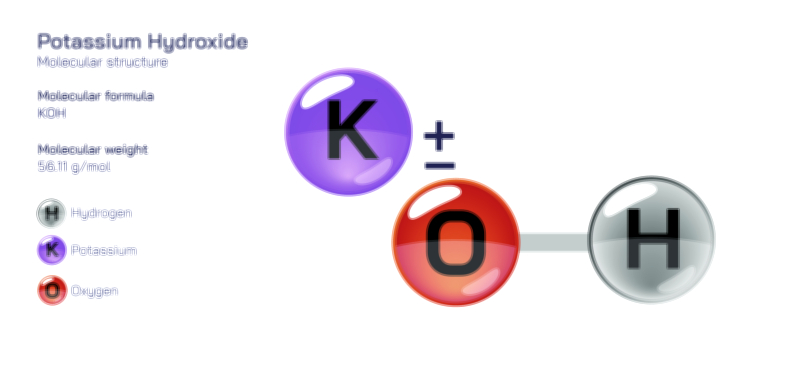
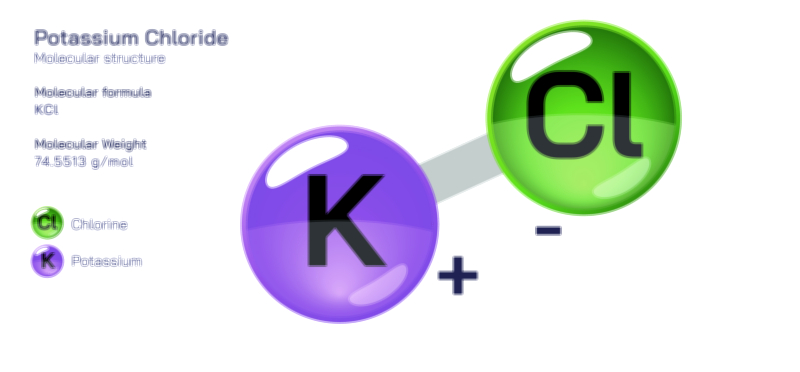
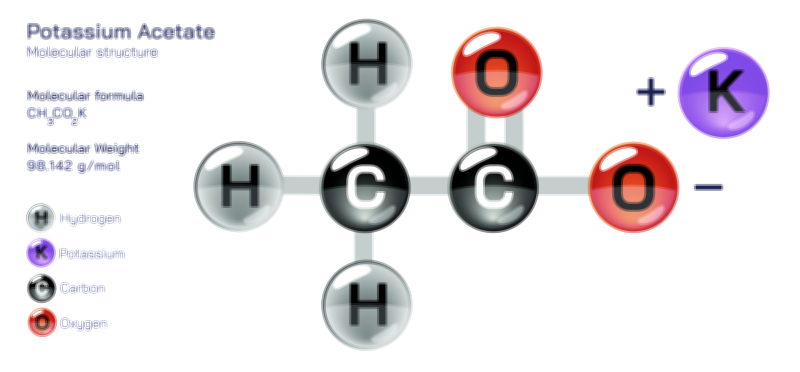
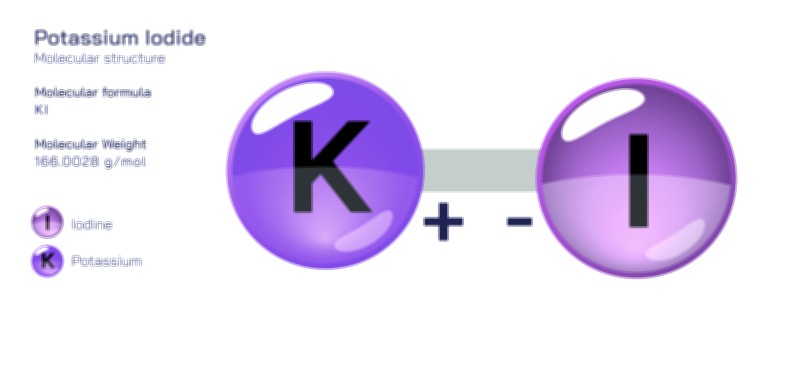
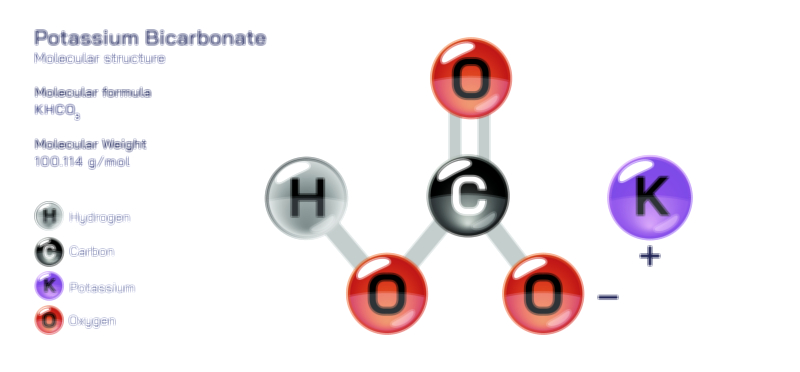
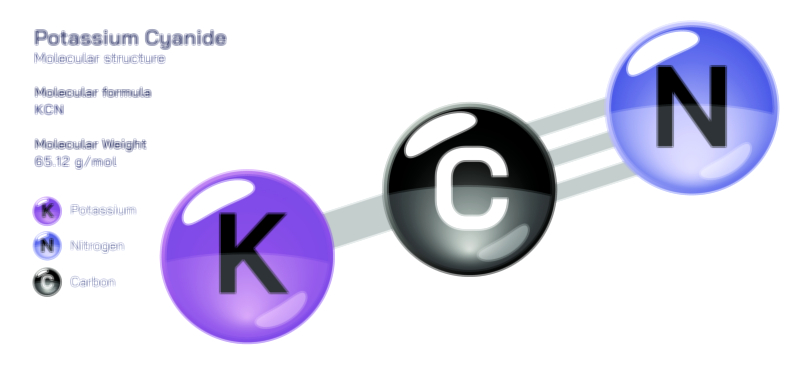
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐂𝐡𝐫𝐨𝐦𝐚𝐭𝐞 𝐂𝐡𝐞𝐦𝐢𝐜𝐚𝐥 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞
Potassium chromate is an important inorganic compound recognized for its bright yellow appearance, strong oxidizing behaviour, and extensive use in analytical chemistry, manufacturing, and industrial processes. Its structure is based on an ionic arrangement consisting of potassium ions and chromate ions held together through electrostatic forces. The chromate ion itself is the defining feature of the compound and contains a central chromium atom surrounded by oxygen atoms in a geometrically balanced configuration that gives the molecule both structural rigidity and strong chemical reactivity. The chromium present in this structure exists in a high oxidation state, and this oxidation state is directly responsible for the distinctive bright yellow colour commonly associated with chromate salts. In solid form, potassium chromate crystallizes in a structure where potassium ions occupy positions between chromate groups, forming a stable three–dimensional lattice that dissolves easily in water and produces a highly alkaline and intensely coloured solution.
Microscopically, the arrangement of potassium chromate reveals how ionic separation in water contributes to its behaviour. When dissolved, it separates completely into free potassium ions and chromate ions. The chromate ion remains strongly bound internally due to the strength of chromium–oxygen coordination, but externally it is free to interact with surrounding ions, metals, and chemical reagents. Because of this structural configuration, potassium chromate becomes a highly efficient indicator and reactive agent in tests designed to identify specific metal ions, especially silver, barium, and lead. Its bright colour makes even small amounts of it visible to the eye, and in analytical chemistry this vivid colour change property is applied in titration reactions and laboratory detection procedures.
The structure of potassium chromate also demonstrates the close relationship it shares with potassium dichromate. Both compounds contain chromium in the same oxidation state, and in solutions they can interconvert depending on the acidity of the environment. Under alkaline conditions, the chromate form remains dominant, displaying a yellow colour. In acidic environments, the structure shifts toward the dichromate form, producing an orange solution. This reversible structural transformation highlights the sensitivity of chromium–oxygen clusters to their surrounding chemical environment and illustrates how the chromate structure is not simply fixed but responsive to pH conditions, making it an important subject in equilibrium chemistry and industrial processing.
Despite its usefulness, potassium chromate must always be handled with strict care. The same structural features that give chromium high oxidation power also make chromates toxic to living organisms and hazardous to health when inhaled, ingested, or absorbed through the skin. The compound can have serious effects on cells and tissues because chromium in this oxidation state interacts aggressively with biological molecules. Due to these risks, protective handling, controlled laboratory procedures, and safe disposal practices are essential wherever potassium chromate is used. Many workplaces and industrial sectors have shifted toward safer alternatives when possible, but potassium chromate continues to remain indispensable in certain specialized scientific and industrial contexts due to the precision, stability, and reactivity that arise directly from its chemical structure.
Potassium chromate stands as a strong example of how the microscopic structure of a compound determines its macroscopic behaviour. The ionic lattice, the chromium–oxygen coordination, and the reversible equilibrium with dichromate form all work together to create a substance that is both scientifically valuable and chemically powerful. Its structure explains its vivid colour, its selective reactivity, its analytical capabilities, its industrial relevance, and its toxicity. Understanding the chemical structure of potassium chromate allows chemists and engineers to harness its benefits safely and selectively, demonstrating how even the most striking and hazardous materials can hold high importance in the world of chemistry when handled with knowledge and care.