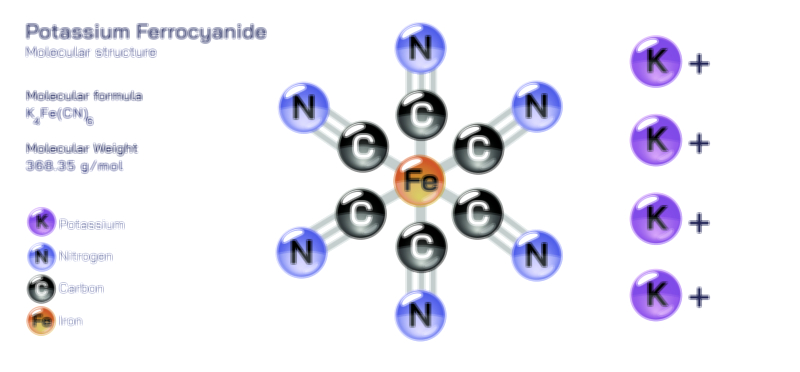
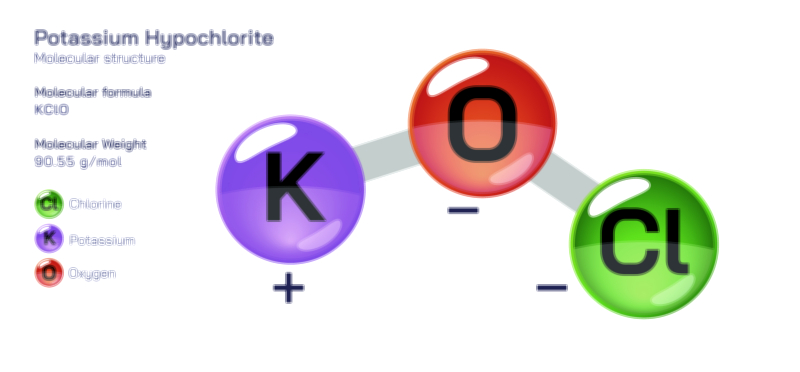
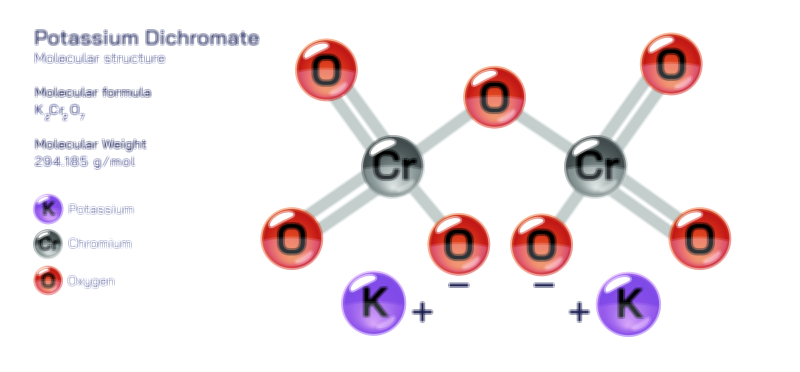
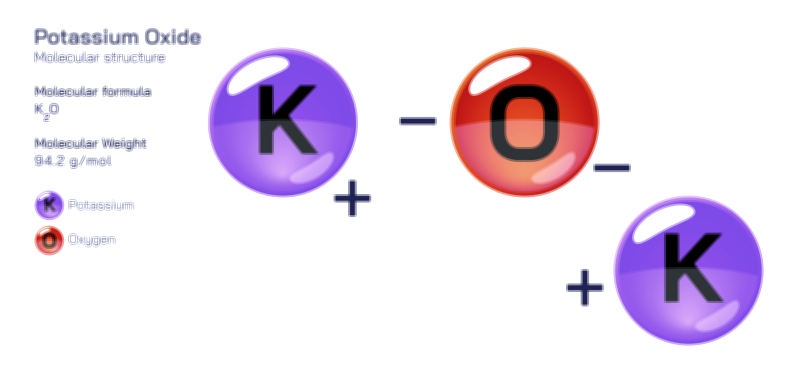ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉĆØÉ£ØÉ×ØÉŁØÉÜØÉŁØÉ× ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉÜØɦØÉØ ØÉöØɼØÉ×Øɼ
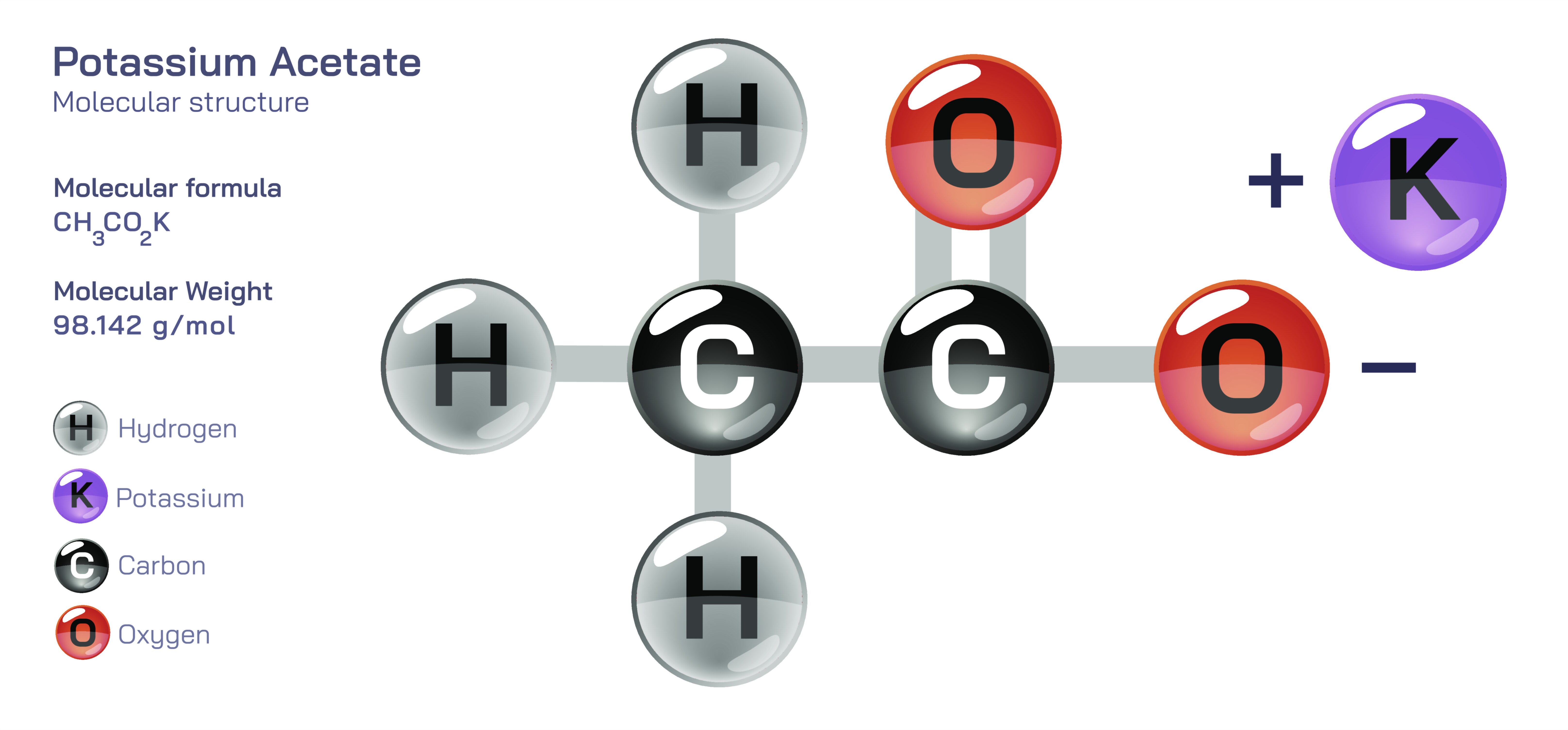
Potassium acetate is an important inorganicŌĆōorganic salt widely used in science, medicine, food processing, laboratory chemistry, and industry due to its balanced reactivity, high solubility, and biological compatibility. Chemically, potassium acetate is produced when acetic acid reacts with potassium hydroxide, forming a salt composed of potassium ions and acetate ions held together through ionic attraction. Its structure consists of a positively charged potassium ion (KŌü║) and a negatively charged acetate ion, which contains a carbon backbone bonded to oxygen atoms arranged in a way that gives the compound stability and solubility. The acetate component is derived from acetic acid, the same acid responsible for the characteristic sourness of vinegar, and it brings into the molecule organic flexibility and compatibility with biological systems, while the potassium component supplies an electrolyte quality that interacts well with bodily fluids, hydration mechanisms, and plant metabolic functions. In solid form, potassium acetate appears as white crystalline granules or a fine powder that dissolves readily in water and alcohol, producing a clear, neutral-to-slightly alkaline solution. This combination of structural simplicity and functional versatility allows potassium acetate to play a significant role in preserving food, de-icing roads, supporting biochemical reactions, stabilizing medical formulations, and controlling pH in sensitive environments.
The structural behaviour of potassium acetate demonstrates how small changes in molecular arrangement can produce wide-ranging effects in practical applications. The acetate ion acts as a buffering agent, meaning it can help maintain a stable pH in many chemical and biological systems. This ability comes from the fact that acetate is weakly basic and can either accept or donate protons depending on the environment, preventing sudden swings in acidity. The potassium ion, meanwhile, supports ionic conductivity, hydration balance, and mineral supplementation when used in biological or nutritional formulations. The high solubility of potassium acetate in water is one of its most valuable characteristics because it distributes evenly without leaving residue, making it ideal for liquid-based systems such as intravenous solutions, dialysis fluids, laboratory reagents, and de-icing sprays. Unlike salts that release chloride ions and contribute to corrosion, potassium acetate is considered significantly less damaging to metal surfaces, concrete structures, soil composition, and the environment, which explains its growing use as a replacement for calcium chloride and sodium chloride in winter road maintenance and aviation runway de-icing solutions. The compound also has a relatively low freezing point, and solutions made from it lower the freezing temperature of water considerably, preventing ice buildup and making roadways safer in cold climates without causing the same level of chemical damage typically associated with traditional road salts.
In the medical and pharmaceutical fields, potassium acetate has long been valued for its electrolyte-supporting and buffering properties. It is included in intravenous fluids to help restore potassium levels in patients experiencing electrolyte depletion due to illness, dehydration, metabolic disorders, or prolonged medical treatment. Because potassium is vital for nerve conduction, muscle contraction, cardiac rhythm regulation, and cellular energy production, medical-grade potassium acetate provides a controlled and predictable method for supplementing the mineral safely. It is also used in dialysis formulations to balance electrolyte concentrations while removing waste from the blood in individuals with kidney dysfunction. In emergency treatment for metabolic acidosis, potassium acetate can be administered to help counteract an excessively acidic internal environment by supporting the bodyŌĆÖs buffering systems. Its compatibility with the human body and its predictable physiological effect allow physicians and pharmacists to use it confidently in a variety of therapeutic contexts. Beyond intravenous solutions, potassium acetate is also employed in preserving and stabilizing medical specimens, laboratory media, and biochemical assays.
Food and agricultural industries make use of potassium acetate for its non-toxic, flavour-neutral, and antimicrobial characteristics. It functions as a preservative and acidity regulator in packaged foods, especially in meat-based and processed foods where controlling microbial growth is critical for safety and shelf life. Because acetate interferes with the metabolic processes of certain bacteria without contributing harmful toxicity, small amounts of potassium acetate help inhibit spoilage and maintain freshness. It also helps stabilize pH and reduce water activity in food systems, limiting conditions that allow bacteria to multiply. In agriculture, potassium acetate serves as a highly efficient source of potassium in liquid fertilizers. Plants absorb acetate-based potassium more readily during periods of drought or low soil moisture compared to conventional potassium chloride fertilizers, because it reduces osmotic stress and mitigates salt shock. This makes potassium acetate valuable not only in standard crop nutrition programs but also in precision agriculture for improving nutrient uptake and plant resilience.
In chemical laboratories and industrial processing, potassium acetate has a wide footprint due to its role as a buffering compound, a catalyst supporter, and a solvent-enhancing agent. Analytical chemists use potassium acetate to prepare buffer solutions that preserve reaction stability, control ionic strength, and support the functioning of enzymes during biochemical experiments. It is often chosen when chloride ions must be avoidedŌĆösuch as in the testing of sensitive metal components or in preparing solutions for chemical reactions where halogens interfere with desired pathways. In industrial manufacturing, potassium acetate is used as a polymer additive, textile-printing chemical, corrosion inhibitor, and fuel additive. It contributes to improving the efficiency of polyester production, enhancing dye absorption in fabrics, and increasing stability in oil-based lubricants. Its predictable thermal and ionic properties help manufacturers control product quality in large-scale production.
Another well-known application of potassium acetate lies in portable heat packs. When a supersaturated solution of potassium acetate is sealed inside a flexible plastic container, it remains liquid until triggered by a metal disc or bending action. Once activated, the salt crystallizes rapidly, releasing stored heat in an exothermic reaction and producing warmth for an extended period. This heating mechanism is safe, reusable, and non-toxic, and it demonstrates how phase transitions of chemical salts can become practical tools for everyday life. After the heat pack cools, reheating it dissolves the crystals again, returning the system to its supersaturated liquid state and preparing it for reuse. This simple mechanism highlights how the molecular behaviour of potassium acetate translates into user-friendly thermal technology.
Potassium acetateŌĆÖs environmental profile is another reason for its increasing demand across sectors. Because it contains no chlorides and breaks down naturally into substances that do not persist or bioaccumulate, it is regarded as a more environmentally responsible option in several applicationsŌĆöespecially in winter road safety and de-icing where large quantities of chemicals enter ecosystems. It has significantly lower corrosive impact on bridges, airport runways, vehicles, concrete surfaces, and underground utilities compared to traditional salts. This results in longer infrastructure life and reduced maintenance costs, allowing cities and airports to maintain safety without compromising structural longevity.
Across medicine, food science, agriculture, chemical engineering, and infrastructure safety, potassium acetate demonstrates how one compound can become valuable in completely different environments based on the unique interaction of structure and function. The dual nature of its compositionŌĆöan essential mineral cation paired with an organic buffering anionŌĆögives it a versatility that few simple salts achieve. Its high solubility supports its use in medicine and nutrition, its non-corrosive de-icing properties protect public infrastructure, its antimicrobial behaviour enhances food preservation, its buffering capacity stabilizes laboratory reactions, and its crystallization heat release supports reusable heating systems. The ongoing improvement of processing technologies and sustainability efforts ensures that potassium acetate continues to expand into new fields of science and industry. Although nearly invisible in everyday life, potassium acetate quietly supports safety, comfort, nutrition, agriculture, healthcare, and technology, illustrating the silent yet profound influence that chemical materials have on modern living.