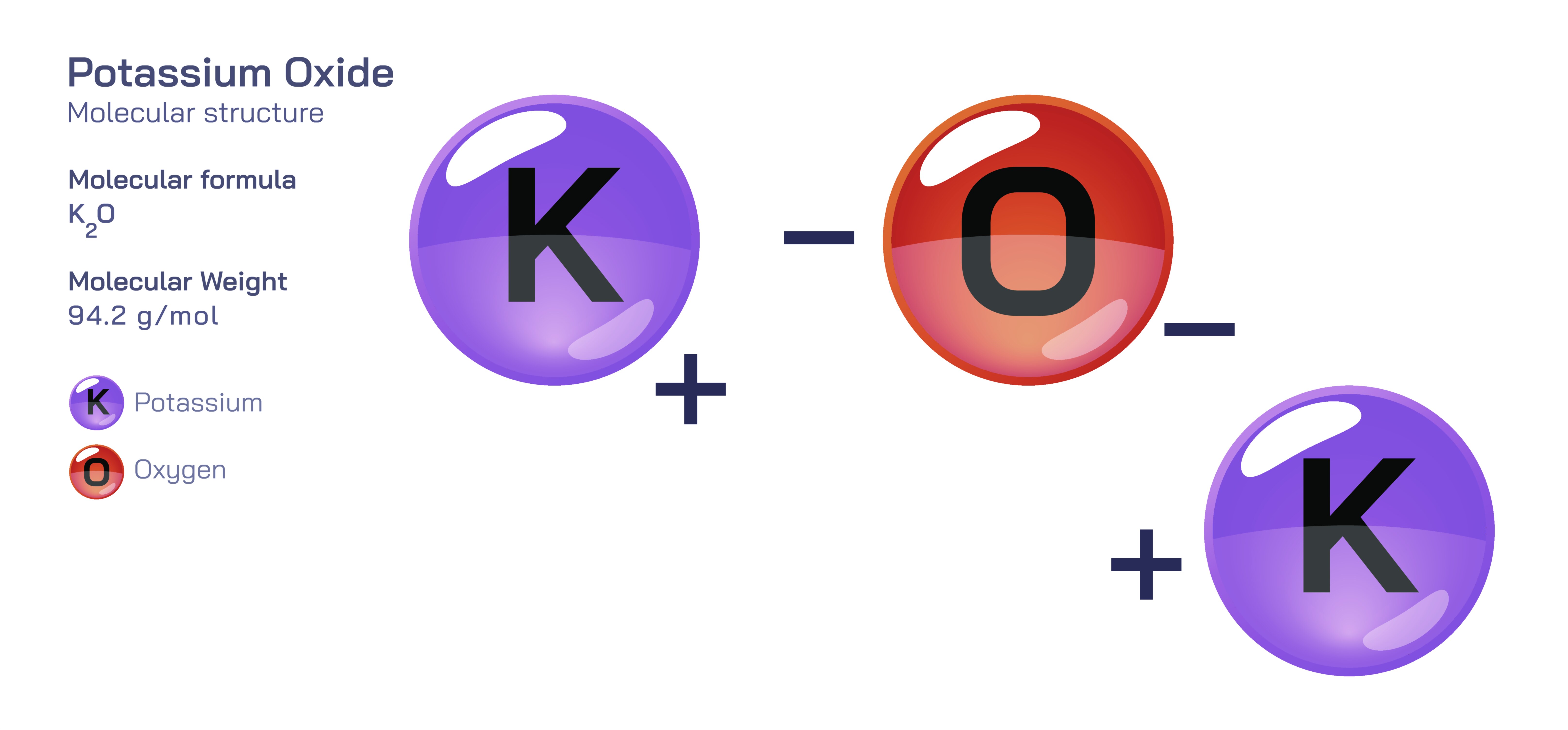𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐎𝐱𝐢𝐝𝐞 (𝐊₂𝐎) 𝐌𝐨𝐥𝐞𝐜𝐮𝐥𝐚𝐫 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞 𝐚𝐧𝐝 𝐂𝐡𝐞𝐦𝐢𝐜𝐚𝐥 𝐏𝐫𝐨𝐩𝐞𝐫𝐭𝐢𝐞𝐬.
Potassium oxide is an inorganic compound whose identity arises from a strong ionic interaction between potassium ions and oxide ions, forming a highly reactive alkaline oxide that demonstrates powerful chemical basicity and strong oxygen affinity. At the structural level, potassium oxide appears deceptively simple: it contains two potassium atoms bonded with one oxygen atom, expressed through the empirical formula K₂O. However, the actual structural story becomes meaningful when viewed through electron transfer and solid-state arrangement. Each potassium atom donates one electron to the oxygen atom, converting the neutral potassium atoms into K⁺ ions and reducing the oxygen atom to an O²⁻ oxide ion. This full electron transfer generates a purely ionic attraction between the potassium ions and the oxide ion. Within the solid lattice, these ions arrange themselves into a repeating crystalline framework in which the oxide ion is surrounded by potassium ions positioned symmetrically to neutralize the charge. This electrostatic structure produces a strong, rigid, salt-like solid that remains stable only under dry conditions. The lattice does not rely on directional covalent forces but rather on the uniform electrostatic pull between oppositely charged ions. Every structural property of potassium oxide in its solid form—its high melting point, brittleness, and strong affinity for humidity—arises directly from this ionic architecture.
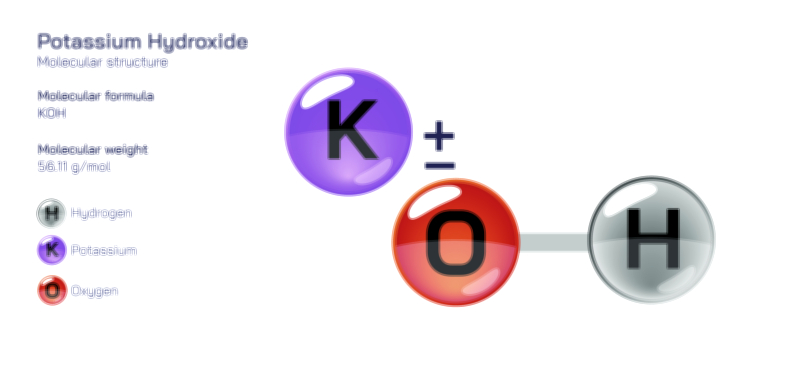
The structure of potassium oxide gains practical meaning only when its reactivity in the presence of water is considered. Because the oxide ion is an extremely strong base with a powerful attraction toward protons, it cannot remain stable in the presence of water. When potassium oxide encounters moisture, the oxide ion rapidly reacts with water molecules, breaking the hydrogen–oxygen bonds in water and producing hydroxide ions. This reaction turns potassium oxide into potassium hydroxide, a transformation that occurs with the release of significant heat because the resulting hydroxide ions are much more stable in aqueous form than the original oxide ions were in the solid state. This transition is not an incidental side reaction but instead an unavoidable chemical fate for potassium oxide in ambient conditions. It demonstrates that the compound exists structurally not to remain a static mineral in the environment but to behave as a precursor to strong alkaline solutions. The oxide ion itself does not persist in water because its charge density and proton affinity lead it to transform immediately into hydroxide. Therefore, potassium oxide is best understood structurally as the anhydrous form of potassium hydroxide, and its existence is meaningful primarily wherever water is absent. As soon as it dissolves, the molecular identity shifts from oxide-based structure to hydroxide-based chemistry.
In molten or high-temperature environments, potassium oxide exhibits a structure suited for oxygen mobility. The oxide ion can act as an oxygen donor, a characteristic that allows potassium oxide to participate in processes that require oxygen transfer. In high-temperature metallurgical applications, the compound reacts with acidic oxides and impurities in molten materials, forming stable salts that separate from the metal. This behaviour arises from the oxide ion’s tendency to neutralize oxide species with high acidity, enabling slag formation and purification processes. On the other hand, potassium oxide does not act like elemental oxygen, nor does it support combustion. It does not serve as an oxidizing agent in organic combustion because the oxygen atom is held in a reduced state, and its release is not energetically favourable unless a highly acidic oxide reacts with it. However, it readily supports the formation of silicates and aluminates in high-temperature ceramics and glass manufacturing. In these environments, potassium oxide functions as a flux agent, lowering melting temperatures and modifying the physical structure of glass by replacing network-forming oxide units with network-modifying potassium ions. Through this structural role, potassium oxide improves thermal expansion control, transparency, and mechanical strength of industrial glass compositions, demonstrating how its ionic nature influences macroscopic material performance.
The structure of potassium oxide also determines its chemical compatibility beyond water. Because the oxide ion is highly basic, the compound reacts vigorously with acids to produce potassium salts. In these reactions, the oxide ion captures protons from the acid and becomes water, while the anion from the acid bonds with potassium to form a salt such as potassium sulfate, potassium carbonate, or potassium chloride depending on the acid involved. These acid–base interactions are not reversible because the formation of water from oxide and protons eliminates the oxide structure permanently. Therefore, in any environment where proton sources exist, potassium oxide cannot maintain its identity. The compound also reacts with carbon dioxide to produce potassium carbonate by converting the oxide ion into carbonate through a structural association of oxygen with carbon dioxide. This transformation demonstrates that potassium oxide is not stable in air over prolonged periods if atmospheric carbon dioxide is present and humidity allows mobility. It reflects a recurring theme in potassium oxide chemistry: the oxide ion is a starting point from which other potassium compounds develop based on environmental contact.
In industrial contexts, the structural reactivity of potassium oxide is so powerful that the compound itself is rarely handled directly except under controlled conditions. However, its chemical significance remains enormous because it serves as a theoretical benchmark for potassium content in fertilizers. Even though potassium oxide itself is not applied to crops, fertilizer compositions are scientifically described using potassium oxide equivalence to express the amount of potassium available for plant nutrition. The K₂O unit, as a structural reference, simplifies nutrient measurement and standardization because it represents the theoretical oxide-based form of potassium. In agriculture, potassium oxide does not act as a fertilizer ingredient directly; instead, potassium salts such as potassium chloride, potassium nitrate, or potassium sulfate supply potassium to the soil. The structural use of K₂O arises from standardization rather than application, proving that its identity has scientific value even when not physically present.
The structural features of potassium oxide also determine its behaviour in laboratory synthesis and materials science. As a precursor compound, it establish a ready route to potassium hydroxide, potassium carbonates, and other potassium salts. When chemists require a highly reactive potassium compound in nonaqueous synthesis, potassium oxide becomes a strong base and oxygen carrier. Because of its ionic simplicity and the discreet presence of K⁺ and O²⁻ ions, it allows control over reactivity without introducing unwanted byproducts associated with carbonate, nitrate, or halide counterions. This explains its value in catalytic systems, ceramic engineering, refractory chemistry, and specialized catalyst supports.
Viewed holistically, potassium oxide is not just a formulaic combination of potassium and oxygen but a structural blueprint for one of the strongest basic oxide systems known in alkali chemistry. In the crystalline solid, the O²⁻ ion remains intact only because it is surrounded by potassium ions in a tightly packed electrostatic lattice. In the presence of water, the internal structure transforms rapidly into hydroxide because of the oxide ion’s overwhelming proton affinity. In molten systems, the oxide ion enables glass and ceramics to form through charge-balancing interactions between network-modifying and network-forming ions. In acid systems, the oxide ion is neutralized and converted into a salt. In air, the structure can convert into carbonate over time. Every transformation demonstrates how the structural identity of the oxide ion guides potassium oxide toward predictable chemical outcomes.
Potassium oxide is therefore a paradigm of structure-driven reactivity. Its ionic architecture explains its stability only in dry environments, its extreme basicity in the presence of water, its glass-modifying role in high-temperature materials, its neutralization behaviour in acid–base chemistry, and its conceptual use in agricultural science. It demonstrates that chemical behaviour does not arise from symbolic formulas but from the deeper reality of electron distribution and atomic architecture. The compound embodies the lesson that even the simplest ionic structure can generate wide-reaching effects across chemistry, industry and materials science when the internal arrangement of ions aligns with the laws of thermodynamics, electron transfer and structural transformation.