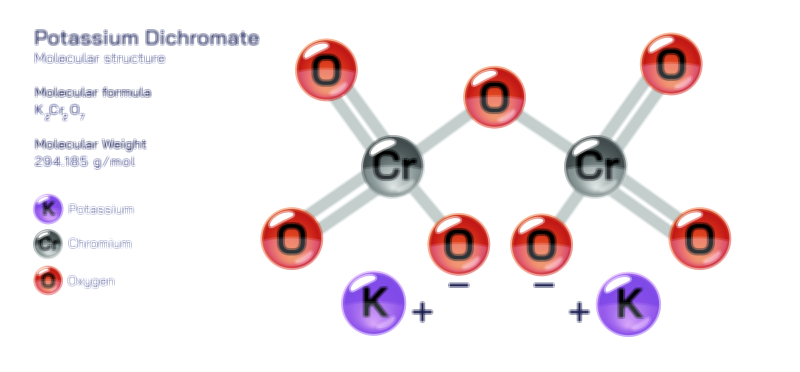
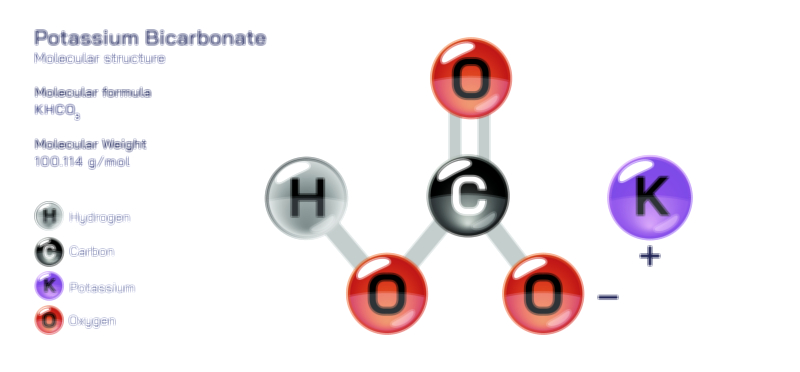
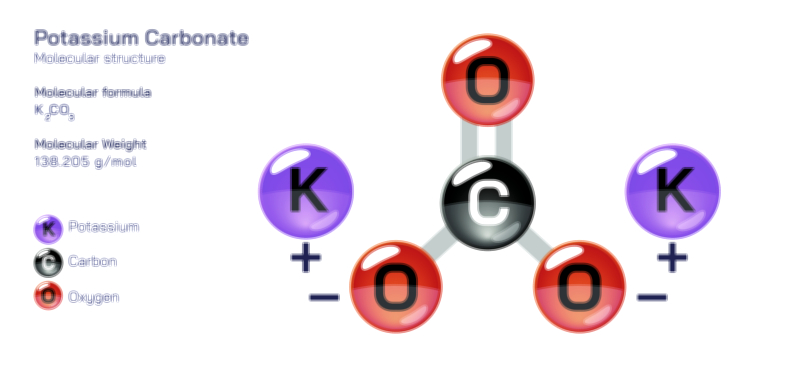
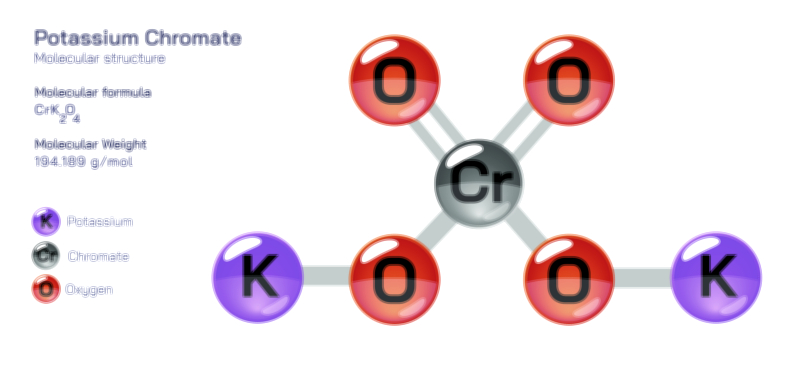
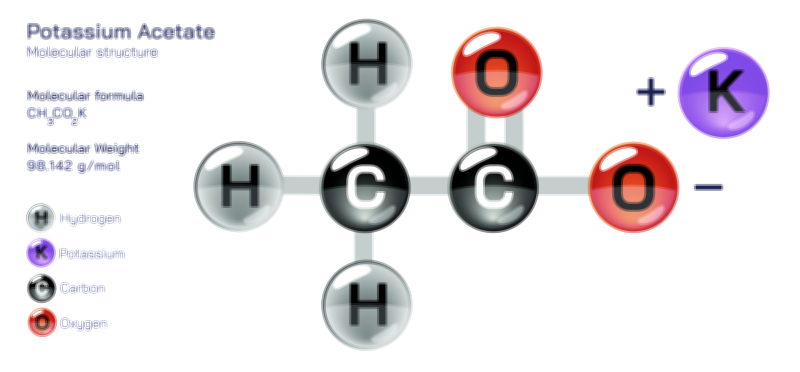
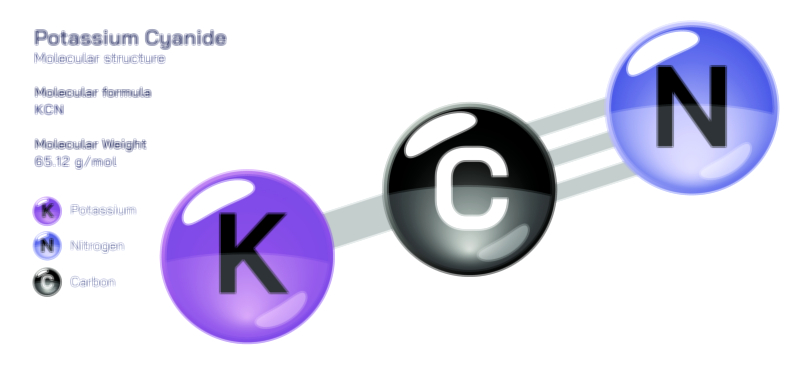
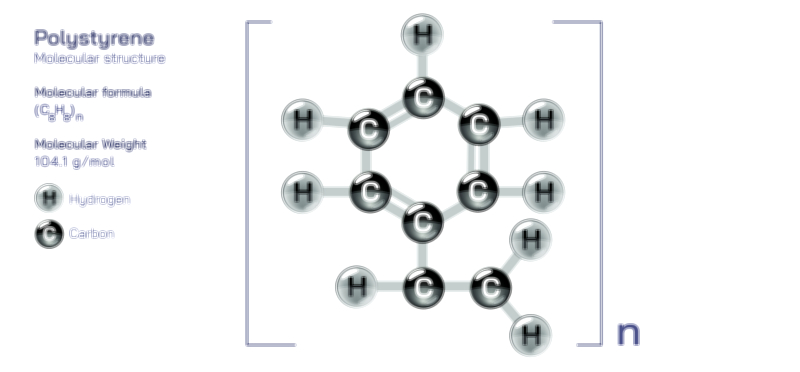
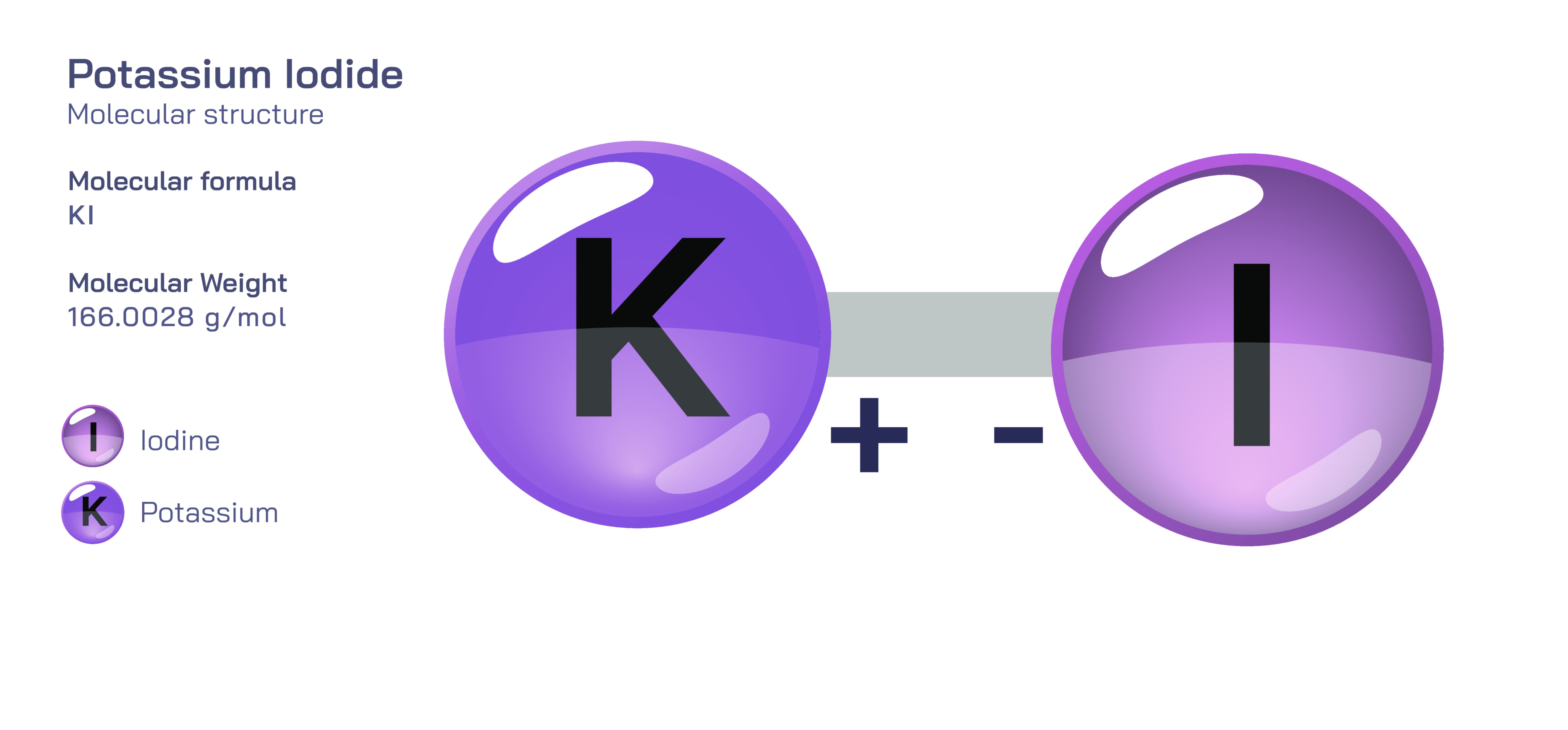ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉłØÉ©ØÉØØÉóØÉØØÉ× ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ŌĆö ØÉāØÉ×ØÉ×ØÉ® ØÉäØÉ▒ØÉ®ØÉźØÉÜØɦØÉÜØÉŁØÉ©ØɽØÉ▓ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓
Potassium iodide is an inorganic compound whose structural identity is grounded in its strong ionic bond and its highly ordered crystalline framework, yet beneath this apparent simplicity lies a molecular architecture that influences matter, chemistry, and biology in profound ways. The foundation of the compound is built on the transfer of one electron from a neutral potassium atom to a neutral iodine atom. When potassium loses its electron, it becomes a positively charged ion with a stable electronic configuration, and when iodine gains that electron, it becomes a negatively charged iodide ion whose outer electron shell becomes complete and energetically satisfied. The attraction between these two ions is not based on shared electrons as in covalent molecules but instead on pure electrostatic force, where opposite charges hold each other firmly in place in a repeating ionic lattice. In a single formula unit this ionic relationship appears simple, but the true structural complexity arises when millions of these ions arrange themselves into a three-dimensional crystalline network in which potassium and iodide ions alternate at mathematically precise intervals. This repeating geometry creates a stable, rigid structure that gives potassium iodide its salt-like appearance, relatively high melting point, and brittle physical behaviour. The cubic crystal lattice is symmetrical, and every potassium ion is surrounded by iodide ions while every iodide ion is surrounded by potassium ions. This fully packed arrangement minimizes repulsion between like charges and maximizes attraction between opposite charges, demonstrating the delicate physical logic embedded within the atomic architecture.
When potassium iodide transitions from the solid crystalline state into aqueous solution, the structural arrangement changes dramatically, but the identity of the ions remains unchanged. Water molecules are able to penetrate and disrupt the ionic lattice because they surround the ions during hydration and prevent them from returning to the crystal arrangement. Potassium ions disperse and remain strongly solvated, drifting through the liquid as stabilized cations that contribute to solution conductivity but otherwise play a passive chemical role. The iodide ion remains the centerpiece of the compoundŌĆÖs behaviour once in solution, and its structural simplicityŌĆöconsisting of a single iodine atom with a complete electron shellŌĆöprovides stability but also a unique capacity for reversible redox behaviour. The large size of the iodide ion and the loosely held outer electrons create a highly polarizable electron cloud, meaning its electron distribution can shift and deform in the presence of electric fields, organic molecules, heavy metals, and polar solvents. This polarizability allows iodide ions to form temporary complexes with organic molecules, to participate in substitution reactions in organic chemistry, and to become an essential participant in electron-transfer processes. The same structural characteristic is responsible for the tendency of the iodide ion to be oxidized back to elemental iodine in the presence of strong oxidizing agents, producing a visible brown or purple colour that not only represents a chemical transformation but also reveals the structural flexibility of the iodide ion under the right conditions.
This structural ability to cycle between the stable iodide state and the oxidized iodine state is particularly significant because it gives potassium iodide analytical, industrial, and biological relevance. In analytical chemistry, iodideŌĆÖs reversible redox behaviour forms the basis of iodometric titrations and starch-iodine tests, which rely on the visual transformation of iodide into molecular iodine and back again. These recognizable colour transitions are not merely aesthetic; they depict structural shifts in the electron distribution of iodine and reveal that the iodide ionŌĆÖs structural stability is balanced with responsiveness under specific conditions. In biological systems, the structural identity of the iodide ion becomes even more crucial. The iodide ion is selectively absorbed by the thyroid gland because its size and charge perfectly match the structure of iodine-processing proteins called sodium-iodide symporters. These proteins discriminate among ions with extraordinary precision, and the structural form of iodide allows it to pass through this selective biological channel while other halide ions such as chloride or bromide do not serve the same hormonal role. Once inside the thyroid gland, iodide is partially oxidized and incorporated into the structure of thyroid hormones. This biological mechanism depends completely on the iodide ion retaining its identity during transport, and it illustrates how a chemically simple ion can be essential to complicated physiological processes that regulate metabolism, growth, and neurological development.
The internal structure of potassium iodide also determines its behaviour beyond biological systems. In industrial applications, iodideŌĆÖs polarizability and redox capacity allow it to function as a catalyst component in certain organic transformations and as a stabilizing agent in photographic processing. Its ability to form stable yet reversible complexes with transition metals becomes important in materials processing, where iodide can prevent unwanted oxidation or support crystal growth. The shape and electron density of the iodide ion also influence its behaviour in solid-state mixtures. When potassium iodide crystals are exposed to ultraviolet light or ionizing radiation, electron displacement inside the iodide ion can create halogen defects and trapped electron sites, producing colour centres known as F-centres. These subtle structural interactions illustrate that even though the ionic lattice of potassium iodide appears visually uniform, at the atomic level it contains a dynamic interplay of charged particles and electronic transitions that scientists can study to understand radiation effects and crystal imperfections.
Despite these complex structural consequences, the potassium ion itself remains structurally stable and chemically restrained across all phases. Whether in the solid lattice, in molten form, or dissolved in solution, the potassium ion does not undergo redox changes and does not form covalent bonds with other atoms or groups. Its chemical role consists almost entirely of balancing the charge of the iodide ion and providing electrical neutrality to the lattice or the solution. This lack of reactivity is itself structurally meaningful because it allows the iodide ion to exhibit full chemical expression without interference from the cation, enabling potassium iodide to function as a clean iodide-delivery compound in medicine, agriculture, and nutrition. A compound that combines a highly stable ion with a chemically responsive ion results in a material that is both predictable and versatile, with the potassium ion providing structural foundation and the iodide ion providing chemical and biological activity.
Viewed through the lens of structureŌĆōfunction relationships, potassium iodide demonstrates how a compound that appears straightforward on paper embodies a highly significant molecular architecture. The ordered crystal lattice explains its physical stability and salt-like properties. The ionic dissociation in water explains its conductivity, solubility, and electrolyte behaviour. The polarizable electron cloud of the iodide ion explains its broad participation in organic chemistry, redox reactions, analytical testing, and biological hormone formation. The inertness of the potassium ion explains why the compound can deliver iodide safely without introducing additional reactivity. Together, these structural attributes form a molecular system that is both simple and powerful, proving that complexity in chemistry does not always arise from large molecular frameworks. Sometimes, the deepest scientific value originates from the precise balance of a single metal ion and a single non-metal ion whose structural and electronic complementarity supports applications ranging from human health and industrial chemistry to materials science and environmental processes.