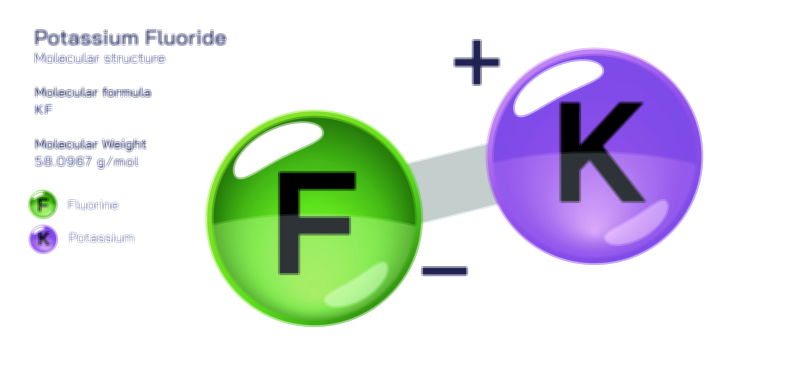
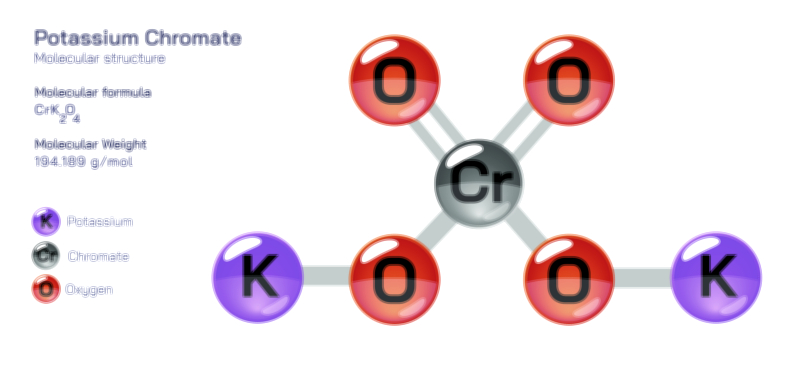
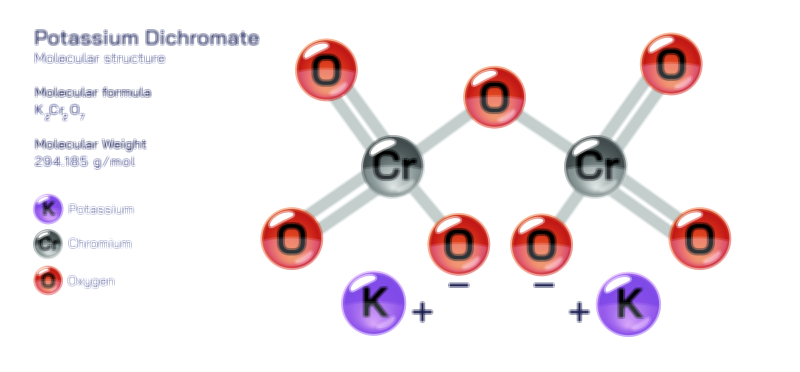
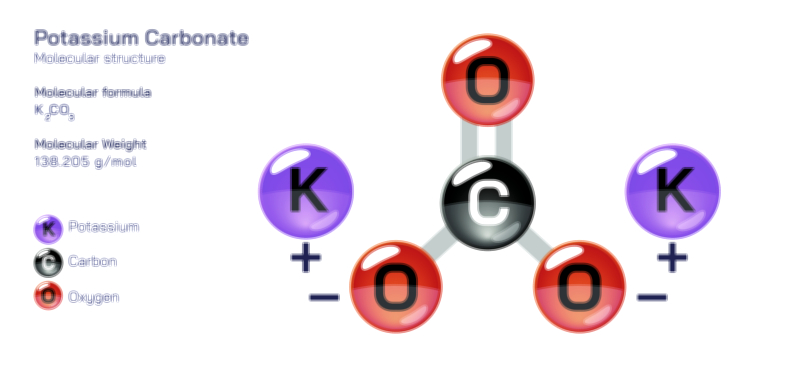
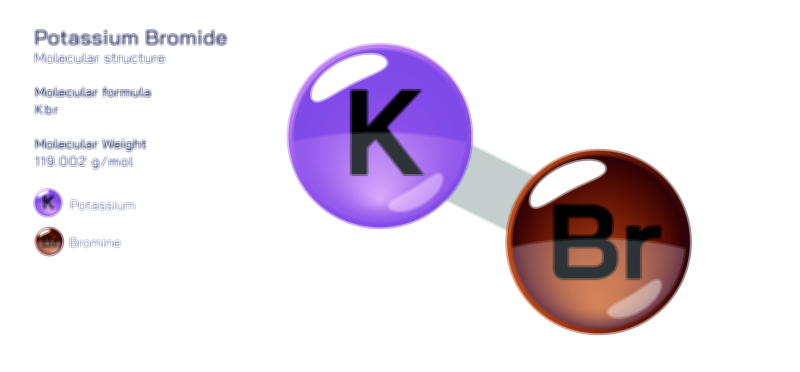
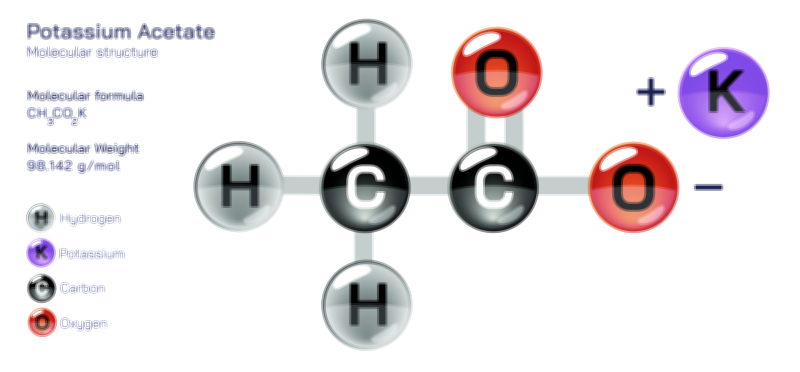
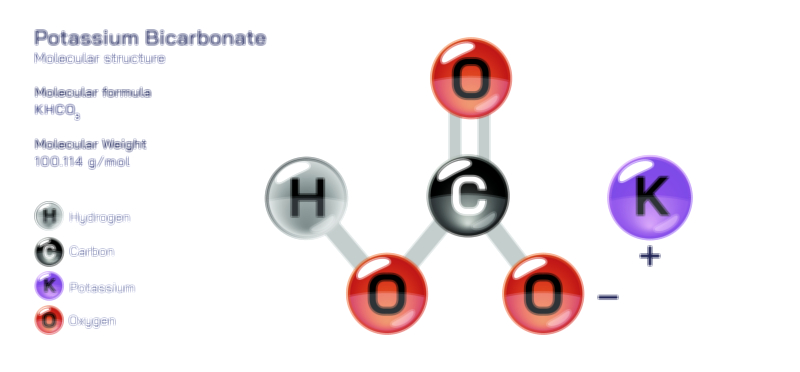
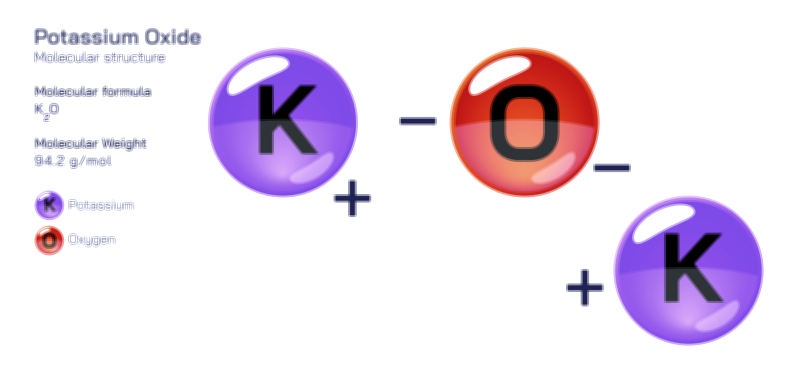ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉģØÉ×ØɽØɽØÉ©ØÉ£ØÉ▓ØÉÜØɦØÉóØÉØØÉ× ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×
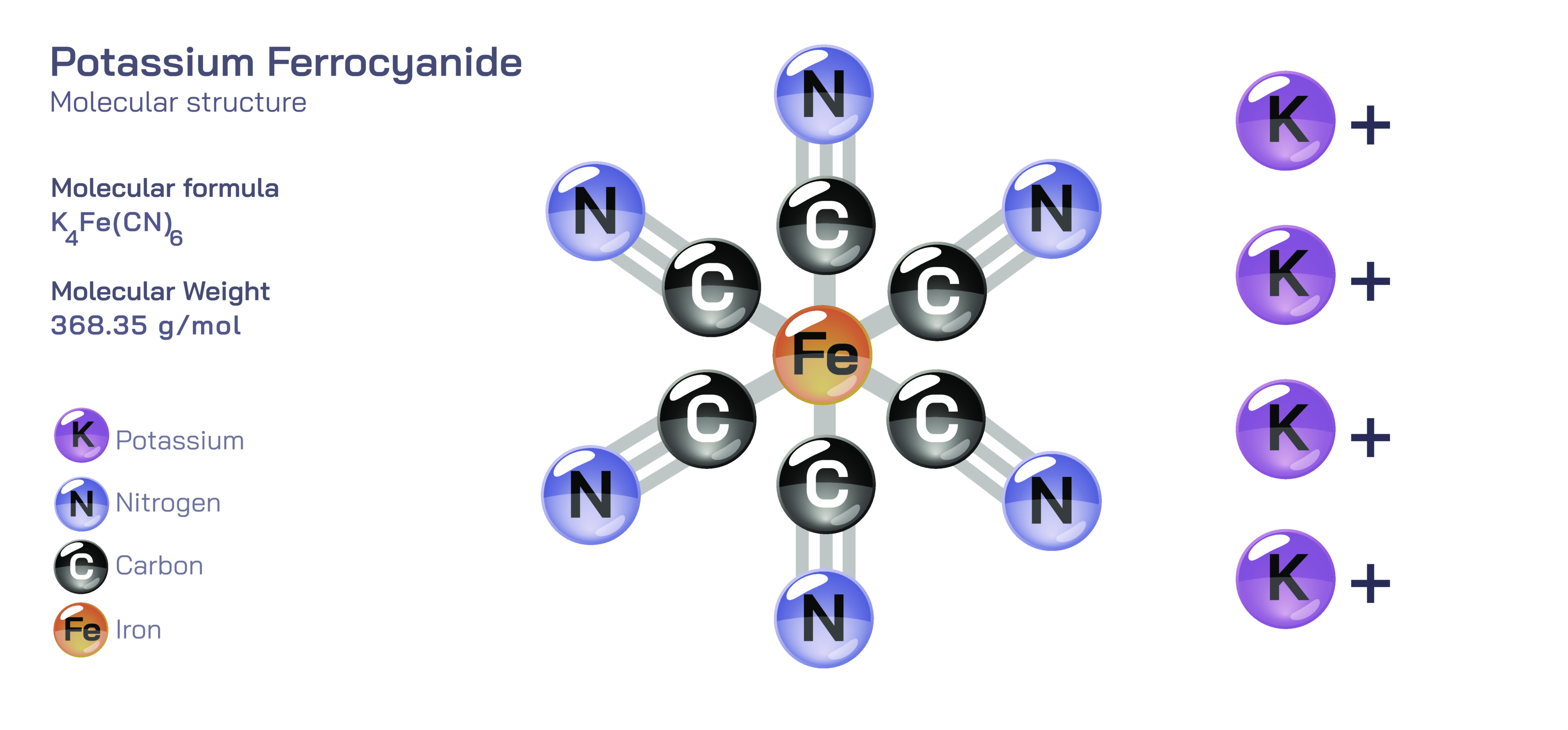
Potassium ferrocyanide possesses one of the most elegant and widely studied molecular architectures in inorganic and coordination chemistry, combining the simplicity of an ionic salt with the sophistication of a complex metalŌĆōligand cluster. At the centre of the molecular arrangement lies an iron atom that exists in the ferrous state, surrounded symmetrically by six cyanide ligands bonded through their carbon ends. Together, these ligands form a perfectly organized octahedral geometry around the iron, giving the complex both internal rigidity and electronic stability. This tightly coordinated ion is called the ferrocyanide ion, and it contains the true structural identity of the compound. The carbonŌĆōnitrogen bonds inside the cyanide groups remain intact and unaffected by their attachment to iron, and the six ligands act collectively to share electron donation in a balanced pattern that stabilizes the metal centre against chemical disruption. The charge of the ferrocyanide ion is carried externally because all six cyanide groups are part of the internal structure, leaving a negative charge across the entire cage-like formation. This is where potassium ions play their role, not by joining the complex chemically but by arranging themselves around it through ionic attraction to maintain electrical neutrality and build a crystalline lattice. The potassium ions have no effect on the geometry of the ferrocyanide unit but instead create the static framework that allows the compound to exist as a solid material.
The structure remains just as meaningful when potassium ferrocyanide dissolves in water. The potassium ions separate immediately into solution while the ferrocyanide ion persists intact, preserving its octahedral shape rather than breaking apart into free cyanide. This highly stable behaviour explains why the compound dissolves safely and predictably without releasing toxic free cyanide under normal conditions. The internal bonding of iron to the six cyanide ligands is so strong that it prevents dissociation, and this tight structure is the reason ferrocyanide differs completely from potassium cyanide in reactivity and biological impact. The ferrocyanide unit acts as a closed coordination cage in which the cyanide groups remain bound and unavailable for independent interaction. Even in the dissolved state, the complex retains its original identity, showing that the true chemistry of the compound lies within the octahedral coordination framework rather than in the outer potassium shell.
The ferrocyanide structure also influences how potassium ferrocyanide behaves in analytical chemistry and industrial processes. When the central complex encounters iron in a higher oxidation state, especially ferric ions, the arrangement of cyanide ligands allows the formation of bridges between one ferrocyanide ion and another metal centre. This network creates the deeply coloured material known as Prussian blue, one of the oldest synthetic pigments in history and a clear visual demonstration of how the ferrocyanide complex can bond externally while preserving its internal geometry. The structural ability of cyanide ligands to link metal centres through controlled bridging without releasing themselves from iron makes the compound useful in pigment formation, metal precipitation, and specific qualitative chemical tests. This behaviour underscores the dual nature of ferrocyanide: internally locked and stable, but externally available for predictable coordination with different transition metals.
The crystalline state of potassium ferrocyanide reflects the same structural themes that define its ionic and dissolved forms. The lattice appears as a repeating arrangement in which each ferrocyanide anion sits in an organized location surrounded by potassium ions and, in many cases, by coordinated water molecules that support crystal packing and structural balance. The water is not bonded directly to the iron or the cyanide but becomes part of the extended lattice framework, helping to stabilize the compound physically rather than chemically. This ordered solid-state architecture helps the material maintain its shape, durability, and solubility profile. The ferrocyanide unit itself, however, remains unchanged regardless of whether the compound is present in a solid crystal, dissolved in solution, or integrated into pigment structures.
The molecular structure of potassium ferrocyanide therefore defines every aspect of its behaviour. The stable octahedral arrangement around iron explains the lack of free cyanide release, the gentle reactivity profile, and the relative safety of the compound compared with other cyanide salts. The external potassium ions explain its solubility and crystalline nature without interfering with the complex core. The capacity of cyanide ligands to form additional bridges when exposed to certain metal ions explains its importance in analytical chemistry and pigment science. Potassium ferrocyanide reveals how a compound can appear simple in formula yet extraordinarily rich in structural detail, illustrating how careful coordination at the atomic level creates materials that are chemically stable, functionally useful, and structurally sophisticated across both physical and applied sciences.