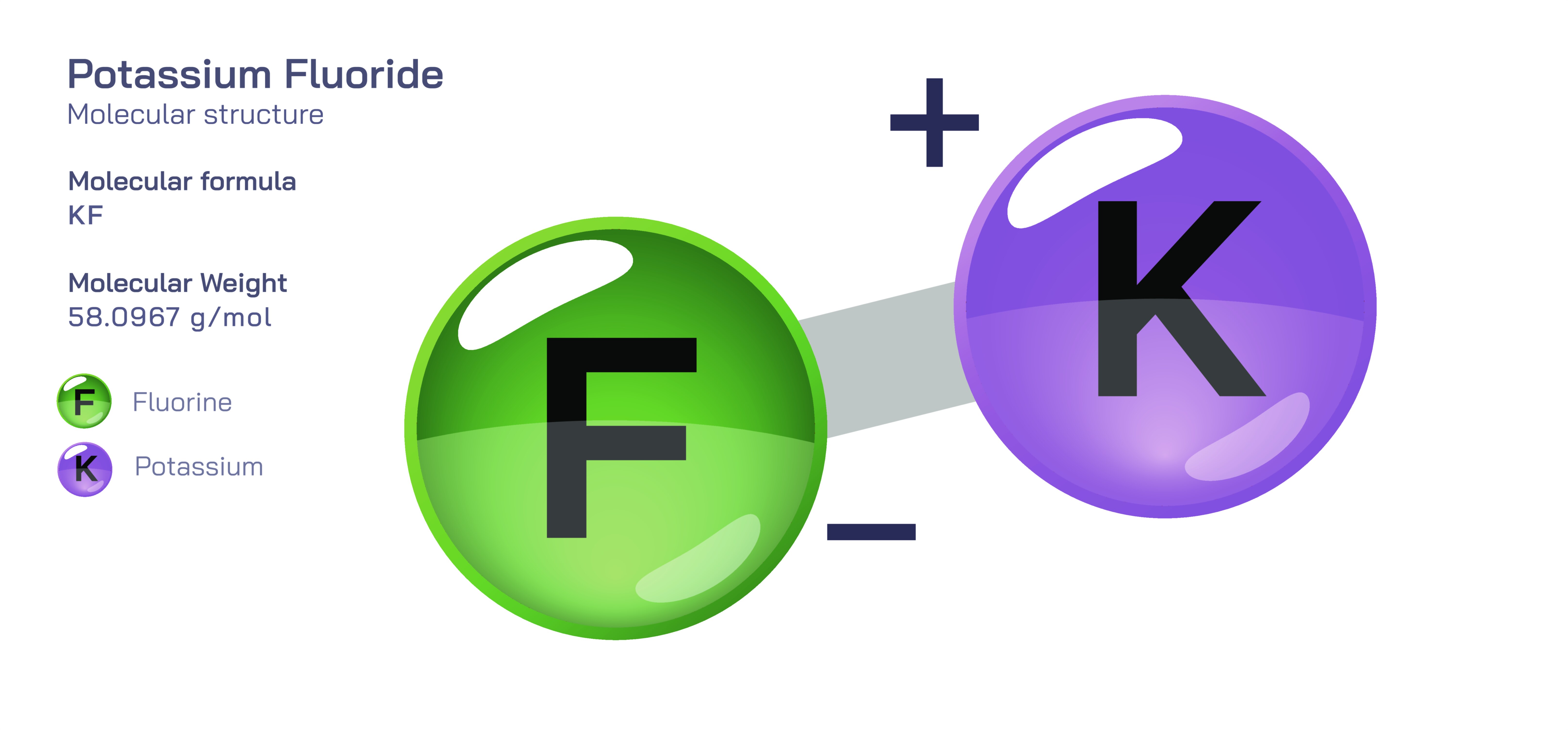
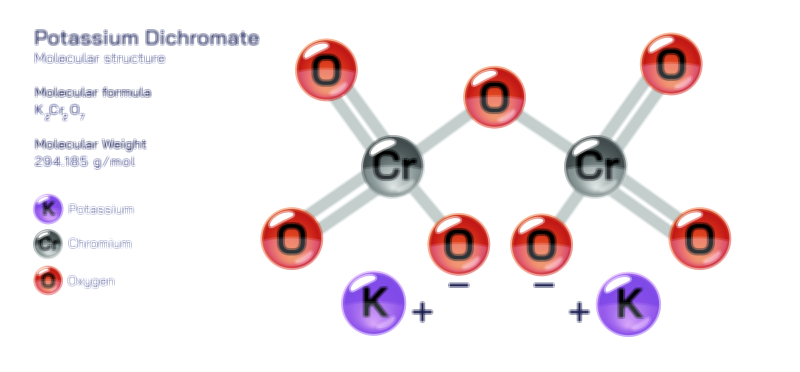
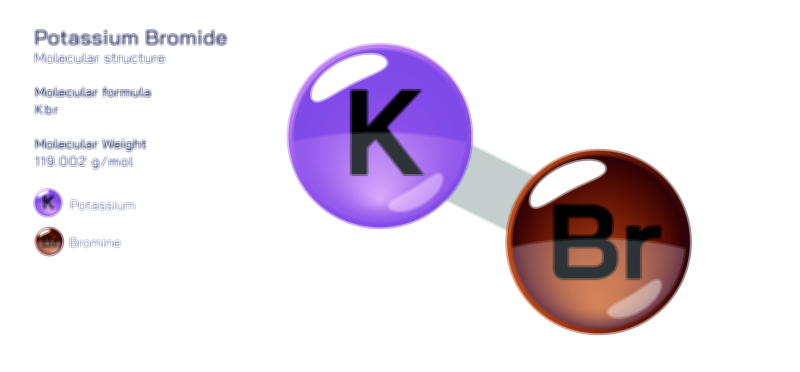
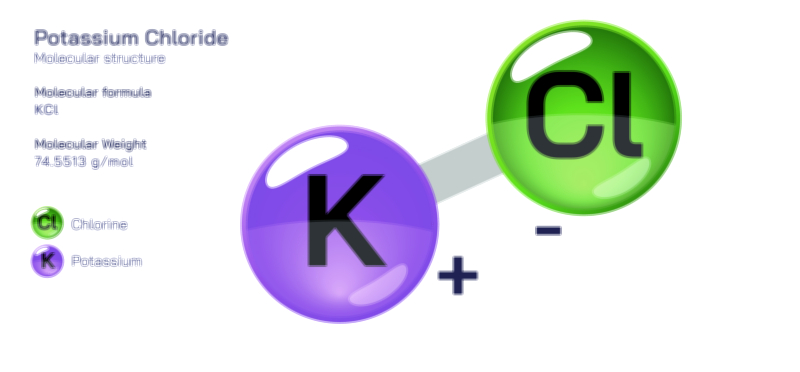
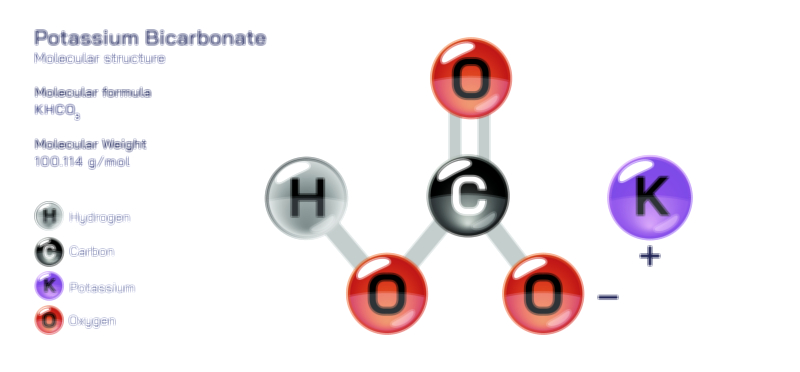
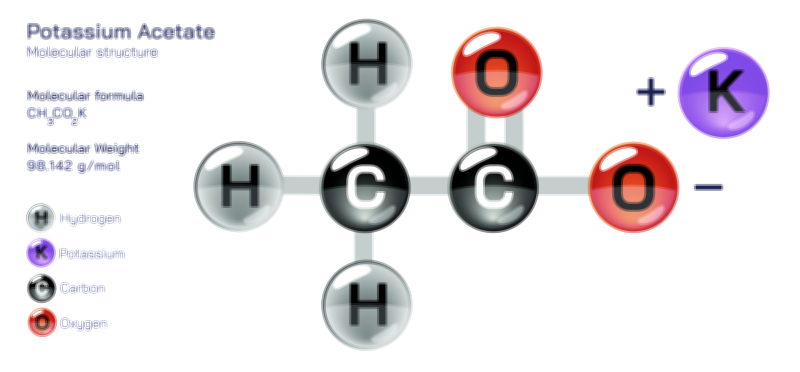
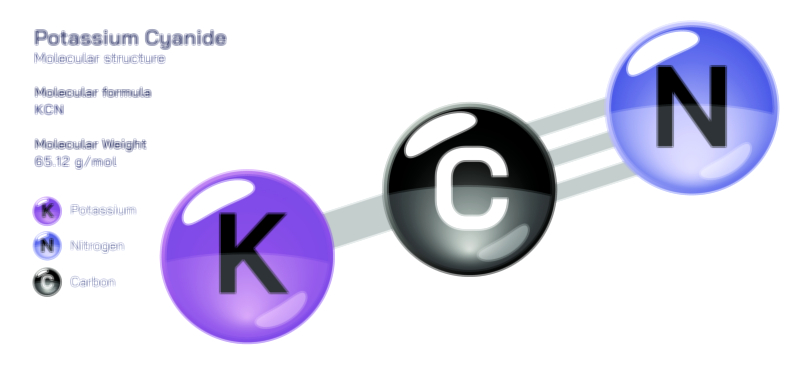
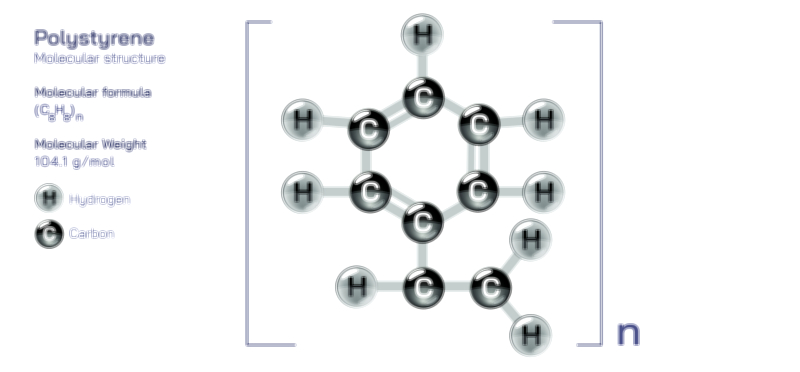
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐅𝐥𝐮𝐨𝐫𝐢𝐝𝐞 𝐂𝐡𝐞𝐦𝐢𝐜𝐚𝐥 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞
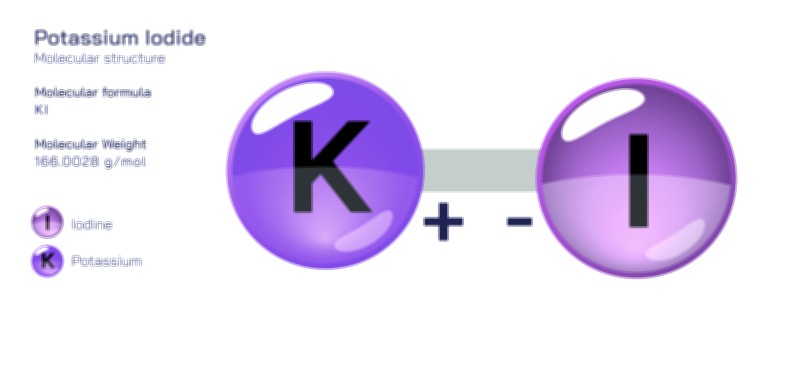
Potassium fluoride is an inorganic compound defined by the strong ionic attraction between potassium ions and fluoride ions, forming a clear and orderly crystalline framework that determines its physical behaviour and chemical properties. At the microscopic level, potassium fluoride is composed of one potassium atom that carries a positive charge and one fluorine atom that carries a negative charge after accepting an electron. This complete transfer of an electron from potassium to fluorine generates ions with opposite charges, creating a powerful electrostatic pull that binds them together. In the solid state, these alternating potassium and fluoride ions repeat in a continuous three-dimensional lattice that adopts a symmetrical cubic geometry. Each potassium ion is surrounded by fluoride ions and each fluoride ion is surrounded by potassium ions, forming a fully packed crystalline pattern that produces structural stability, a relatively high melting point, and clean, salt-like crystal formation. Because this attraction is purely ionic rather than covalent, the material remains firm and brittle rather than flexible, highlighting how atomic bonding style directly influences macroscopic characteristics.
When potassium fluoride dissolves in water, the crystal lattice breaks apart as water molecules surround and separate the ions, releasing them into solution while preserving their original charge states. This dissolution occurs easily because both potassium and fluoride ions interact strongly with water molecules; potassium stabilizes through hydration, and fluoride has a strong affinity for hydrogen bonding. Once dissociated, the ions exist freely in solution rather than as neutral molecules, and this ionic separation is responsible for the conductive, highly reactive behaviour of dissolved potassium fluoride. The fluoride ion remains structurally simple even after dissolution, consisting of a single fluorine atom stabilized by its complete outer electron shell, and this configuration explains why it behaves as a highly reactive anion that can attract hydrogen, attack silicon-oxygen bonds, and participate in etching, catalysis, and substitution reactions. By contrast, potassium ions remain relatively inert in solution, acting mainly as counter-ions that maintain charge balance while fluoride drives most of the chemistry.
The internal geometry of potassium fluoride in solid form also gives insight into how it transforms under different conditions. Because the ionic lattice is based purely on charge attraction rather than directional covalent interactions, the compound does not show complex molecular shapes in the way coordination complexes do. Instead, its structure changes only when outside forces break the ionic packing, such as melting or dissolving. The symmetry of the lattice is responsible for the compound’s transparency and uniform refractive behaviour in certain crystalline forms, and its clean cleavage reflects the repeating geometric arrangement of ions across crystal planes. The simplicity of the structure makes potassium fluoride a prototypical model for understanding ionic bonding, crystal packing, and lattice behaviour in classical solid-state chemistry.
The fluoride ion’s strong attraction toward silicon-containing compounds also has structural origins. Fluorine achieves a highly stable electronic configuration through electron gain, and once stabilized, it interacts aggressively with atoms that can support strong bond formation, especially silicon. This is why potassium fluoride, upon dissolution, becomes part of etching and glass-reactive processes where fluoride disrupts the silicon-oxygen network in glass and ceramics. This behaviour does not arise from complex multi-atom molecular shapes but directly from the inherent electronic state of the fluoride ion, showing that even simple ionic compounds can produce significant industrial and chemical effects when the resulting ions are highly reactive.
Whether examined as a solid crystal or as a dissolved electrolyte, potassium fluoride demonstrates how much information can be derived from a compound’s structural simplicity. The crystalline ionic lattice explains its hardness, symmetry, solubility, and melting behaviour. The full electron transfer between potassium and fluorine explains the strength of the bond in the solid yet the readiness with which the compound separates into ions in solution. The stability of the fluoride ion’s completed electron shell explains the chemical power it displays during reactions. Although the chemical formula of potassium fluoride appears straightforward, its structure is a precise example of how ionic bonding shapes matter, showing that even the most basic arrangement of ions can influence materials science, reactivity, physical behaviour, and industrial applications in profound ways.