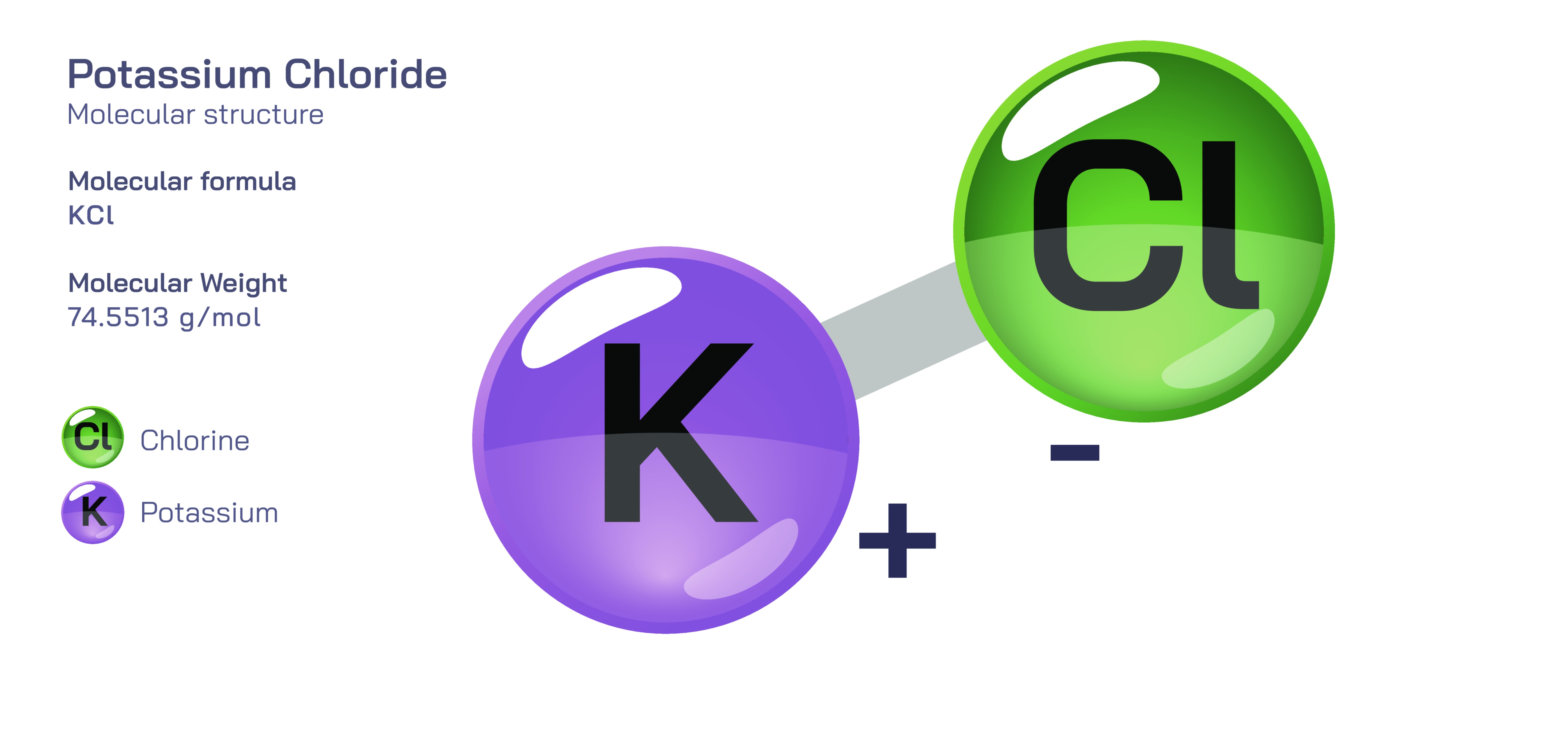
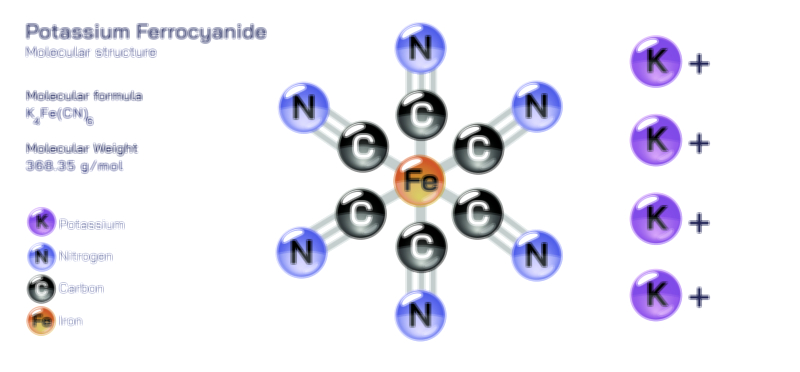
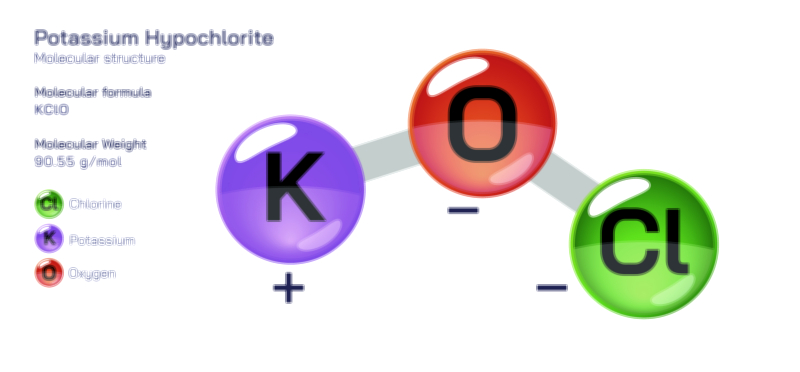
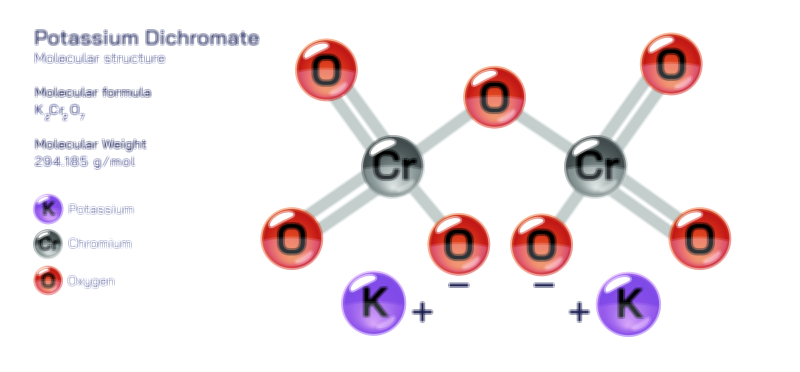
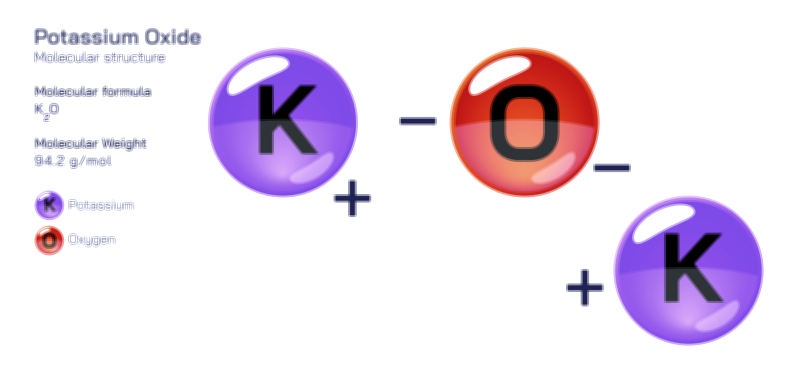𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐂𝐡𝐥𝐨𝐫𝐢𝐝𝐞 𝐂𝐡𝐞𝐦𝐢𝐜𝐚𝐥 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞
Potassium chloride is one of the simplest yet most fundamentally important inorganic compounds in science, industry, medicine, and biology, and its significance arises directly from the nature of its chemical structure. It is formed from the ionic bonding of two essential elements: potassium and chlorine. In its pure solid crystalline state, potassium chloride consists of a balanced, lattice-like arrangement of positively charged potassium ions and negatively charged chloride ions. These opposite charges attract each other strongly and create a stable cubic crystal grid, where each potassium ion is surrounded symmetrically by chloride ions and each chloride ion is surrounded symmetrically by potassium ions. This repeating arrangement extends in all directions through the crystal, giving potassium chloride its structural rigidity, characteristic cubic geometry, and high melting point. The strength of the ionic attraction makes potassium chloride solid at room temperature, while the organized lattice structure explains why it forms transparent or white cubic crystals that can fracture cleanly along defined planes. Although the compound appears externally simple, its ionic lattice is responsible for every physical and chemical behaviour it displays.
Dissolving potassium chloride in water reveals the full functional power of its structure. Once the solid salt is placed in water, the lattice breaks apart as solvent molecules surround and separate the ions. Potassium ions (K⁺) and chloride ions (Cl⁻) remain electrically stable on their own and disperse uniformly throughout the liquid, creating a strongly conductive electrolyte solution. This dissociation is possible because the compound’s ionic bond is strong enough to hold the crystal together in solid form yet weak enough to release freely moving ions in solution. The structure’s ability to shift between a rigid lattice and dissolved free ions allows potassium chloride to interact with biological tissues, electrical systems, and chemical environments in essential ways. When introduced into the human body through food, supplements, or intravenous fluids, the dissolved potassium ions play key roles in nerve conduction, muscle contraction, acid–base balance, and cellular energy transport, while chloride ions contribute to osmotic balance, digestive acidity, and the maintenance of fluid equilibrium. The ionic nature of potassium chloride therefore supports essential physiological processes that keep living organisms functioning.
The cubic lattice structure of potassium chloride is also responsible for its high solubility in water and its lack of reactivity with atmospheric gases under normal conditions. Because the lattice contains a single type of cation and a single type of anion arranged in a highly symmetric geometry, water molecules can approach both ions without interference from complex bonding patterns. This is a major reason why potassium chloride dissolves easily in water but remains chemically stable in dry air. In contrast to salts that hydrolyze or decompose in moisture, potassium chloride does not break down into secondary products but simply dissociates, meaning it can be dissolved and recrystallized repeatedly without losing identity. This stability is essential for laboratory formulations, standard reference solutions, and food-grade materials where purity and reliability are mandatory. In industrial crystallizers, potassium chloride forms beautiful, well-defined cubic crystals, which confirm the structure visually and demonstrate how strongly geometry influences macroscopic properties.
The structural simplicity of potassium chloride also explains its electrical behaviour. In the solid state, ions are fixed in the lattice and cannot move freely, so the material does not conduct electricity. However, once melted or dissolved, the ions are liberated and can travel in response to an electric field, turning potassium chloride into an excellent conductor. This transformation underlines how the mobility of ions rather than their mere presence determines conductivity. In chemical processing, this behaviour allows potassium chloride to participate in electrolysis reactions that produce potassium hydroxide and chlorine gas, and these products become raw materials for fertilizers, disinfectants, pharmaceuticals, and numerous industrial chemicals. The ionic structure enables efficient splitting during electrolysis because the bonding between potassium and chloride is purely electrostatic and not covalent. This illustrates how atomic-level forces can influence large-scale manufacturing practices.
Potassium chloride’s structure also determines its ability to substitute sodium chloride in many roles. The ions that form potassium chloride have almost the same size ratio as the ions in sodium chloride, and both compounds adopt the same type of cubic lattice. As a result, potassium chloride has a taste, solubility profile, and physical behaviour similar to ordinary salt, which makes it useful as a sodium-reduced dietary alternative for individuals with hypertension or cardiovascular sensitivity. However, the difference in ionic composition means the compound has distinct physiological effects: potassium helps relax blood vessel walls and balance electrolytes, while chloride supports acid–base regulation. Although the crystal structure resembles common table salt, the biological effects differ strongly because potassium ions interact differently with nerve channels, muscle tissues, and renal filtering mechanisms. This contrast shows how materials that look nearly identical externally can have very different interactions inside the human body depending on their ionic composition.
The structural makeup of potassium chloride also plays a central role in environmental geochemistry. Its crystal lattice is the defining structure of a natural mineral called sylvite, found in evaporite geological deposits where ancient seas have concentrated and dried over time. These potassium chloride deposits serve as a major global source of potassium fertilizers, which are essential for plant nutrient programs that support worldwide food production. The soil, like the human body, requires potassium to activate enzymes, regulate osmotic balance, and support water movement through plant tissues. Because potassium chloride dissolves rapidly in soil moisture, it releases potassium ions that roots can absorb immediately, demonstrating once again how ionic structure translates into biological and agricultural value. The chloride component, although useful in moderated amounts, must be managed carefully in salt-sensitive crops. This reminds us that even structurally simple compounds require thoughtful application depending on biological context.
Understanding the chemical structure of potassium chloride provides insight into why this salt plays such a wide-ranging role in the world. Its symmetric cubic lattice explains its hardness, crystalline appearance, and stability. Its ionic dissociation explains its solubility, conductivity, and biomedical importance. Its electrical properties explain its relevance in industrial electrolysis and chemical production. Its similarity to sodium chloride explains its sensory familiarity while simultaneously highlighting physiological differences. A compound so structurally simple nonetheless influences nutrition, physiology, geochemistry, agriculture, laboratory science, pharmaceuticals, and chemical engineering every day. Potassium chloride demonstrates how a basic ionic structure can support extremely varied functions once it enters different physical, biological, and industrial environments. It is a perfect example of how the invisible architecture of atoms governs the visible properties of matter, shaping both natural systems and human technology in ways that are easy to overlook yet essential for life and progress.