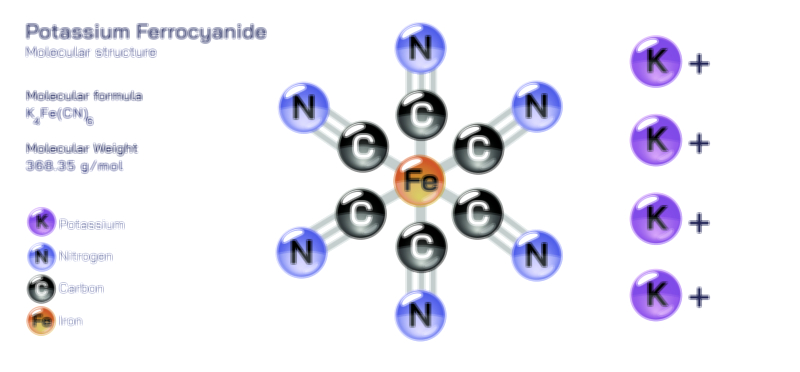
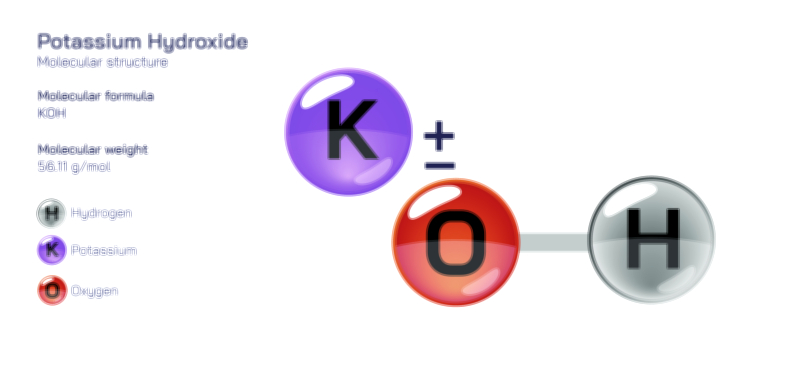
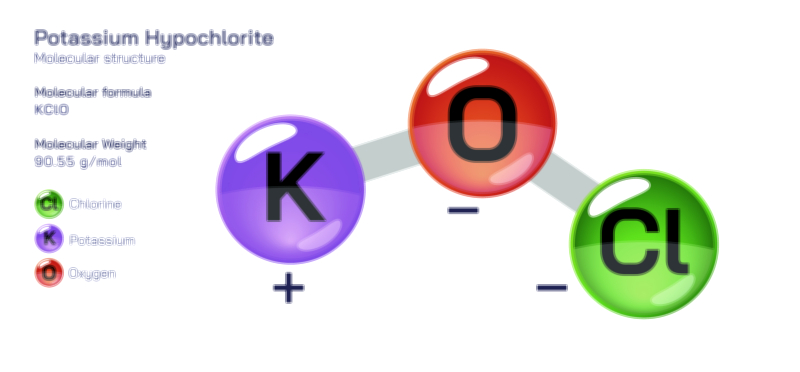
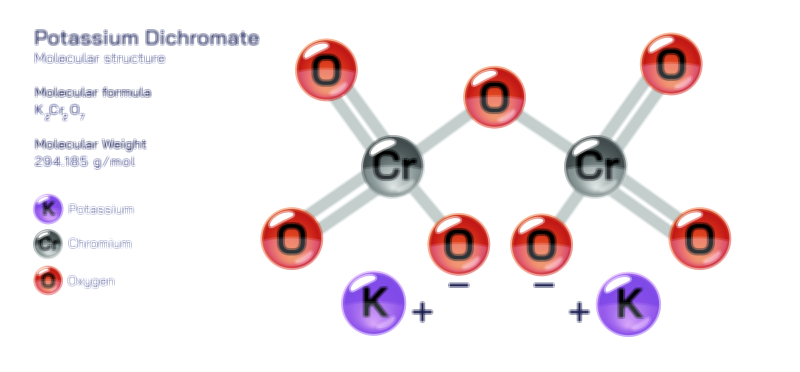
ØÉÅØÉ©ØÉŁØÉÜØɼØɼØÉóØÉ«ØÉ” ØÉüØÉóØÉ£ØÉÜØɽØÉøØÉ©ØɦØÉÜØÉŁØÉ× ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉÜØɦØÉØ ØÉĆØÉ®ØÉ®ØÉźØÉóØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØɼ
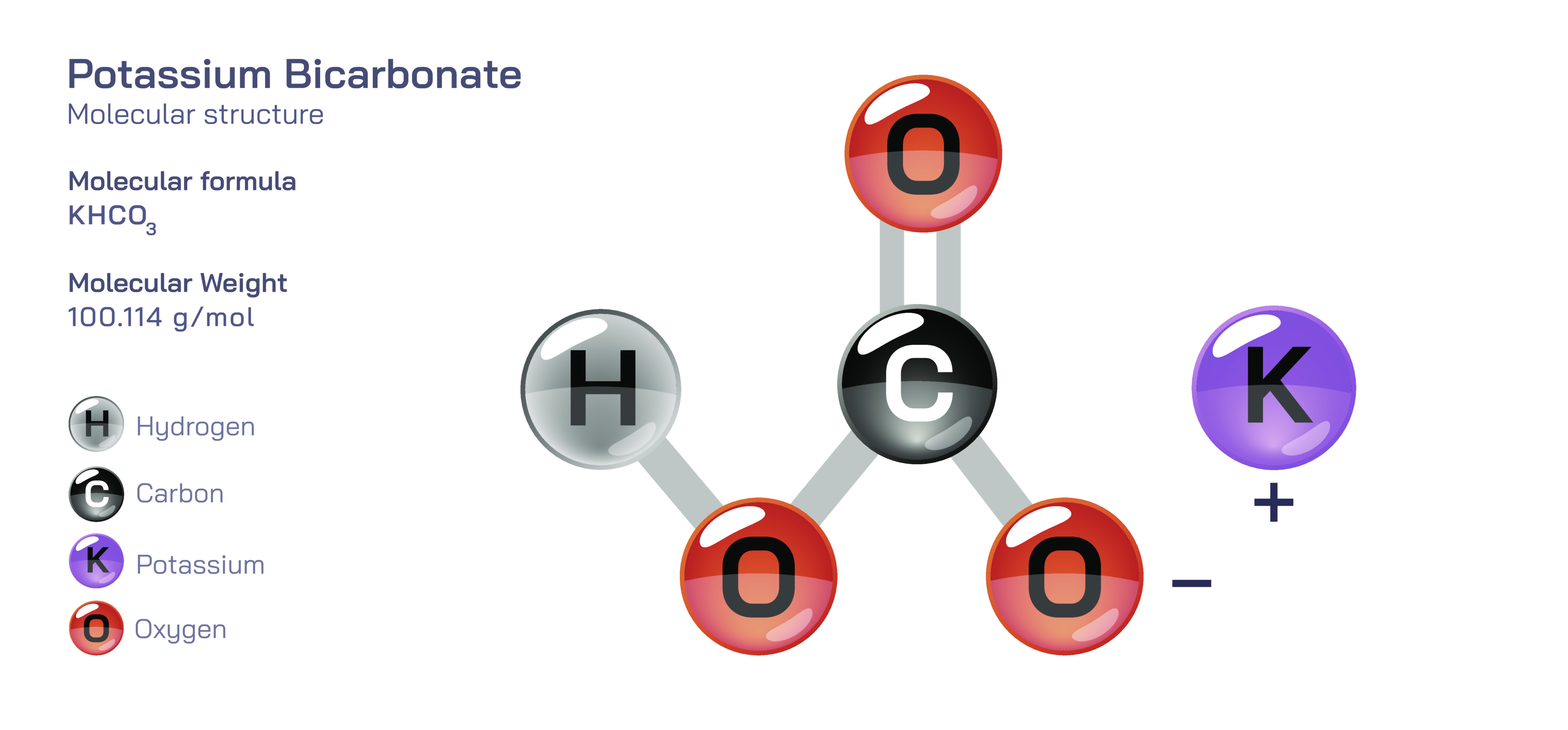
Potassium bicarbonate is a versatile inorganic compound widely used in food chemistry, agriculture, fire safety, pharmaceuticals, and environmental management due to its balanced alkalinity, gentle reactivity, and high solubility. Chemically, it is represented by a salt formed from potassium ions and bicarbonate ions. The bicarbonate ion originates from carbonic acid and consists of a carbon atom bonded to oxygen atoms in a molecular arrangement that allows the compound to act as a mild base capable of reacting with acids while releasing carbon dioxide. Combined with the potassium ion, this structure forms a stable crystalline solid that is white, odourless, and freely soluble in water. When dissolved, potassium bicarbonate dissociates completely into KŌü║ and HCOŌéāŌü╗ ions, creating a buffering environment that helps regulate pH without causing corrosive changes. This gentle chemical behaviour explains why potassium bicarbonate finds widespread use in biological systems, food formulations, controlled agricultural nutrition, extinguishing agents, and medical preparations. While simple in composition, the bicarbonate structure gives the compound broad effectiveness across chemical, physiological, and industrial processes.
A key property of potassium bicarbonate is its ability to act as a buffering agent. When dissolved in solution, the bicarbonate ion balances acidity by neutralizing excess hydrogen ions, producing carbon dioxide and water in the process. This behaviour makes it especially useful in formulations that require stable pH control. Unlike strong alkaline compounds that create sharp changes in chemical conditions or pose safety risks, potassium bicarbonate adjusts acidity gently and predictably, which is critical for biological systems and sensitive chemical reactions. Its high solubility allows it to distribute evenly without forming residue, while the potassium component contributes essential electrolytes that support nerve signalling, cellular metabolism, hydration balance, and muscle function when used in physiological or nutritional applications. Taken together, these attributes give potassium bicarbonate a unique blend of buffering efficiency and biological safety.
In the food industry, potassium bicarbonate is widely used as a leavening agent and acidity regulator. When exposed to moisture and heat or combined with acidic ingredients, it releases carbon dioxide gas, causing dough and batter to rise. It is often used as an alternative to sodium bicarbonate in sodium-restricted diets because it performs the same leavening function without increasing sodium intake. Beyond leavening, potassium bicarbonate helps maintain flavour balance and pH stability in beverages, dairy products, confections, and processed foods. It acts as a mild antimicrobial agent in some formulations by reducing excess acidity that encourages microbial growth. In winemaking, potassium bicarbonate helps correct high acidity levels during fermentation, improving flavour and balance while minimizing sensory defects. Its neutral taste and predictable chemical action make it favourable for food processing where precision and consistency are essential.
In agriculture, potassium bicarbonate serves as both a nutrient source and a protective agent. As a fertilizer, it provides readily available potassium without introducing chlorides or sulfates, which may stress certain plants or accumulate in sensitive soils. This makes it particularly suitable for greenhouse cultivation, hydroponics, and high-value crops such as fruits and vegetables, where precise nutrient balance is important. Its mild alkalinity also helps improve plant resilience during stress conditions, especially when soil or water becomes overly acidic. Beyond nutrition, potassium bicarbonate is an effective fungicide used to control powdery mildew, black spot, and other fungal diseases in vineyards, orchards, and vegetable farms. When applied on leaves, its bicarbonate component disrupts fungal cell balance and prevents spore viability, while its potassium content nourishes plants rather than damaging them. This dual benefit has increased its popularity as an environmentally friendly alternative to synthetic fungicides.
Potassium bicarbonate plays an essential role in fire safety systems, particularly in dry chemical fire extinguishers designed for Class B and Class C fires involving flammable liquids and electrical equipment. When sprayed onto flames, potassium bicarbonate interrupts the combustion process by neutralizing free radicals and creating a protective barrier between heat and oxygen. It performs more efficiently than sodium bicarbonate in high-temperature fire situations due to its greater fire-suppressing strength and ability to maintain its extinguishing effect longer. For this reason, it is widely used in industrial plants, aviation safety equipment, fuel storage facilities, automotive workshops, and environments where ignition risk is high. Potassium bicarbonate-based extinguishing powders flow smoothly, resist caking, and leave relatively less residue, making cleanup easier.
In medicine and pharmaceuticals, potassium bicarbonate supports the treatment of conditions related to electrolyte imbalance and metabolic acidosis. Doctors may prescribe it to correct low potassium levels in patients who lose electrolytes through illness, dehydration, or certain medications. Its bicarbonate component helps neutralize acidity in the bloodstream, supporting patients with conditions that involve acid accumulation. Its controlled alkalinity also helps in relieving mild acid indigestion in some antacid formulations. Unlike stronger alkaline salts, potassium bicarbonate provides a gradual buffering effect that does not cause large pH swings or gastrointestinal irritation when used appropriately under clinical guidance. It is also added to oral rehydration solutions and sports supplements to support recovery from physical exertion and dehydration.
Potassium bicarbonate also contributes to laboratory and industrial chemical processes. It is used in buffer preparation, pH stabilization, analytical reagent formulation, and chemical synthesis where gentle alkalinity is required. In water treatment systems, especially in aquaculture and controlled ecosystems, potassium bicarbonate raises alkalinity safely without adding harmful salts and helps maintain stable conditions for aquatic organisms. It is also used in fermentation industriesŌĆösuch as biofuel and enzyme productionŌĆöto regulate pH and improve productivity.
Across food science, agriculture, fire protection, medical therapy, environmental management, and industrial chemistry, potassium bicarbonate demonstrates how a single compound can adapt to multiple roles because of its structural balance between potassium and bicarbonate ions. Its ability to neutralize acidity, distribute cleanly in water, act as a nutrient in plants and humans, and suppress combustion gives it value that extends far beyond its simplicity. Whether supporting nutrition, protecting crop health, ensuring fire safety, stabilizing biochemical reactions, or improving flavour in winemaking and baking, potassium bicarbonate quietly contributes to everyday life and industrial efficiency in ways most people never see.