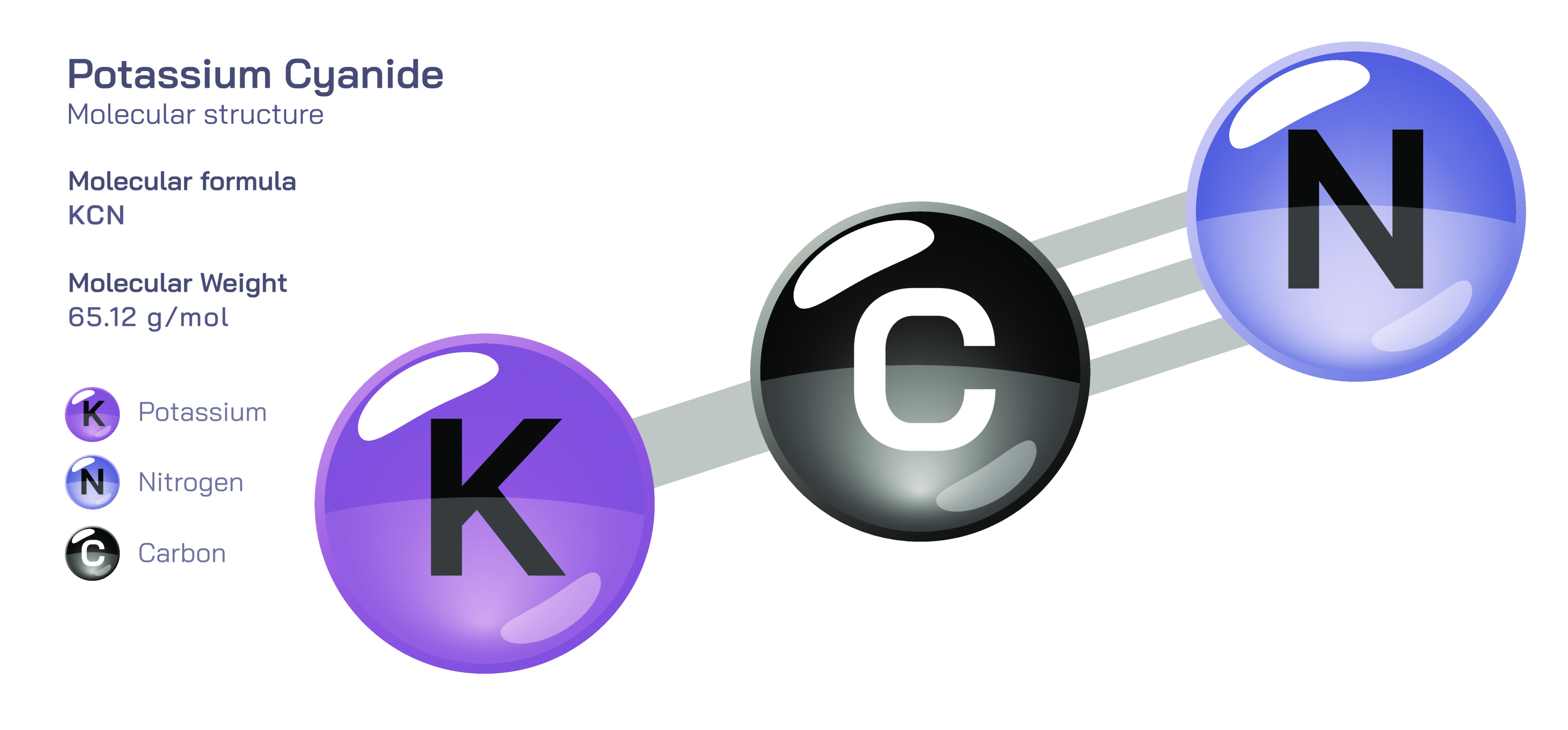
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐂𝐲𝐚𝐧𝐢𝐝𝐞 𝐂𝐡𝐞𝐦𝐢𝐜𝐚𝐥 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞
Potassium cyanide is an inorganic compound composed of potassium ions and cyanide ions arranged in a strongly ionic crystalline lattice. In its pure state, it appears as white, crystalline granules resembling common table salt. The chemical structure is simple in formula but notable in bonding behaviour: the potassium ion (K⁺) is positively charged, while the cyanide ion (CN⁻) carries a negative charge. These opposite charges attract one another and stabilize the compound in its solid form.
At the molecular level, the cyanide ion is the dominant structural component. It consists of one carbon atom and one nitrogen atom bonded tightly by a strong triple bond. This short, rigid bond creates a linear shape, making CN⁻ a compact and highly stable ion internally. Within the solid lattice, potassium ions surround cyanide ions in an ordered arrangement held together through electrostatic (ionic) forces rather than shared electrons. Because the cyanide ion is already structurally rigid, its geometry does not change when incorporated into the salt—only the ionic packing around it shifts to form the lattice.
When potassium cyanide dissolves in water, the lattice separates completely, and the ions become free in solution. The potassium ion remains fully solvated and largely inert, while the cyanide ion stays intact as a carbon–nitrogen triple-bonded pair. This dissociation is the most important structural feature of potassium cyanide because the cyanide ion in free form has strong chemical affinity toward certain metals—particularly those in transition-metal complexes—and can bind tightly to them. The ability of the cyanide ion to form extremely stable coordination complexes with metals stems from the presence of the nitrogen atom’s electron pair along with the electron density in the carbon–nitrogen bond.
In the crystalline solid, the ionic lattice provides stability, but in aqueous environments, the free CN⁻ ion becomes highly reactive due to its strong electron-pair donating ability. Even though the potassium–cyanide combination is structurally simple, the nature of the cyanide ion makes the compound chemically powerful.
Outside the lattice, the rigid linear CN group remains unchanged, and it is this persistent microscopic structure that governs potassium cyanide’s behaviours in industrial metal extraction, electroplating chemistry, and analytical chemistry methods—situations where dissolved cyanide binds selectively to metals.
It is important to emphasize that although the structure of potassium cyanide can be described scientifically, cyanide compounds are extremely hazardous. The same electronic properties that allow CN⁻ to bond strongly with metals also allow it to interfere with biological enzymes that contain metal centres. For this reason, potassium cyanide must only be handled in strictly controlled laboratory or industrial environments by trained professionals following safety protocols.
In summary, the chemistry of potassium cyanide is rooted in its straightforward but highly significant structure:
• a potassium ion (K⁺) forming the ionic component of the lattice, and
• a cyanide ion (CN⁻) with a linear carbon–nitrogen triple bond, internally covalent but externally ionic in relation to potassium.
The ionic lattice explains its crystalline physical form, while the cyanide ion’s linear triple-bonded structure explains its unique reactivity in solution. Although simple in composition, the structure determines all observable characteristics, making potassium cyanide a powerful and tightly regulated compound in materials science and chemistry.