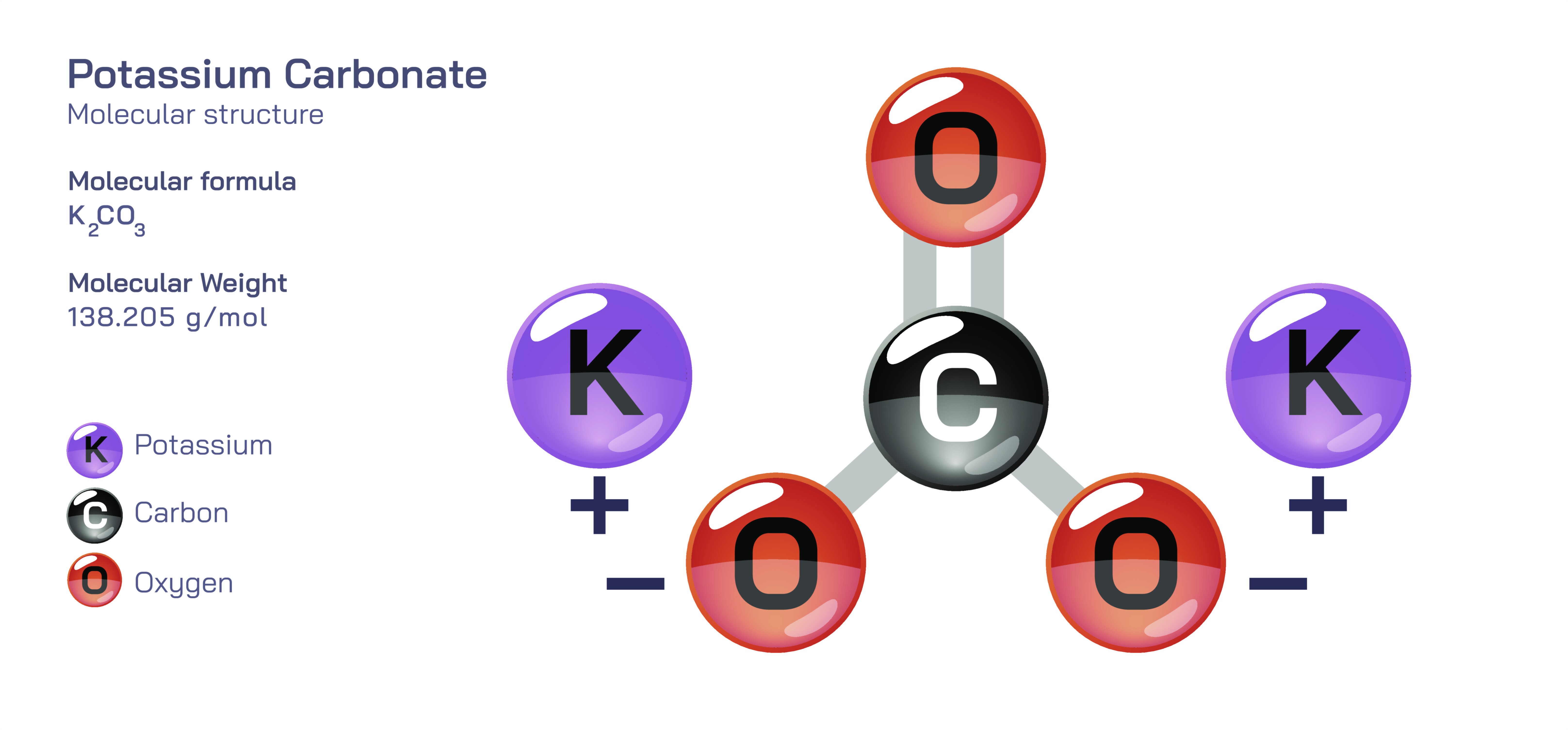
Potassium Carbonate — Chemical Structure, Properties, Industrial Uses, and Practical Significance
Potassium carbonate is a highly important inorganic compound that plays a major role in industrial chemistry, food processing, glass manufacturing, and laboratory applications. Although often overshadowed by more commonly discussed alkaline compounds, potassium carbonate represents a cornerstone material in sectors where precise alkalinity, controlled reactivity, and reliable solubility are required. It appears as a white, granular or powdered solid that dissolves readily in water to form a strongly alkaline solution. Its chemical identity is defined by a compound formed from potassium, carbon, and oxygen in a stable arrangement that gives rise to predictable and versatile interactions in aqueous systems. The structure is based on a carbonate group bonded to two potassium atoms, and this composition gives the compound its characteristic basic behavior and solubility. Unlike corrosive strong alkalis such as potassium hydroxide, potassium carbonate offers a high degree of alkalinity in a milder solid form that is easier to store, transport, and handle in large quantities across industrial environments. At the same time, its capacity to regulate acidity, promote ion exchanges, and participate in precise chemical reactions makes it indispensable in fields that require fine control over pH and reaction pathways.
Even without viewing detailed chemical diagrams, it is possible to appreciate the significance of the compound by considering how its internal structure influences its behavior. The carbonate portion of the molecule acts as the key reactive site and is strongly attracted to hydrogen ions. This affinity allows potassium carbonate to neutralize acids efficiently, forming potassium salts and releasing benign by-products such as carbon dioxide and water in many reactions. The potassium ions themselves contribute to the compound’s high solubility because they readily dissociate and disperse in water, allowing the alkaline properties to activate instantly after dissolution. Once dissolved, the compound increases the pH of the solution and provides buffering capacity, meaning it can stabilize acidity levels over time rather than allowing sudden fluctuations. This buffering ability explains why potassium carbonate appears frequently in environments where long-term pH stability is essential to product performance, safety, or flavor consistency. Unlike some alkaline compounds that leave behind strong tastes or undesired residues, potassium carbonate functions with a controlled and predictable profile, making it suitable even in sensitive chemical systems.
Historically, potassium carbonate has roots in early human industrial activity because it could be obtained from the ashes of burned vegetation. For centuries, people extracted alkaline salts by filtering water through plant ash and evaporating the solution to obtain a mixture dominated by potassium carbonate. This material was crucial for traditional soap making, textile processing, leather tanning, and early glassmaking. With the advent of modern chemical production, the compound is now manufactured industrially through refined processes that allow large-scale, high-purity output, supporting global demand across multiple sectors. Although technology has evolved dramatically from the early days of ash filtration, potassium carbonate has retained its role because no alternative has matched its particular blend of solubility, alkalinity, and compatibility with organic and inorganic systems.
Glass production remains one of the most significant uses of potassium carbonate. When used as a fluxing agent in glass formulation, it lowers the melting point of silica and improves the clarity, hardness, and brilliance of specialty glass. Potassium-based glass exhibits a distinct optical character and enhanced resistance to mechanical wear, making it desirable for high-quality glassware, laboratory containers, smartphone screen materials, and heat-resistant products. The compound helps achieve these properties without making the glass brittle, which is critical for durability. Potassium carbonate also contributes to the formation of certain ceramics and enamel coatings, where it participates in the fusion of minerals at high temperature and influences the surface appearance and hardness of the final material.
In the food industry, potassium carbonate is valued not as a reactive chemical but as a precise regulator of acidity and a textural enhancer. It appears in controlled amounts in bakery products, where it helps modify dough structure, improve leavening in specific formulations, and adjust flavor balance without the sodium content that accompanies many other food-grade alkaline compounds. It plays a traditional role in the preparation of certain noodles, pretzels, and confectionery products, where its alkalinity contributes to firm texture, characteristic color, or distinct taste profiles that cannot be replicated easily with substitutes. Because potassium is a biologically essential mineral, the compound finds a place in low-sodium food technology as well, offering a functional alternative where reducing sodium intake is medically or nutritionally desirable. When used at safe regulated levels, it allows food manufacturers to support flavor and stability while meeting dietary reformulation goals.
Potassium carbonate is also indispensable in chemical industries where it participates as a reactant, processing agent, or catalyst support material. In pharmaceutical synthesis, it enables reactions where controlled alkaline conditions are required to activate molecular groups or neutralize acidic intermediates. In the manufacture of dyes, detergents, personal care products, and specialty chemicals, it provides alkalinity without introducing corrosive hazards associated with stronger bases. The compound is also important in laboratory environments where accurate pH adjustment is necessary without interfering with organic reactions or leaving behind strong ionic residues. In many organic reactions, the choice of base can determine whether the targeted product forms correctly or whether side reactions disrupt the synthesis; potassium carbonate occupies an optimal balance that allows reactions to proceed cleanly and predictably.
Beyond chemical manufacturing and food processing, potassium carbonate plays a role in water treatment and environmental systems. It is used to correct overly acidic water conditions and support mineral balance in industrial water loops, cooling systems, and aquaculture environments where stable pH is essential for biological and mechanical safety. Because the compound dissolves completely and does not introduce environmentally persistent contaminants when used responsibly, it offers a dependable option in applications where effectiveness must pair with ecological responsibility. It also supports fire suppression formulations in certain specialty extinguishing systems, where its chemical profile allows smothering of flames without leaving corrosive residues that might damage equipment in enclosed spaces.
Safety considerations with potassium carbonate are defined not by toxicity but by alkalinity. Although the compound is not corrosive in the way strong bases are, concentrated solutions can irritate the skin, eyes, and respiratory passages because of the elevated pH. For this reason, industrial handling includes protective gear, dust suppression measures, and ventilation systems to avoid inhalation of fine particles. When dissolved in water, the dilution process generates heat, so adding the solid gradually to water under controlled stirring minimizes splashing and thermal shock. In controlled environments, potassium carbonate remains one of the more manageable alkaline compounds because it is chemically stable, nonflammable, and free from volatile by-products. Disposal typically involves controlled dilution or neutralization with acids, and because its reaction products are not environmentally hazardous, neutralized forms can be processed safely through conventional waste systems.
Potassium carbonate’s broad impact stems from the fact that it serves as both a functional industrial material and a supporting compound that allows other technologies to perform efficiently. Whether contributing to glass that is clearer and more durable, food that rises and browns properly, pharmaceuticals that require exact chemical conditions, or water systems that must maintain balanced pH, the compound demonstrates the far-reaching importance of precision chemistry in everyday life. Its utility lies in its reliability: it dissolves consistently, reacts predictably, and adapts to systems ranging from high-temperature mineral fusions to delicate biological formulations. The compound also illustrates a recurring theme in industrial science — that some of the most transformative substances are not always dramatic or visibly reactive, but instead quietly shape the materials, structures, and processes that define modern living.
The story of potassium carbonate is therefore one of evolution and continuity. It originated in the early days of civilization as a product of natural ash and has since become a refined, high-purity compound powering some of the world’s most advanced manufacturing sectors. Its presence spans ancient craft, modern technology, nutritional science, and laboratory innovation alike. Understanding potassium carbonate is not only a matter of identifying its structure or categorizing it as an alkaline compound, but appreciating how its predictable reactivity and versatile behavior make it a foundational ingredient in industries and products that support daily life.