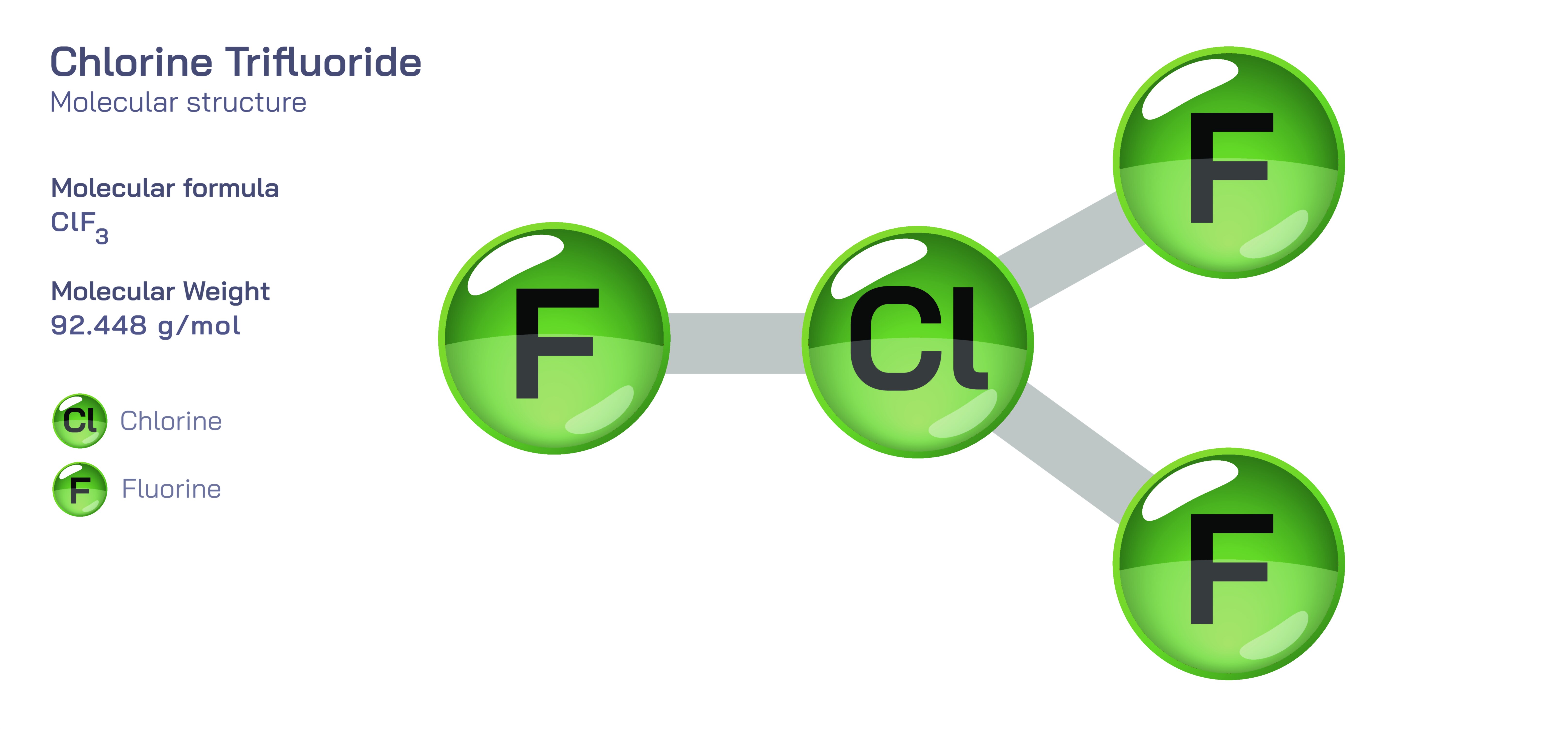
Chlorine Trifluoride — Reactive Halogen Compound
Chlorine trifluoride is one of the most extraordinary, dangerous, and chemically powerful compounds ever discovered, earning a near-legendary status in the world of reactive halogens and extreme oxidizers. Known for its unmatched reactivity, its ability to ignite materials typically considered nonflammable, and its reputation for being nearly impossible to contain safely, chlorine trifluoride stands as a striking example of chemistry pushed to its reactive limits. Although composed of only one chlorine atom and three fluorine atoms, this compound behaves far beyond what its simple formula suggests. It possesses a volatile, aggressive character that challenges conventional understanding of chemical stability and reactivity. To appreciate chlorine trifluoride in full, one must explore its molecular structure, extreme oxidizing power, industrial applications, historical significance, and the profound safety considerations that surround it. The compound demonstrates how specific arrangements of atoms can yield behavior so potent that it borders on the surreal, reminding us that chemical reactivity is deeply rooted in microscopic structures that dictate macroscopic extremes.
At the molecular level, chlorine trifluoride (ClF₃) is a highly asymmetric and reactive interhalogen compound belonging to the family of halogen fluorides. Its structure is shaped like a bent or T-shaped arrangement due to the influence of nonbonding electron pairs on the central chlorine atom. This geometry creates an uneven distribution of charge and contributes to the molecule’s high polarity and aggressive chemical behavior. Chlorine, normally less electronegative than fluorine, is forced into an unusual oxidation state through bonding with three strongly electronegative fluorine atoms. This elevates the chlorine to a deeply electron-deficient state, driving the compound to act as a ferocious oxidizer. Even without an external ignition source, chlorine trifluoride can release large amounts of energy simply by encountering materials capable of being oxidized. It is so reactive that it can attack substances typically considered resistant to chemical reaction, including metals, glass, ceramics, concrete, sand, and many organics. The strength of the chlorine–fluorine bonds and the eagerness of fluorine to pull electrons from virtually anything create conditions under which reactions occur spontaneously, sometimes explosively.
One of the most remarkable characteristics of chlorine trifluoride is its ability to ignite substances normally considered nonflammable. Most fuels require both oxygen and an ignition source to burn, but chlorine trifluoride carries its own oxidizing power so intensely that it bypasses the need for atmospheric oxygen entirely. When it comes into contact with organic materials such as cloth, paper, wood, or oils, it reacts instantly, often generating flames or violent decomposition. What makes ClF₃ even more astonishing is that it can cause combustion in materials that are not only nonflammable but normally used to extinguish fires. Substances like asbestos, sand, and even concrete can serve as fuel when exposed to chlorine trifluoride. Its reactivity with glass and ceramics arises from its ability to break silicon–oxygen bonds and release large amounts of energy as it forms stable fluorides. This degree of chemical aggressiveness makes the compound extremely difficult to store or transport using conventional containers.
Because of its destructive nature, chlorine trifluoride requires specialized handling and containment. It must be stored in containers made of specific metals such as nickel, Monel, or special steel alloys that can rapidly form protective fluoride layers on their surfaces. These protective layers prevent further attack from the compound, acting as a chemically resistant barrier. However, even these materials are safe only under completely dry conditions. The presence of moisture—even traces of water vapor—can disrupt the protective film and trigger a violent reaction. Handling chlorine trifluoride therefore demands an environment free from air humidity, organic residues, incompatible substances, or even microscopic impurities. Industrial systems designed to contain ClF₃ operate using meticulous engineering protocols, rigorous monitoring, and emergency safeguards, emphasizing how a compound’s molecular nature dictates the need for extreme caution at every step.
Despite its hazards, chlorine trifluoride holds important industrial applications that highlight its unique strengths. One of its most significant uses is in the semiconductor industry, where it functions as an effective cleaning and etching agent for removing metal residues and unwanted contamination inside high-temperature reactors. Because ClF₃ operates in its gaseous state and does not require plasma to initiate reaction, it can access narrow passages and complex internal surfaces that other cleaners cannot reach. This ability to strip deposits without needing mechanical scraping or harsh thermal cycling makes it valuable in maintaining silicon wafer processing equipment. Semiconductor fabrication demands exceptionally pure and clean internal environments, and chlorine trifluoride’s potent oxidizing nature helps achieve those conditions. Its use in this industry illustrates how even the most dangerous chemicals can become indispensable when managed with precision and respect.
Historically, chlorine trifluoride attracted attention in nuclear fuel processing as well. It was investigated for its ability to convert uranium compounds into volatile uranium hexafluoride, a key intermediate in nuclear fuel refinement. Its reactivity made the process more efficient in theory, though its extreme hazards limited its widespread adoption. At various points in history, its energy density and reactive nature also led to experimental interest as an incendiary or military material, but these applications proved far too dangerous and unpredictable to pursue. Its potential for explosive decomposition, violent reactivity with common materials, and the difficulty of controlling it safely made ClF₃ unsuitable for battlefield use despite its formidable chemical power. The challenges associated with its containment, transport, and stability overshadowed any tactical advantage.
The safety profile of chlorine trifluoride must be understood to appreciate fully its place in chemistry. Human exposure to even small concentrations of its vapors can cause severe respiratory damage, chemical burns, and tissue destruction. Contact with skin or eyes produces catastrophic injuries, not because of heat alone but because the compound aggressively attacks organic molecules, dissolving proteins, fats, and structural tissues. Inhalation of its decomposition products, such as hydrogen fluoride or chlorine-containing acids, further compounds the hazards. Emergency response teams require specialized training, equipment, and protocols when working near chlorine trifluoride, often relying on inert gas purging, vacuum systems, and remote handling technologies to minimize exposure. These dangers highlight how profoundly a molecule’s electron distribution and bonding behavior influence real-world safety considerations.
Environmentally, chlorine trifluoride seldom persists in its original form due to its extreme reactivity. If released, it quickly reacts with water vapor in the atmosphere to produce hydrochloric and hydrofluoric acids, both of which pose environmental hazards but are less reactive than ClF₃ itself. Although industrial use is tightly regulated, accidents involving chlorine trifluoride have historically caused severe damage due to its ability to destroy infrastructure and organic matter. These historical incidents have reinforced the necessity of rigorous containment and monitoring, making ClF₃ a prime example of how high-energy chemicals require extraordinary engineering controls. The compound’s environmental footprint, while potentially severe in immediate accidents, does not manifest as long-term persistence because it readily decomposes into other forms.
Chemically, chlorine trifluoride represents an extreme case of halogen behavior. Halogens are known for high reactivity, but the combination of chlorine and fluorine amplifies this characteristic to a peak rarely matched by other substances. Understanding its behavior deepens our broader understanding of chemical bonding, electronegativity, oxidation states, and energy release during bond formation. Researchers studying ClF₃ often use it as a benchmark for evaluating oxidation strength, reactivity limits, and the structural basis for energetic reactions. Its existence challenges chemists to consider how molecular structure creates the conditions for explosive or aggressive behavior and how even simple combinations of atoms can produce unexpectedly powerful effects.
Chlorine trifluoride symbolizes the dual nature of chemical innovation: it is simultaneously a tool of precision in advanced technologies and a substance of extreme hazard. Its uses in semiconductor fabrication contribute to the creation of devices that define modern life, from smartphones to computers to renewable energy systems. Yet its inherent danger serves as a reminder that technological advancement often relies on understanding and safely harnessing the forces of nature at their most intense. The compound’s legacy underscores the importance of balancing innovation with responsibility, engineering controls with scientific insight, and technological utility with unwavering respect for chemical power.
Ultimately, chlorine trifluoride stands as a testament to the profound impact that molecular structure has on behavior, reactivity, and application. It exemplifies how a small molecule, built from common halogens, can generate extraordinary effects—from igniting materials that resist flame to refining silicon for advanced electronics. Its story intertwines the beauty of chemical theory with the sobering realities of industrial safety. It is a compound that demands caution, precision, and knowledge, embodying the principle that understanding the microscopic world grants immense power—but also immense responsibility.