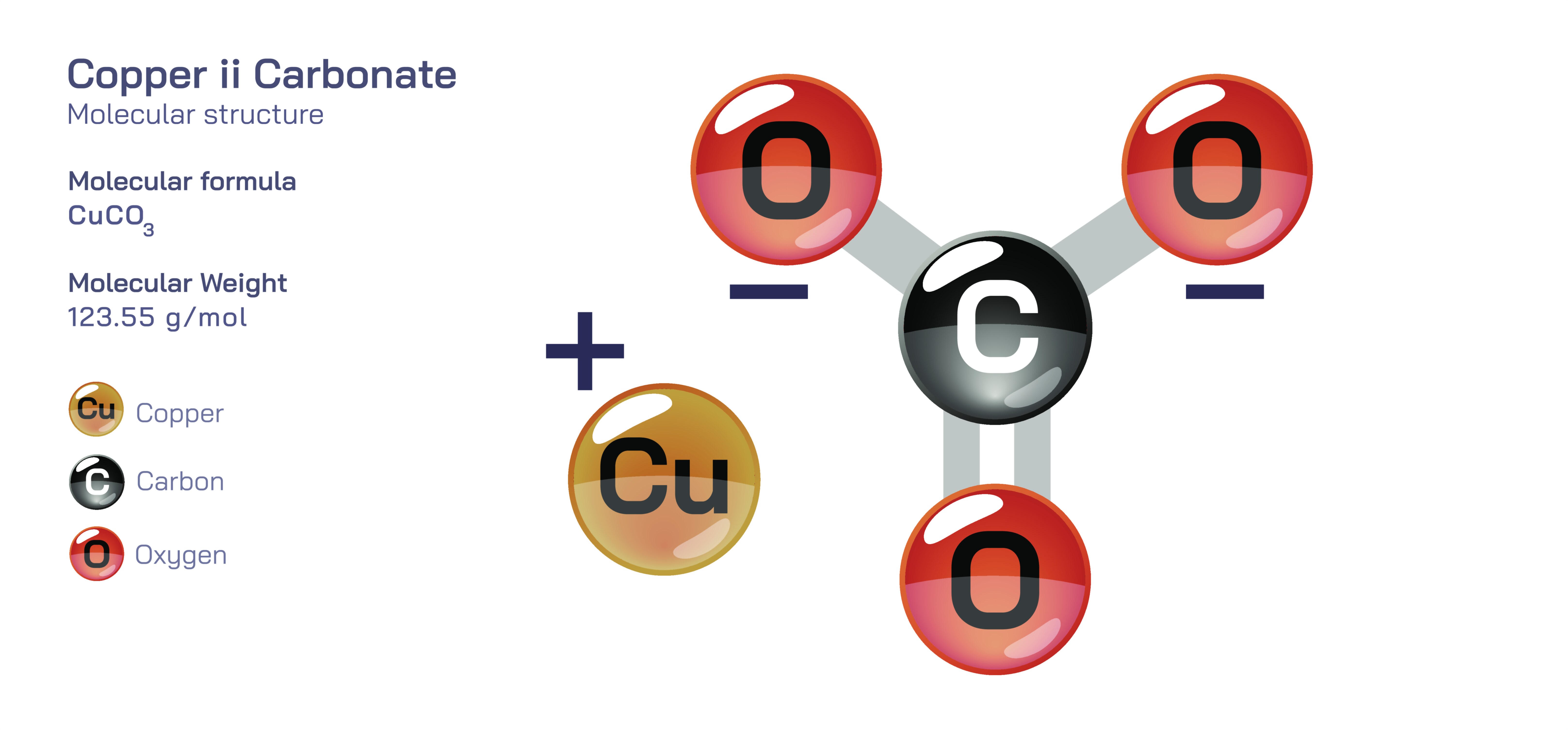
Copper (II) Carbonate — Inorganic Salt Compound
Copper (II) carbonate is an inorganic salt compound that represents one of the most recognizable and historically significant materials associated with copper chemistry, distinguished by its characteristic green to blue-green coloration and its role in natural mineral formation, industrial applications, pigment history, and fundamental chemical science. Often encountered in nature as basic copper carbonates such as malachite and azurite, copper (II) carbonate itself highlights the close interplay between copper ions, carbonate groups, and environmental conditions that shape mineral structures. Although the pure anhydrous form of copper (II) carbonate is rarely found in isolation outside laboratory synthesis, its basic forms dominate geological settings where copper deposits react with carbon dioxide and water in the atmosphere. This compound not only illuminates the principles of mineral chemistry but also provides practical insight into reactivity, decomposition, precipitation, color formation, and environmental transformations. Its relevance spans from ancient art pigments to modern material science, making it an excellent example of how a simple inorganic salt can connect diverse areas of knowledge.
Copper (II) carbonate is typically represented chemically as a material consisting of copper ions in the +2 oxidation state paired with carbonate anions. However, in real-world environments, copper(II) ions tend to form basic carbonates rather than the simple neutral carbonate, especially under aqueous conditions. The two most common natural forms—malachite and azurite—reflect this tendency. Malachite, with its vivid green coloration, consists of copper carbonate hydroxide, while azurite, known for its deep blue tones, also contains hydroxide groups but with a different stoichiometric ratio. These minerals form over long geological timescales when copper-bearing rocks undergo weathering and interact with carbon dioxide-rich groundwater or atmospheric moisture. The presence of hydroxide in these minerals demonstrates how water plays a crucial role in coordinating with copper ions, stabilizing structures, and influencing color. The transformation from azurite to malachite under continued exposure to moisture further illustrates the dynamic nature of copper carbonate chemistry, as small changes in environmental conditions cause shifts in structure, hydration, and visual identity.
The distinctive colors associated with copper carbonates have occupied a central place in human cultural and artistic history. Malachite, in particular, has been used as a pigment for thousands of years, appearing in ancient Egyptian artifacts, medieval manuscripts, Renaissance paintings, and various decorative arts. The compound’s brilliant green hue arises from the interaction between copper ions and the carbonate-hydroxide coordination environment, affecting d-orbital splitting and electronic transitions that determine how visible light is absorbed and reflected. Such color behavior makes copper (II) carbonate a powerful example of ligand field theory, illustrating how the geometry and ligand types around a transition metal ion shape its optical properties. Artists valued malachite for its strong, opaque color, although its sensitivity to acids and weathering meant it required careful handling. Over time, the pigment could undergo transformations, sometimes darkening or degrading if exposed to acidic materials or moisture-rich environments. These limitations helped inspire the development of synthetic pigments in more recent centuries, yet malachite remains an important historical example of copper-based color chemistry.
Laboratory preparation of copper (II) carbonate or its basic forms typically involves the reaction between soluble copper(II) salts such as copper sulfate or copper chloride and a carbonate source like sodium carbonate or potassium carbonate. When these solutions are mixed, a blue-green precipitate forms immediately, reflecting the rapid association between copper ions and carbonate ions. This precipitation serves as a classic demonstration of double displacement reactions and solubility rules in inorganic chemistry. The insolubility of copper (II) carbonate in water forces it out of solution, producing a solid that can be filtered, washed, and dried. The exact composition of the precipitate can vary depending on temperature, pH, concentrations, and the ratio of reactants, making the experiment a platform for exploring how solution conditions influence product formation. The ability of copper carbonate to form complex mixtures with hydroxide also helps illustrate how metal ions interact with carbonate solutions across different pH levels, transitioning between carbonate, bicarbonate, and hydroxide complexes based on environmental conditions.
One of the most notable chemical properties of copper (II) carbonate is its thermal decomposition. When heated strongly, the compound breaks down into copper(II) oxide and carbon dioxide gas. This decomposition is frequently used in educational demonstrations to show how heating can induce chemical transformation, releasing gaseous products that visibly escape or cause bubbling in enclosed setups. The process highlights principles of thermal stability, lattice energy, and the relative strength of metal–oxygen versus metal–carbonate interactions. The copper(II) oxide produced through decomposition is a black solid widely used in ceramics, pigments, and catalysis. This thermal behavior links copper carbonate to a broader family of metal carbonates that decompose into oxides, each with its own temperature threshold and structural characteristics. Understanding this decomposition pathway is essential in fields like materials science, geology, and industrial synthesis where controlled heating processes shape the final material properties.
Copper (II) carbonate also participates in acid-base chemistry in a way that reflects the typical behavior of metal carbonates. When exposed to acids—even weak ones such as acetic acid—the compound reacts vigorously, producing carbon dioxide gas and dissolving to form soluble copper(II) salts. This effervescence offers a clear visual indication that carbonates contain chemically bound carbon dioxide, ready to be released upon acid contact. The reaction is frequently used as a test for carbonate presence in unknown substances. This behavior also has environmental relevance: acidic rainwater interacting with copper-containing materials produces copper salts that dissolve and enter waterways. Understanding these reactions is crucial for evaluating corrosion, degradation, and environmental dispersal of copper compounds. As copper carbonate dissolves under acidic conditions, it transitions through complex aqueous species that influence mobility, bioavailability, and ecological impact.
The environmental chemistry of copper (II) carbonate is especially important because copper is both an essential micronutrient and a potential contaminant. In soils, copper carbonate plays a role in the natural cycling of copper, forming through weathering processes and interacting with organic matter, humic substances, and microbial communities. Its low solubility helps regulate copper availability to plants and microorganisms, preventing toxic accumulation but ensuring adequate supply for enzymatic and metabolic processes. When soils become too acidic, however, copper carbonate dissolves, leading to higher levels of soluble copper that can disrupt biological systems. This dual role—protective under neutral conditions but vulnerable under acidic conditions—illustrates how inorganic salts contribute to environmental balance. Researchers studying soil chemistry and agricultural health often analyze copper carbonate behavior to predict nutrient distribution, contamination risks, and long-term stability of copper-containing fertilizers or pesticides.
Copper (II) carbonate also contributes to understanding corrosion processes, particularly the formation of patina on copper surfaces. Over time, copper exposed to moisture, oxygen, and carbon dioxide gradually transforms into a complex mixture of copper carbonate hydroxides that form a protective green layer known as patina. This weathered coating, seen on statues, roofs, and architectural accents, acts as a natural barrier that slows further corrosion. The patina’s composition varies depending on environmental pollutants, humidity, and exposure patterns, but copper carbonate compounds play a defining role in its characteristic color and protective function. Studying the formation and evolution of patina layers provides insight into long-term metal stability, cultural heritage conservation, and architectural material behavior in outdoor environments.
Industrial applications of copper (II) carbonate are tied to its role as a precursor material. It serves as a starting point for producing copper(II) oxide, copper salts, and various copper-based catalysts. In ceramic manufacturing, its controlled decomposition contributes to glazes and finishes that require specific coloration or chemical properties. In certain chemical syntheses, copper carbonate supplies a slow-release source of copper ions, useful when rapid dissolution is undesirable. Its ability to produce carbon dioxide upon acid contact also makes it significant in reaction pathways where gas evolution influences reaction kinetics, structure formation, or pressure regulation. In all these cases, copper (II) carbonate acts not merely as a passive salt but as a chemically active intermediate capable of transforming under controlled conditions.
Beyond industrial uses, copper carbonate’s optical properties and mineral heritage support scientific research in mineralogy, crystallography, and environmental geochemistry. By studying how copper ions coordinate with carbonate groups in different hydration states and geometries, scientists gain insights into how minerals form, how metals migrate in the earth’s crust, and how environmental factors influence crystal structure. These studies extend to advanced materials research where synthetic analogs or modified copper carbonates help develop functional materials for sensors, coatings, catalysts, or electronic applications. The principles learned from this compound—relating structure, bonding, and reactivity—directly inform the design of new materials with tailored behaviors.
In educational contexts, copper (II) carbonate continues to serve as a versatile demonstration material. Its precipitation reaction helps students visualize how ions form solids; its effervescence with acids illustrates gas evolution; its decomposition into copper oxide highlights thermal transformations; and its distinctive color connects chemical structure to optical behavior. These characteristics make copper carbonate a powerful teaching tool for illustrating general chemistry concepts, helping learners connect theoretical principles to sensory observations. Its ability to bridge mineralogy, coordination chemistry, environmental science, and material synthesis makes it a uniquely rich subject for exploration.
Ultimately, copper (II) carbonate exemplifies the complexity hidden within seemingly simple inorganic compounds. Its existence spans geological, chemical, artistic, environmental, and industrial dimensions, each demonstrating how copper interacts with carbonate ions to produce vivid colors, stable minerals, reactive intermediates, and protective surface coatings. Through its behavior in precipitation, decomposition, coordination, and environmental cycling, copper (II) carbonate reveals the broader themes of inorganic chemistry: the influence of structure on properties, the adaptability of transition metal compounds, and the profound ways in which simple salts shape both natural landscapes and human creations.