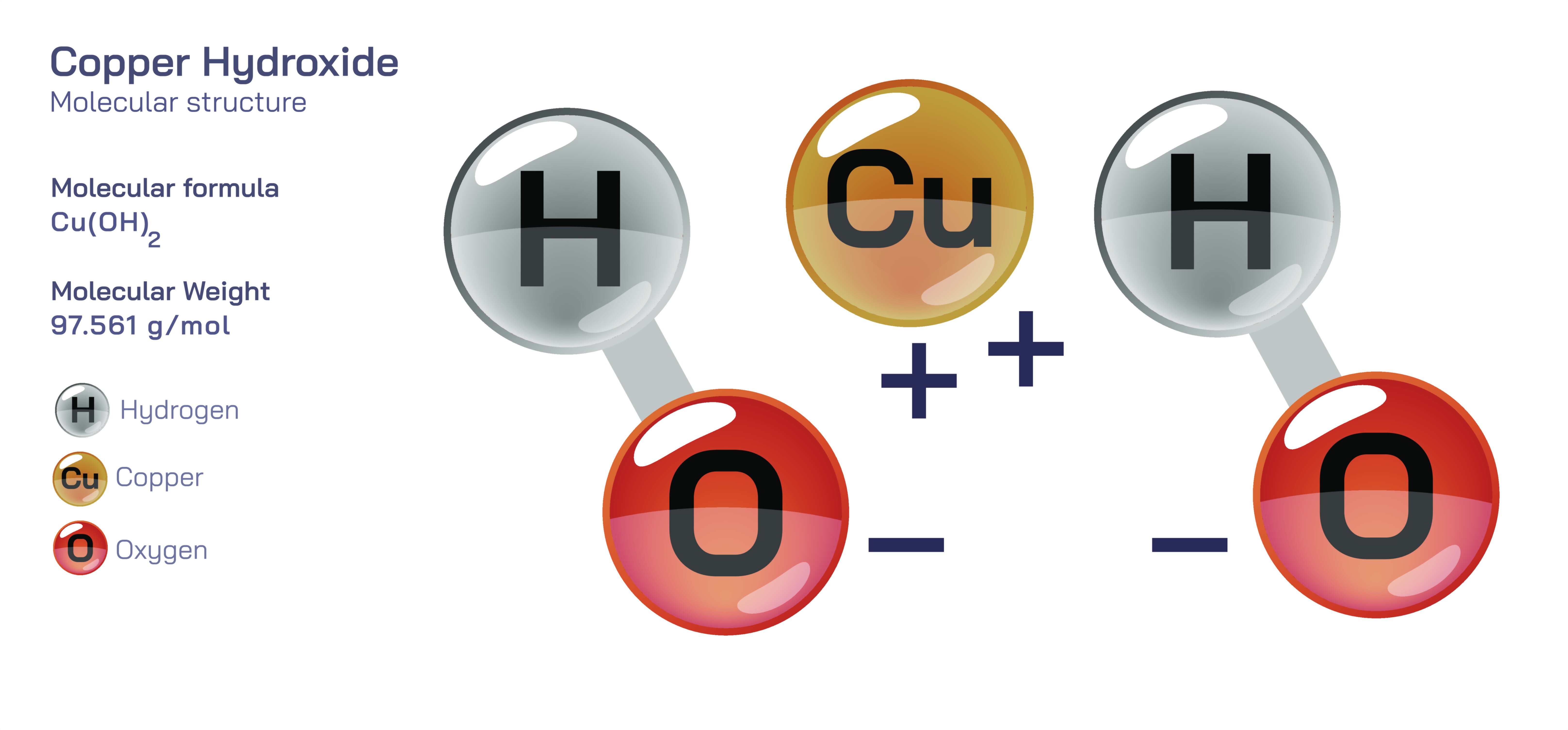Copper Hydroxide — Inorganic Base Compound
Copper hydroxide is an intriguing inorganic base compound that occupies a meaningful place in both classical and modern chemistry due to its distinctive properties, characteristic blue coloration, and participation in a wide range of chemical, environmental, industrial, and biological processes. Represented by the composition involving copper in its +2 oxidation state combined with hydroxide groups, this compound illustrates how transition metals form hydroxides that behave not merely as simple bases but as complex, reactive, and structurally rich materials. Copper hydroxide is typically encountered as a vivid, gelatinous blue solid that forms when soluble copper(II) salts react with alkali hydroxides. Its appearance alone makes it one of the more visually recognizable inorganic substances, but beyond its aesthetic qualities, copper hydroxide demonstrates important concepts of solubility, precipitation, amphoteric interactions, coordination behavior, and redox reactivity. These characteristics make it central to understanding inorganic chemistry, environmental science, metallurgy, catalysis, and even biological systems.
One of the most defining features of copper hydroxide is its low solubility in water. When a solution of copper sulfate or copper chloride is mixed with sodium hydroxide or potassium hydroxide, the copper ions rapidly combine with hydroxide ions to form a gelatinous precipitate. This process visually captures the principle of precipitation reactions, in which ions that were previously dispersed in solution reorganize into an insoluble solid. Students often observe that the formation of copper hydroxide proceeds instantly, highlighting the strong tendency of copper(II) to pair with hydroxide in aqueous environments. The precipitate’s rich blue color, distinct from the deeper blue of many hydrated copper complexes, emerges from the electronic transitions of copper ions interacting with the hydroxide ligands. This color shift reveals how even small changes in the chemical environment surrounding a metal ion can alter its optical properties. In this way, copper hydroxide serves as a vivid demonstration of ligand field effects and the sensitivity of transition metals to coordination geometry.
Although copper hydroxide is classified as a base, its behavior in solution is more nuanced than that of conventional alkali hydroxides. Rather than fully dissociating to produce hydroxide ions, it remains predominantly insoluble, acting more as a weak base and a structural precursor to other copper-containing species. Its interactions with acids demonstrate its basic character: when exposed to acidic conditions, copper hydroxide dissolves, forming soluble copper(II) salts while releasing water. These reactions are often used in laboratory settings to prepare various copper salts in a controlled manner. Conversely, in the presence of strong bases, copper hydroxide can convert into complex ions such as tetrahydroxocuprate species, highlighting its amphoteric tendencies. This dual behavior—reacting with both acids and strong bases—offers insight into how metal hydroxides can occupy intermediate roles between simple bases and more complex coordination compounds. Such amphoterism reflects the adaptability of copper chemistry and emphasizes the relationship between ligand concentration, solubility, and coordination environment.
Copper hydroxide also plays a significant role as an intermediate in redox reactions. Because it contains copper in the +2 oxidation state, it can undergo reduction to copper(I) species or further transformation into copper oxide under heating. When copper hydroxide is gently heated or dehydrated, it converts into black copper(II) oxide, a transformation that reveals how hydroxides can serve as precursors to metal oxides. These decompositions demonstrate thermal stability principles, dehydration pathways, and structural rearrangements within inorganic solids. In reducing environments, copper hydroxide may be converted into copper(I) oxide or even metallic copper, providing accessible examples of redox transitions that highlight how electrons can shift the oxidation state and structural identity of the compound. These redox pathways are foundational for understanding processes such as mineral formation, metallurgy, corrosion, and catalytic cycles involving copper.
In industrial and technological applications, copper hydroxide finds use in areas ranging from agriculture to manufacturing. One of its primary roles is as an active ingredient in fungicides and pesticides. Copper compounds have long been recognized for their antimicrobial properties, and copper hydroxide, with its low solubility and sustained reactivity, is particularly suitable for forming protective coatings on plant surfaces. When applied to crops, it slowly releases copper ions that disrupt fungal growth, making it widely used in agriculture to protect fruits, vegetables, and ornamental plants. Its effectiveness as a fungicide arises from copper’s ability to interfere with microbial enzymes, proteins, and cellular processes. This biological interaction emphasizes that copper hydroxide, although simple in composition, participates in complex biochemical mechanisms that influence plant health and agricultural productivity.
In addition to its agricultural uses, copper hydroxide serves important functions in materials science and composite manufacturing. It can be utilized as a precursor in the synthesis of copper-based catalysts, pigments, and ceramics. By controlling the conditions under which copper hydroxide decomposes or reacts with other compounds, researchers can produce materials with specific structural or catalytic properties. For example, copper hydroxide can be transformed into nano-sized copper oxide particles, which have applications in catalysts, electronics, and antimicrobial coatings. This ability to act as a starting material for advanced functional materials highlights its versatility and importance in synthetic chemistry. In certain polymer formulations, copper hydroxide contributes to flame-retardant behavior or thermal stability, demonstrating how inorganic additives can enhance the performance of composite materials.
Copper hydroxide is also relevant in environmental chemistry, particularly in the context of water treatment and pollution control. Copper compounds interact strongly with organic molecules, suspended solids, and biological matter. In wastewater treatment systems, copper hydroxide can form through reactions between copper-containing effluents and alkaline conditions, altering the mobility, solubility, and environmental impact of copper. Understanding how copper hydroxide behaves in natural water bodies is crucial for predicting its influence on ecosystems. While copper is an essential trace element for plants and animals, elevated concentrations can produce toxic effects, reducing biodiversity and affecting aquatic life. Studying the formation, dissolution, and transformation of copper hydroxide helps environmental scientists evaluate contamination risks, design remediation strategies, and regulate industrial discharge. This environmental dimension underscores the importance of understanding inorganic compounds not only in controlled laboratory settings but also within the complex context of natural ecosystems.
In biological systems, copper hydroxide does not exist as a stable compound, yet its underlying chemical principles help explain how copper interacts within living organisms. Copper ions participate in enzyme catalysis, electron transfer, oxygen transport, and metabolic processes. When copper ions accumulate excessively or interact with cellular components outside regulated pathways, they can induce oxidative stress, damaging proteins, membranes, and genetic material. Studies involving copper hydroxide provide insight into how copper ions bind to biological molecules and how organisms regulate metal concentrations. These insights contribute to fields such as toxicology, biochemistry, and medicine, where metal homeostasis plays a critical role in health and disease. Even though copper hydroxide itself is not a biological species, its chemistry informs our understanding of how copper behaves in environments ranging from soil to living cells.
In laboratory education, copper hydroxide serves as an accessible and visually striking material for demonstrating key chemical principles. Whether illustrating precipitation reactions, demonstrating amphoterism, exploring complex ion formation, or showing thermal decomposition into copper oxide, the compound provides clear and engaging visual evidence of fundamental concepts. Students can observe how solution color, pH, ligand concentration, and temperature influence the identity and behavior of metal compounds. These demonstrations help bridge abstract theoretical concepts with tangible observations, making copper hydroxide a valuable teaching tool. Its sensitivity to conditions also reinforces the idea that chemistry is deeply dependent on environment and context—a principle that resonates across all branches of the discipline.
Copper hydroxide also carries historical significance as one of the copper compounds recognized early for its vivid color and utility. In the past, copper salts contributed to pigments, ceramics, and surface treatments, although modern materials have replaced many of these applications with more stable compounds. Nonetheless, copper hydroxide remains part of the lineage of materials that shaped early chemistry and practical craft. Its ability to impart blue-green tones and its readiness to transform into other copper oxides or basic salts reflect the long-standing interest in copper-based materials across cultures and eras.
Ultimately, copper hydroxide exemplifies the dynamic and interconnected nature of inorganic chemistry. It showcases how a transition metal interacts with hydroxide ligands, how solubility and coordination shape the compound’s behavior, how redox transformations connect one copper species to another, and how a single material can influence agriculture, industry, environmental systems, and scientific study. Its vivid blue color and characteristic reactions make it memorable, but its deeper significance lies in how it bridges fundamental chemical understanding with real-world applications. Through copper hydroxide, one can appreciate not just the properties of a single compound but the broader themes of structure, reactivity, and transformation that define the chemistry of metals in diverse environments.