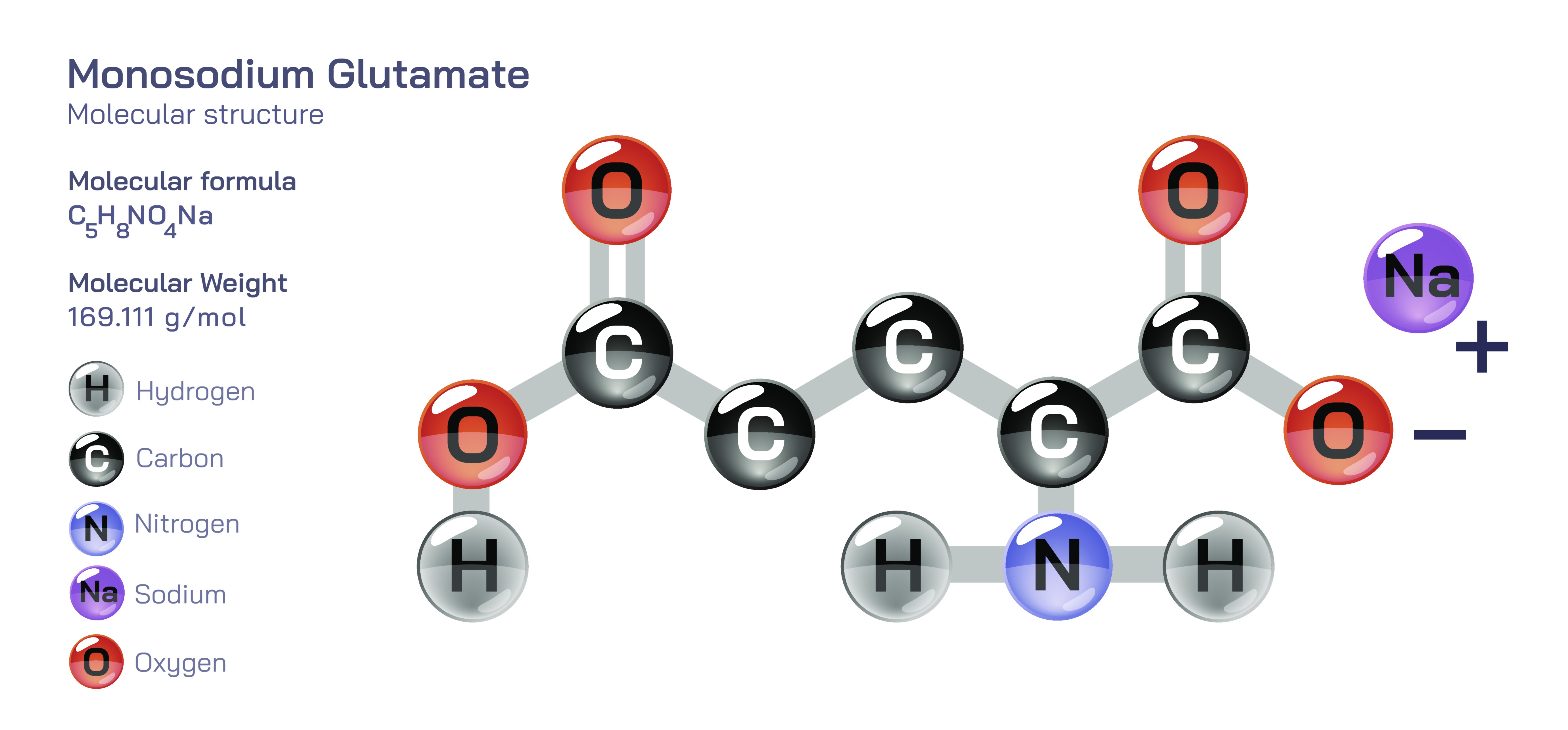
Monosodium Glutamate — Molecular Structure and Composition
Monosodium glutamate, commonly known as MSG, is an organic–inorganic compound whose molecular architecture directly explains its distinctive biochemical behavior, sensory influence on taste, and wide-ranging roles in food science, nutrition, and cellular metabolism. Its chemical formula, C₅H₈NO₄Na, tells only part of the story; the functional properties that distinguish MSG originate from the way its atoms are arranged to form a sodium salt of the amino acid glutamic acid. At the core of the molecule is the glutamate anion, derived from glutamic acid—an amino acid naturally found in proteins—and this anion consists of a five-carbon backbone that includes both an amino group (–NH₂) and two carboxyl groups (–COO⁻). In MSG, one of these carboxyl groups remains negatively charged, while the other exists in a protonated or partially deprotonated state depending on the surrounding pH. The sodium ion bonds ionically to this negatively charged carboxylate group, balancing the charge and generating a crystalline salt that dissolves rapidly in water. Once dissolved, the sodium ion separates completely, allowing the free glutamate anion to interact with taste receptors, enzymes, and metabolic pathways. This dissociation is the pivotal structural event behind MSG’s ability to enhance flavor and participate in cellular chemistry: the sodium atom is neither the source of the taste nor the biologically active portion—the glutamate anion is.
Glutamate’s structure is what enables MSG to stimulate the sensory system in a way distinct from sweet, salty, sour, bitter, and fatty taste sensations. The amino acid contains a side chain that terminates in a carboxylate group, giving it a strongly polar region capable of forming hydrogen bonds, coordinating with water molecules, and fitting precisely into the binding pocket of the umami receptors on the tongue. These receptors—specifically the metabotropic glutamate receptor subtype present in taste buds—are triggered not by sodium but by the stereochemistry of the glutamate molecule itself. When free glutamate from dissolved MSG binds to these receptors, it activates signaling pathways that the brain interprets as savory depth. This taste, called umami, is the same flavor experience produced by natural glutamate released during protein breakdown in foods such as aged cheese, tomatoes, mushrooms, seaweed, fermented products, cured meat, broths, and slow-cooked meats. From a molecular perspective, glutamate’s ability to create umami is not accidental; it arises from the specific arrangement of its functional groups around the carbon backbone, which allows it to interact with sensory proteins that evolved to recognize amino acid–rich foods as nutritious.
Once inside the body, MSG behaves not as an artificial additive but as glutamate—the same molecule produced from digestion of dietary proteins. In the digestive system, proteins are broken down into amino acids, and glutamic acid is one of the most abundant among them. MSG simply provides the glutamate form without requiring enzymatic cleavage from protein chains. The sodium ion separates, contributing to total dietary sodium in a quantity lower per gram than table salt because MSG is only about one-third sodium by mass. The glutamate anion enters cellular metabolism through well-established pathways, especially the glutamate–glutamine cycle, which supports nitrogen shuttling, cellular energy exchange, neurotransmitter balance, and protein synthesis. In the brain, glutamate plays a central role as the most abundant excitatory neurotransmitter, but dietary glutamate from MSG does not cross the blood–brain barrier in healthy biological systems any more readily than glutamate from food proteins. Its fate is largely determined by intestinal cells, which metabolize a large portion of glutamate for energy, and by various tissues that use it in amino acid interconversion. These biochemical processes reflect the structural compatibility between glutamate and metabolic enzymes—a compatibility embedded in the molecule’s composition long before MSG became part of human cuisine.
In food systems, the molecular behavior of MSG extends beyond direct taste stimulation. Free glutamate enhances the perception of body, roundness, and savory depth in foods by intensifying the volatility and perception of aromatic compounds associated with richness and cooked complexity. It also broadens flavor spread across the palate because glutamate interacts with multiple oral receptors and influences salivation, both of which assist in distributing flavors through the mouth. The compound does not create flavor on its own; rather, it amplifies existing savory signals in protein-rich or umami-linked foods. This amplification is a direct reflection of glutamate’s molecular affinity for sensory receptors, and the sodium cation contributes indirectly by supporting dissolution and ionic mobility of the glutamate anion in food matrices. In culinary science, MSG often replaces larger quantities of table salt because it enhances flavor intensity while delivering less sodium overall. This property is once again structural: glutamate provides the majority of the flavor effect, while sodium plays a supporting but necessary ionic role in ensuring the molecule dissolves, stabilizes, and disperses efficiently.
The crystalline structure of MSG reveals how composition influences physical behavior. In its solid state, molecules organize into a lattice in which glutamate anions and sodium ions alternate, each stabilizing the other electrostatically. Water, when present, integrates into the lattice through hydrogen bonding, so MSG may appear as a monohydrate under certain storage conditions, though this does not alter its nutritional, metabolic, or sensory properties. Upon dissolution, the uniform distribution of charged ions produces a solution with high ionic mobility and rapid receptor activation, which is why MSG added to hot broth or sauce begins to influence flavor almost immediately. Because the anion is relatively large and carries multiple hydrogen-bonding sites, glutamate remains fully solvated even at high concentrations and does not form aggregates that reduce taste perception.
The environmentally and biologically benign nature of MSG follows directly from its structure. Glutamate is a universal biological molecule across plants, animals, fungi, and microbes—so widespread that no organism requires adaptation to process it. Plants synthesize glutamate as part of nitrogen assimilation; microbes use it for protein production; animals employ it in metabolism and neurotransmitter cycles. When MSG enters any biological system, the reactions that follow depend not on whether it came from a seasoning crystal, a tomato, or a fermented soy sauce, but on the molecular structure of the glutamate anion. The compound has no residues that bioaccumulate, no halogenated groups, and no persistent environmental structures. Even in wastewater, glutamate is rapidly consumed by microorganisms because it serves as a nutrient rather than a contaminant.
The historical and cultural trajectory of MSG also reflects its structure. The compound was first isolated from seaweed broth—an umami-rich food—because glutamate in free form produces stronger umami response than when it is locked inside protein chains. Fermentation, curing, slow heating, and aging of foods all naturally release free glutamate through protein breakdown, which explains why traditional cuisines across the world gravitated toward umami-rich techniques long before the chemistry of taste was understood. MSG is chemically indistinguishable from the glutamate produced during these processes; the separation of sodium and glutamate in solution means the sensory and metabolic effects originate from the glutamate molecule itself rather than from its source.
As a whole, monosodium glutamate illustrates a timeless rule of chemistry: molecular properties determine macroscopic behavior. The five-carbon backbone bearing an amino and two carboxylate groups dictates how glutamate interacts with water, taste receptors, metabolic enzymes, and cellular absorptive pathways. The single sodium ion modifies solubility without altering biochemical identity. The entire functional spectrum of MSG—from savory flavor enhancement to participation in metabolic cycles—exists because the molecular geometry aligns with biological recognition systems that were shaped by evolution around the chemistry of free amino acids.
In food, in physiology, and in environmental systems, MSG behaves not as an artificial additive but as a structural expression of glutamate—the compound through which living organisms build protein, regulate nitrogen flow, and signal nutritional value. In this way, monosodium glutamate stands as a clear reminder that the effects of a substance are ultimately rooted not in its reputation but in its molecular structure, composition, and the biological logic that responds to it.