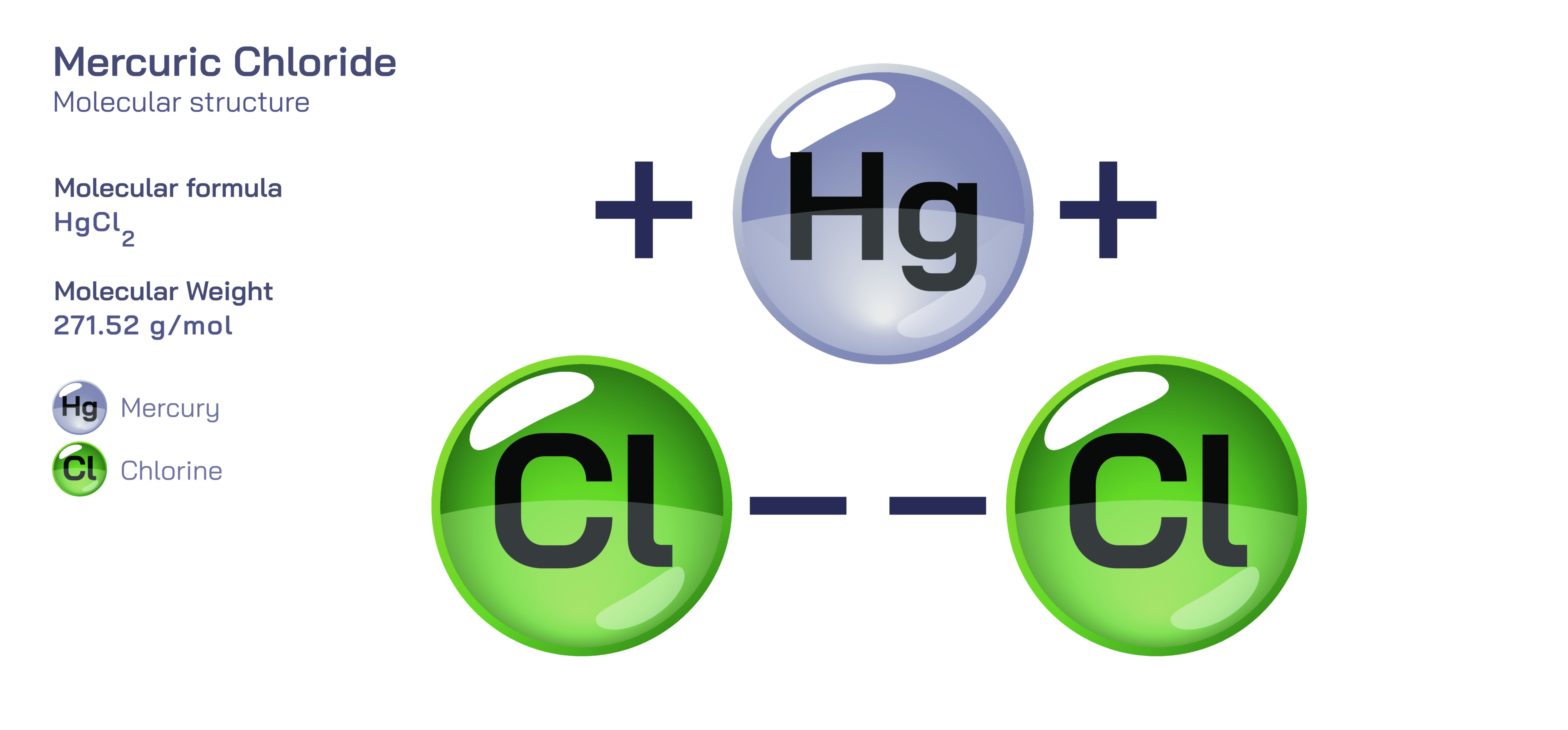Mercuric Chloride — Molecular Structure and Composition
Mercuric chloride is an inorganic compound represented by the formula HgCl₂, and its structural and chemical identity illustrate how the arrangement of atoms in a molecule can shape not only its reactivity and solubility but also its biological interaction, industrial relevance, toxic effects, and historical role in science and medicine. At the molecular level, mercuric chloride consists of one mercury atom in the +2 oxidation state bonded to two chloride ions through linear covalent coordination, giving the entity a strictly linear geometry. Unlike ionic salts such as sodium chloride, HgCl₂ has a strong covalent character in the gas phase and in organic solvents, although in aqueous solution its hydration expands its coordination sphere and allows chloride ions to interact more freely with surrounding water. This hybrid behavior places the molecule between the categories of purely ionic and molecular compounds, explaining its ability to dissolve in polar solvents such as water and alcohol while simultaneously displaying solubility in some organic liquids. The mercury atom, with its high atomic weight and electron-rich d orbitals, contributes to strong polarization of the Hg–Cl bonds, lending the compound high affinity for sulfur-containing biological groups and nucleophilic centers. These interactions later became a key reason why mercuric chloride played an important role in disinfection and chemical preservation, while simultaneously becoming well known as a dangerous toxic compound. The linear configuration of mercury and chloride therefore does not simply describe the shape of the molecule—it determines how mercuric chloride moves between solid, dissolved, and biologically active states.
Unlike many metal chlorides that easily form hydrates, mercuric chloride crystallizes as a stable anhydrous solid under normal conditions, forming flat, prismatic crystals that are heavy, white, and highly corrosive to organic material. On the nanoscale, these crystals are organized by layers of linear HgCl₂ units stacked and stabilized by van der Waals forces, which allow crystalline sheets to slide yet remain cohesive. When placed in water, the strong electronegativity of chloride and the high polarizability of mercury promote a stepwise dissociation process, initially forming solvated HgCl₂, followed by the gradual formation of complex ions such as HgCl₃⁻ and HgCl₄²⁻ when excess chloride is available. These chloride complexes illustrate mercury’s flexibility in coordination chemistry—its valence shell can expand beyond simple twofold linear geometry in aqueous environments, allowing multiple chloride ligands to bind and altering the biological mobility of the compound. This behavior of forming more highly coordinated anions in the presence of chloride-containing fluids partly explains why mercuric chloride spreads efficiently through living tissues after exposure and why its toxicity is not limited to the initial contact point. The molecule’s affinity for biological sulfur also aligns chemically with its structural identity, because mercury is strongly attracted to sulfhydryl (–SH) groups in proteins, enzymes, and cell membranes, forming stable but biologically disruptive bonds that interfere directly with cellular metabolism.
Historically, mercuric chloride was one of the most widely used chemical antiseptics and preservatives because of this mechanism. Long before the molecular basis of toxicity was understood, observers recognized that the compound killed bacteria and prevented the degradation of tissues. For centuries, surgeons, anatomists, and early experimental scientists used mercuric chloride to sterilize tools, disinfect wounds, preserve biological specimens, and even treat certain infections, all under the erroneous assumption that visible antimicrobial effects necessarily meant therapeutic benefit. Only later did research reveal that the same molecular attraction that allowed mercuric chloride to disrupt bacterial enzymes also targeted human enzymes, nerve tissues, and cellular proteins. The linear HgCl₂ molecule was never selective in its killing power; it disabled life via chemical affinity rather than through biological specificity. This realization shifted the compound from a medical tool to a known poison, transforming it from an early disinfectant to a controlled toxic chemical in modern medical and industrial systems.
The compound’s structure and composition also influence its industrial relevance. Mercuric chloride can act as a catalyst in certain organic reactions, particularly those that require activation of alkenes or alkynes through coordination to electrophilic metal centers. The linear mercury center with its exposed electrophilic character allows carbon-based substrates to interact transiently with the metal, lowering activation barriers and enabling transformations that are otherwise sluggish. In the 19th and early 20th centuries, mercuric chloride catalyzed acetylene hydration to produce acetaldehyde—an important industrial reaction at the time—although subsequent advances replaced mercury-based catalysts with safer alternatives after the effects of mercury exposure became undeniable. Additionally, mercuric chloride has been used in metallurgy and electrochemistry because the molecule facilitates chloride transport and electron exchange when in solution. These roles again highlight the underlying logic of the compound’s structure: the way chloride ions coordinate to mercury, and the way mercury accepts electron density, creates the chemical versatility that enables catalytic and electrochemical functionality.
In environmental chemistry, the molecular composition of mercuric chloride plays a profound role in the transformation of mercury species in aquatic and soil systems. Dissolved HgCl₂ is one of the most mobile forms of inorganic mercury, capable of penetrating porous sediments and interacting with microorganisms. When bacterially processed under anaerobic conditions, it can be converted into organic mercury species such as methylmercury, which accumulate through food chains and are far more toxic than the original inorganic salt. The transformation from mercuric chloride into methylmercury is fundamentally rooted in molecular structure—because the HgCl₂ form is soluble, bioavailable, and reactive toward biological ligands, it becomes a precursor to organic mercury buildup in fish, marine mammals, and ultimately humans. For this reason, the environmental regulation of mercuric chloride concerns not only direct toxicity but also its role as a molecular gateway to more dangerous compounds through biological and geochemical processes.
In biological tissues, the mercury atom in HgCl₂ binds strongly to thiol groups in enzymes, blocking their function. Because metabolic processes rely on proteins with precise tertiary and quaternary structures, the formation of mercury-sulfur bonds can alter protein folding, inhibit energy production pathways, and damage cellular membranes. The affects accumulate most strongly in organs that filter or concentrate blood-borne substances, including the kidneys, liver, and central nervous system. Structural knowledge of mercuric chloride illuminates why symptoms of mercury poisoning—such as tremors, neuropathies, gastrointestinal distress, kidney dysfunction, and cognitive impairment—occur when the compound enters the body. The molecule does not need to convert to another form to cause harm; the two chloride ligands merely provide solubility and mobility, while the mercury center does the damage through binding to essential biological structures. Because HgCl₂ molecules or their chloride complexes can cross biological membranes gradually, the compound has a cumulative effect even at low levels of exposure.
Industry and regulatory science eventually recognized that the structural and compositional logic of mercuric chloride makes it unsuitable for casual or widespread application, regardless of its antimicrobial usefulness. As a result, its use has largely shifted to controlled research settings and specialized industrial contexts where trained personnel handle the compound in sealed environments with strict disposal systems. When mercuric chloride does appear in modern laboratories, it is used in trace amounts for highly controlled applications such as analytical chemistry, histological specimen preservation, semiconductor etching, and high-precision chemical synthesis. Even in these environments, the procedures that govern its use focus on minimizing human exposure and environmental release because its toxicity is not degraded by time or simple dilution.
Across these contexts—from crystallographic structure to environmental fate—the identity of mercuric chloride reminds us that chemistry creates consequences not through symbolism but through atomic structure. A straight line of mercury and two chlorides may appear simple on paper, yet the distribution of electrons around that line determines solubility, catalytic behavior, toxicological targeting, and ecological transformation. The compound exists at the intersection of history and science—once valued as a disinfectant and preservative, later recognized as a dangerous toxicant, and now restricted to niches where its utility is weighed carefully against risk. The molecular structure of HgCl₂ is not merely a theoretical model; it is the blueprint that determines how the compound behaves in water, in living organisms, in industrial systems, and in ecosystems.
By understanding mercuric chloride at the structural level, one sees clearly why the molecule is simultaneously scientifically interesting and biologically hazardous. Its composition explains its past role in medicine, its lingering place in industry, and the strict limits required for its safe handling today. It demonstrates that chemistry can produce substances with extraordinary usefulness and extraordinary danger in the same form—and that responsible engagement with such molecules begins by understanding exactly how the arrangement of atoms governs their power in the natural and technological world.