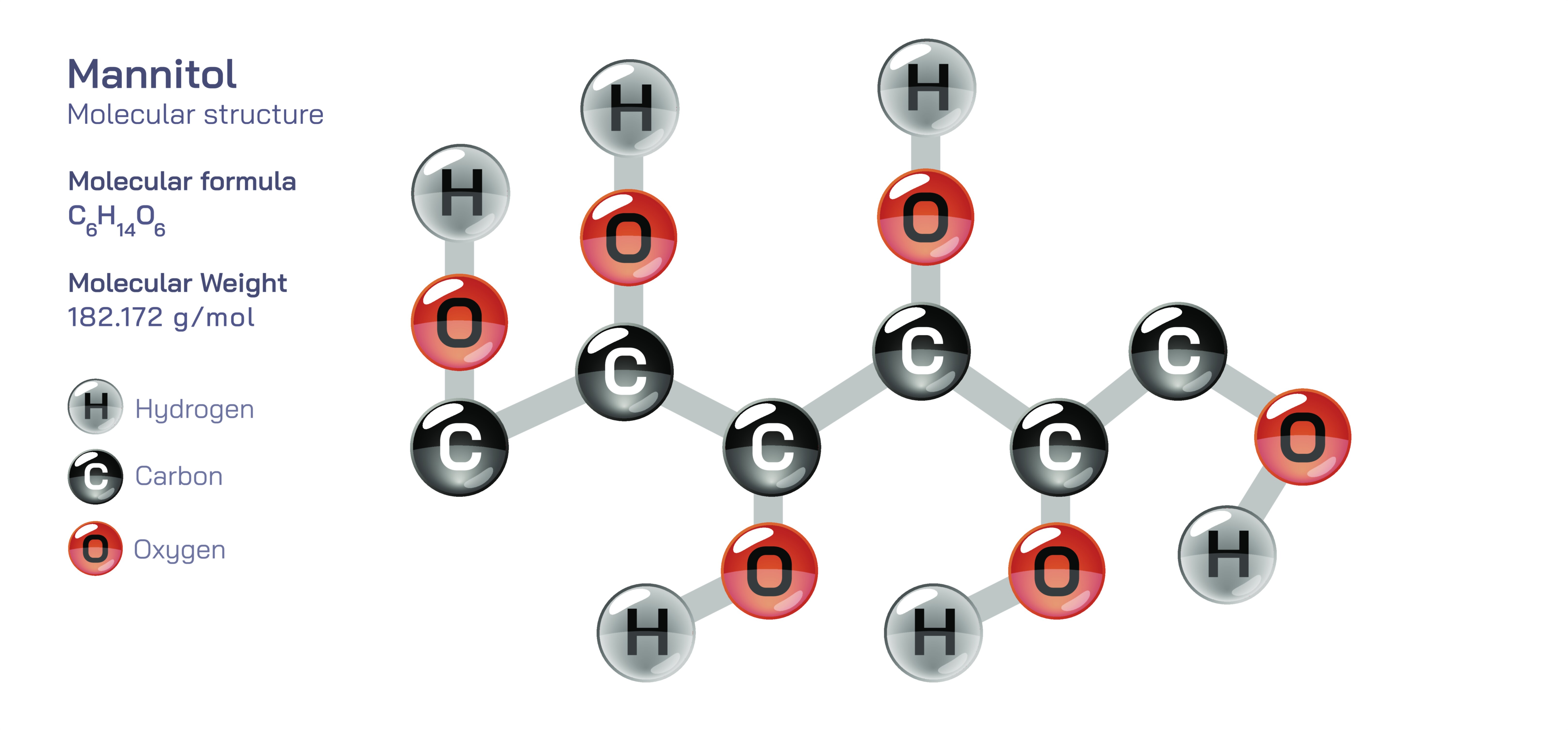
Mannitol — Molecular Structure and Composition
Mannitol is an organic compound of deep biochemical, medical, nutritional, and industrial relevance, and its importance is rooted in the unique architecture of its molecule, which influences not only how it behaves inside the human body but also how it interacts with biological systems, food matrices, pharmaceuticals, and chemical environments. Structurally, mannitol is a polyol, meaning it belongs to the family of sugar alcohols, and its molecular composition consists of six carbon atoms arranged in a linear chain, each bearing a hydroxyl (–OH) group. This gives mannitol the empirical formula C₆H₁₄O₆ and a framework that is closely related to the carbohydrate D-mannose from which it is derived. The spatial arrangement of these hydroxyl groups along the carbon backbone gives the molecule a high density of hydrogen bonding capacity, which in turn governs many of its defining properties such as high water solubility, low reactivity with proteins, chemical stability, and a marked ability to resist metabolism in human tissues. Unlike disaccharides or monosaccharides whose energy yield rapidly increases blood glucose, mannitol is slowly and only partially absorbed in the gastrointestinal tract and does not undergo rapid metabolic oxidation in the liver, which is why it is classified as a low-glycemic sweetener, an osmotic agent, and a non-cariogenic sugar alcohol. The molecular composition of mannitol therefore—six carbons, six hydroxyls, and a straight-chain polyol structure—creates a compound that behaves differently from typical sugars despite tasting sweet and appearing crystalline like them.
Because of its chemical structure, mannitol displays a unique spectrum of physiological and therapeutic functions that extend far beyond nutrition. In medical practice, mannitol is one of the most important osmotic diuretics, meaning it can draw water across biological membranes and increase the volume of fluid excreted through the kidneys. When administered intravenously, mannitol remains largely confined to extracellular fluid compartments and does not cross cell membranes or the blood–brain barrier. This stable extracellular presence generates differences in osmotic pressure that prompt water to move from tissues into the bloodstream, where it can then be filtered out through the kidneys. This property has life-saving applications, particularly in cases of cerebral edema (swelling of the brain) and elevated intracranial pressure following head injury, stroke, or neurosurgical procedures. By reducing water accumulation in brain tissue, mannitol helps lower intracranial pressure without altering electrolyte balance as aggressively as other diuretic interventions. The same osmotic logic applies in ophthalmology, where mannitol is used to lower dangerously high intraocular pressure in acute glaucoma, protecting optic nerves from permanent damage. The molecule’s medical value therefore arises not from caloric energy or nutrient contribution but from the physical consequences of its molecular composition, which make it biologically inert yet osmotically powerful.
Even though mannitol is not a major metabolic fuel, its presence in the diet plays meaningful roles in nutrition and food science. Because humans absorb mannitol inefficiently in the small intestine, a portion reaches the large intestine intact, where gut microbiota can ferment it. This limited digestion makes mannitol significantly lower in calories than sucrose and prevents blood glucose spikes, supporting dietary strategies for individuals managing diabetes, insulin resistance, and conditions requiring controlled carbohydrate intake. Mannitol’s sweetening profile is cooler and less intense than table sugar, lending smooth, refined sweetness to products without contributing to dental caries because oral bacteria do not metabolize it into acids that erode enamel. Its crystalline texture, moisture-binding capacity, and clean mouthfeel have made it a favored ingredient in chewing gums, mints, sugar-free candies, oral healthcare tablets, and specialty bakery products. Because its molecular structure does not brown easily during heating, mannitol is ideal in confectionery applications where heat stability must be maintained without participating in caramelization or Maillard browning. These characteristics reflect how functional properties in food are dictated not simply by sweetness but by intermolecular forces, solubility, and resistance to chemical reactions during preparation and storage.
The stability of mannitol under heat, oxidation, and mechanical stress makes it an invaluable excipient—a compound used not for therapeutic effect but to support the form and delivery of drugs—in pharmaceutical manufacturing. Mannitol can form solid, uniform matrices that stabilize active ingredients during tableting, prevent moisture absorption, and improve dissolution profiles. It commonly appears in chewable tablets and oral-dispersing formulations because its naturally smooth crystalline breakdown produces a pleasant mouthfeel rather than a chalky or gritty sensation. In lyophilized (freeze-dried) injectable medications, mannitol acts as a bulking agent that preserves the structural integrity of delicate biomolecules by promoting a robust glassy matrix around them as water is removed. Many vaccines, protein‐based drugs, enzyme preparations, and monoclonal antibody formulations rely on mannitol for this protective behavior. Once again, these uses arise directly from the molecular capacity of mannitol to form extensive hydrogen bonds and stabilize three-dimensional structures without chemically reacting with active pharmaceutical components.
The biochemical landscape of mannitol extends beyond human use to plants and microorganisms, where it plays an important evolutionary and physiological role. Many plant species produce mannitol as an osmoprotectant—an internal substance that helps regulate water balance and protect cells during environmental stress such as drought, salinity, or cold exposure. Because mannitol does not interfere with cellular metabolism and does not easily crystallize intracellularly, plants use it to manage osmotic pressure while retaining biochemical stability. Fungi, algae, and certain bacteria also synthesize mannitol for similar protective purposes, especially when they encounter oxidative stress or nutrient scarcity. In these organisms, mannitol contributes to antioxidant defense by scavenging reactive oxygen species, which prevents cellular damage during harsh environmental conditions. These natural functions highlight how a structural feature as simple as evenly distributed hydroxyl groups can give a molecule the versatility to act as a sweetener in human food, a medicine in hospitals, and a survival compound in living organisms experiencing stress.
Despite its many benefits, mannitol requires thoughtful handling in diet and medicine. When consumed in excessive quantities—especially in individuals with sensitive digestion or imbalanced gut microbiota—mannitol can exert the same osmotic forces in the intestines that make it useful medically, leading to gastrointestinal discomfort, bloating, or laxative effects. For most people, these effects appear only when intake exceeds normal consumption levels, but they underscore how even a nutritionally gentle molecule can behave differently depending on concentration and context. In clinical settings, careful dosing is essential because mannitol shifts fluid volumes between biological compartments, and improper administration can stress the cardiovascular system or the kidneys in vulnerable patients. Yet when used correctly, these same properties make mannitol indispensable in treating critical conditions such as traumatic brain injury, acute glaucoma, and cases requiring rapid osmotic intervention.
From the perspective of molecular science, the influence of mannitol across so many fields underscores a profound truth about chemistry: small differences in atomic arrangement ripple outward into large differences in how matter behaves. Mannitol is isomeric with sorbitol—another six-carbon sugar alcohol—with the same atomic formula but a slightly different orientation of a single hydroxyl group. That tiny distinction alters melting point, sweetness profile, digestive absorption, industrial compatibility, pharmaceutical performance, and metabolic response. The lesson is that the composition of a molecule tells only part of the story; it is the precise three-dimensional placement of atoms within that composition that determines function. Mannitol, through its linear carbon backbone dotted with six hydroxyl groups in a specific stereochemical pattern, demonstrates that molecular structure orchestrates everything from osmotic balance and therapeutic effect to the sensory qualities of confectionery and the stability of fragile biomedicines.
In every setting where it appears—hospital IV lines, sugar-free snacks, freeze-dried drug vials, dentally safe chewing gums, plant stress-response pathways, and microbial biochemistry—mannitol proves that a molecule is not defined by where it exists, but rather by the internal logic of its atoms and bonds. Its composition enables gentle sweetness without metabolic strain, promotes predictable osmotic behavior in medical emergencies, supports the physical resilience of pharmaceuticals, and protects living cells under harsh environmental changes. Whether viewed through the lens of medicine, nutrition, industry, or biology, mannitol stands as a compelling example of how the design of a molecule quietly shapes its performance and, ultimately, its value across the diverse systems of life and technology.