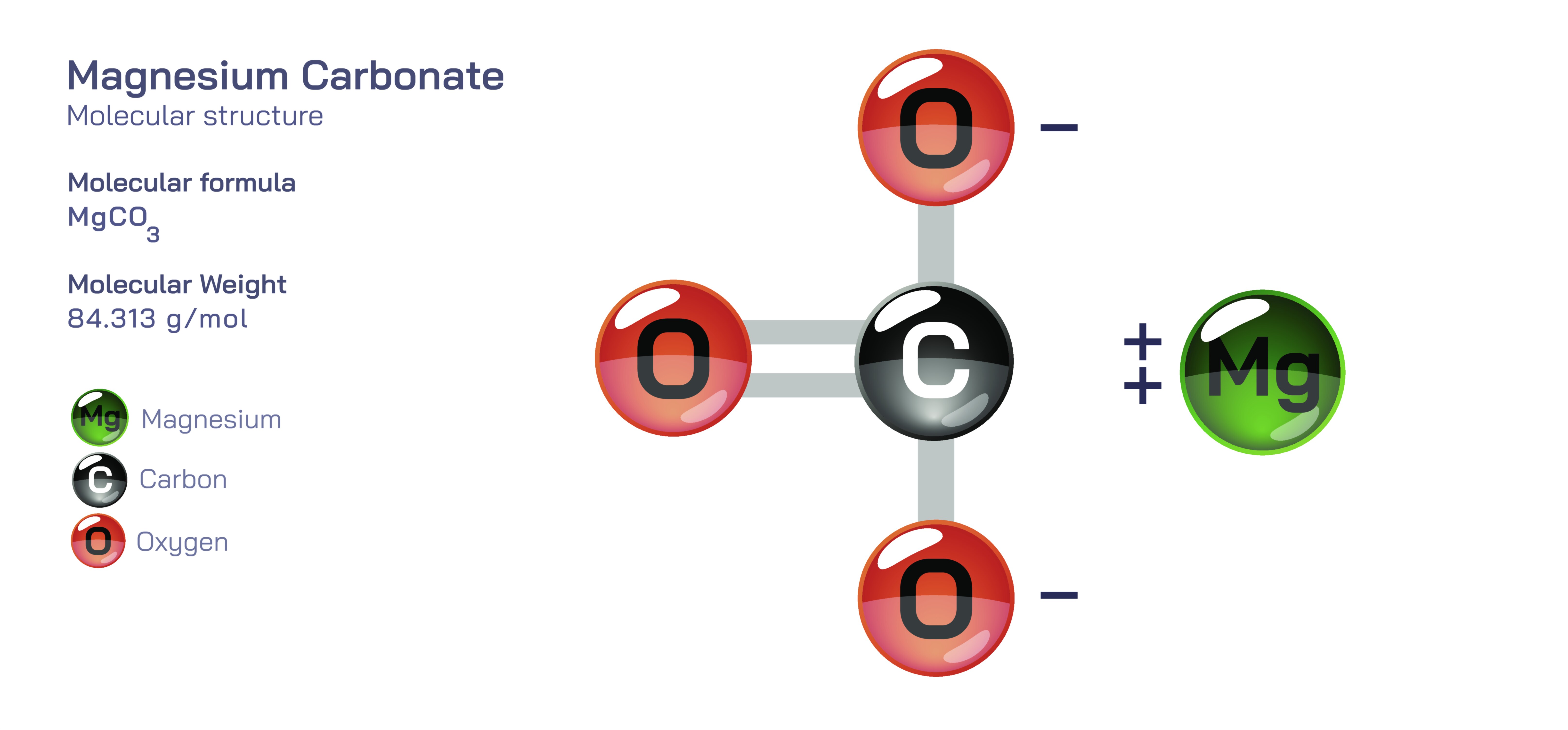
Magnesium Carbonate — Chemical Structure, Physical and Chemical Properties, Natural Occurrence, Industrial Relevance, Biological Roles, and Applications Across Medicine, Sports, Manufacturing, and Environmental Science
Magnesium carbonate is an inorganic compound composed of magnesium cations and carbonate anions, forming a salt that occurs both naturally and synthetically in many physical forms, each with different textures, densities, and industrial uses. Although it is often associated with the white chalk-like powder seen in sports and climbing activities, magnesium carbonate encompasses a broader scientific and technological importance. The compound appears in minerals such as magnesite and dolomite, contributes to soil chemistry, participates in geological processes, aids human metabolic function, and forms the basis of several industrial and pharmaceutical products. Its behaviour reflects the combined properties of magnesium, an alkaline earth metal with strong ionic tendencies, and carbonate, a group known for buffering and structural rigidity. When these ions bond, they create a compound with relatively low solubility, moderate thermal reactivity, and a degree of chemical versatility, allowing magnesium carbonate to serve as both a functional filler and an active chemical agent depending on the environment in which it is used.
Structurally, magnesium carbonate forms a crystalline lattice based on the attraction between positively charged magnesium ions and negatively charged carbonate groups. In nature, this arrangement most commonly appears in the mineral magnesite, which is a dense and stable crystalline form. However, when produced synthetically or subjected to rapid precipitation, magnesium carbonate also forms porous or amorphous variations that contain small amounts of water and develop into light, airy powders. These structural variations largely determine texture and performance. Dense magnesite behaves like a rock and is used in refractory and industrial applications because it withstands high temperatures without deforming. Light basic magnesium carbonate, in contrast, forms a fine white powder with a large surface area, making it ideal for absorbing moisture and improving grip in athletics. Between these two extremes are granular, hydrated, and industrially modified forms that support plastics manufacturing, chemical processing, ceramics, fireproofing, and pharmaceutical formulations.
Magnesium carbonate is characterized by limited solubility in water, but this solubility increases slightly in the presence of carbon dioxide, and it decreases under alkaline conditions. When exposed to acidic conditions, magnesium carbonate reacts readily with acid to form magnesium salts while releasing carbon dioxide. This behaviour explains why magnesium carbonate acts as an antacid: it neutralizes excess stomach acidity while gently releasing gas and water. Technically, this reactivity is not violent or corrosive and is therefore suitable for internal consumption under medical supervision. In geological environments, the reaction of magnesium carbonate with slightly acidic groundwater influences the formation of caves, mineral deposits, and carbonate-based rock systems over millions of years. Under high heat, magnesium carbonate decomposes to form magnesium oxide while releasing carbon dioxide. This thermal transformation is critical for the production of refractory materials because magnesium oxide has excellent resistance to extremely high temperatures, thermal shock, and corrosive environments.
The physical properties of magnesium carbonate also contribute to its famous role in sports and physical training. Gymnasts, rock climbers, weightlifters, and pole vaulters use powdered magnesium carbonate as chalk to improve grip and prevent slipping. The compound absorbs sweat and moisture from the skin while providing a dry surface that increases friction between the hand and equipment. Unlike talc or baby powder, magnesium carbonate does not create a slippery coating because its microscopic texture clings to the skin rather than lubricating it. In addition, because of its lightweight structure and large surface area, it spreads easily across the skin and remains effective without heavy application. Safety also contributes to its popularity; because the compound is chemically mild and poses minimal toxicity when inhaled or in contact with skin, it has become the universal grip aid in athletics and climbing.
Magnesium carbonate has important uses in medicine and nutritional health as well. The compound functions as an antacid in the treatment of occasional heartburn and indigestion. By neutralizing excessive stomach acidity, it helps relieve discomfort and restores digestive balance without abrupt or harsh action. In nutritional supplements, magnesium carbonate serves as a source of dietary magnesium, an essential mineral required for muscle function, nerve signaling, bone health, and energy metabolism. The carbonate form provides magnesium in a stable powder that is easily formulated into capsules, tablets, and chewables. Although it does not dissolve rapidly in the stomach compared with other salts, its slow conversion into bioavailable magnesium can benefit users who require sustained mineral delivery rather than an immediate spike. In pharmaceutical manufacturing, magnesium carbonate is also used as a filler and flow agent to improve tablet consistency, because its fine particle structure prevents clumping and ensures smooth blending.
In industrial applications, magnesium carbonate plays several roles according to its density and structural form. Dense magnesite becomes a key raw material for fire-resistant refractories used in steelmaking, furnaces, kilns, and high-temperature insulation. When converted into magnesium oxide, it contributes to materials that protect factory linings and industrial heating equipment from corrosion and thermal shock. In the plastics and rubber industries, magnesium carbonate acts as a reinforcing filler, improving mechanical strength, reducing shrinkage, and increasing heat tolerance. It also improves flame resistance by releasing carbon dioxide when heated. In the construction sector, magnesium carbonate and its derivatives appear in environmentally oriented cement formulations and fireproof panels, where thermal stability and mineral structure contribute to structural safety.
Environmental science and geology also rely on magnesium carbonate as a major participant in the global carbon cycle. Natural deposition of magnesium carbonate in rocks and sediments removes carbon dioxide from the environment and stores it in stable geological formations. This is one reason carbonate rocks across Earth hold massive archives of atmospheric carbon from past geological eras. Because magnesium carbonate can be formed by reaction between magnesium-rich brines and carbon dioxide, researchers are studying its potential role in carbon sequestration strategies aimed at combating climate change. If engineered efficiently, these processes could permanently store carbon in mineral form, locking it away from the atmosphere.
Magnesium carbonate demonstrates a remarkable combination of chemical stability and selective reactivity, allowing it to function as a material that is simultaneously gentle to biological systems and robust in industrial environments. It does not trigger the harsh corrosive or toxic reactions associated with many metal compounds, and yet it transforms into magnesium oxide—which is critical to high-temperature metallurgy—when heated. It is soft enough to cling to skin for athletic grip, yet strong enough to contribute to the production of structural ceramic and refractory materials. It supports digestion and metabolic balance in the human body while also enabling the manufacture of plastics and fireproof construction materials. Viewed through these lenses, magnesium carbonate is not simply a white powder but a compound with broad relevance that stretches across sports, medicine, geology, manufacturing, nutrition, and climate science.
At its most fundamental level, magnesium carbonate represents the intersection of physical mineral structure, chemical adaptability, and practical utility. Its versatility comes from the nature of its ionic bond and its predictable behaviour under heat, moisture, pressure, and pH change. Whether supporting an athlete’s grasp on a barbell, protecting a furnace from extreme temperatures, strengthening a material under mechanical stress, supplementing essential minerals in the human body, or participating in long-term environmental cycles, magnesium carbonate illustrates how a single chemical compound can integrate into many aspects of human life and natural processes.